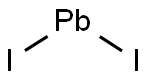
Lead(II) iodide
- CAS No.
- 10101-63-0
- Chemical Name:
- Lead(II) iodide
- Synonyms
- PbI2;Super PbI2;Lead(II) diiodide;LT-S9147;diiodolead;LEAD IODIDE;LEAD DIIODIDE;LEAD(+2)IODIDE;plumbousiodide;LEAD(II) IODIDE
- CBNumber:
- CB2771616
- Molecular Formula:
- I2Pb
- Molecular Weight:
- 461.01
- MOL File:
- 10101-63-0.mol
- MSDS File:
- SDS
- Modify Date:
- 2024/3/26 17:16:27
| Melting point | 402 °C(lit.) |
|---|---|
| Boiling point | 954 °C(lit.) |
| Density | 6.16 g/mL at 25 °C(lit.) |
| Flash point | 954°C |
| storage temp. | Keep in dark place,Inert atmosphere,Room temperature |
| solubility | Soluble in concentrated solutions of alkali iodides and sodium thiosulfate. Insoluble in alcohol and cold hydrochloric acid. |
| form | beads |
| color | Yellow to orange |
| Specific Gravity | 6.16 |
| Water Solubility | Partially soluble in water. Freely soluble in sodium thiosulfate solution. Soluble in concentrated solutions of alkali iodides. Insoluble in alcohol and cold HCl.Soluble in concentrated solutions of alkali iodides and sodium thiosulfate. Insoluble in alcohol and cold hydrochloric acid. |
| Hydrolytic Sensitivity | 0: forms stable aqueous solutions |
| λmax | 539nm(neat)(lit.) |
| Sensitive | Light Sensitive |
| Merck | 14,5411 |
| Solubility Product Constant (Ksp) | pKsp: 8.01 |
| Exposure limits |
ACGIH: TWA 0.05 mg/m3; TWA 0.01 ppm NIOSH: IDLH 100 mg/m3; TWA 0.050 mg/m3 |
| Stability | Stable. May discolour upon exposure to light. |
| InChIKey | RQQRAHKHDFPBMC-UHFFFAOYSA-L |
| CAS DataBase Reference | 10101-63-0(CAS DataBase Reference) |
| NIST Chemistry Reference | Lead diiodide(10101-63-0) |
| EPA Substance Registry System | Lead(II) iodide (10101-63-0) |
SAFETY
Risk and Safety Statements
| Symbol(GHS) |    GHS07,GHS08,GHS09 |
|||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Signal word | Danger | |||||||||
| Hazard statements | H302+H332-H360Df-H373-H410 | |||||||||
| Precautionary statements | P201-P202-P273-P301+P312-P304+P340+P312-P308+P313 | |||||||||
| Hazard Codes | T,N | |||||||||
| Risk Statements | 61-20/22-33-50/53-62 | |||||||||
| Safety Statements | 53-45-60-61 | |||||||||
| RIDADR | UN 2291 6.1/PG 3 | |||||||||
| WGK Germany | 3 | |||||||||
| F | 8 | |||||||||
| TSCA | Yes | |||||||||
| HazardClass | 6.1 | |||||||||
| PackingGroup | III | |||||||||
| HS Code | 28276000 | |||||||||
| NFPA 704 |
|
Lead(II) iodide price More Price(20)
| Manufacturer | Product number | Product description | CAS number | Packaging | Price | Updated | Buy |
|---|---|---|---|---|---|---|---|
| Sigma-Aldrich(India) | 900168 | Lead(II) iodide 99.999% trace metals basis, perovskite grade | 10101-63-0 | 5G | ₹13996.73 | 2022-06-14 | Buy |
| Sigma-Aldrich(India) | 554359 | Lead(II) iodide AnhydroBeads?, ?10?mesh, 99.999% trace metals basis | 10101-63-0 | 5G | ₹10262.1 | 2022-06-14 | Buy |
| Sigma-Aldrich(India) | 211168 | Lead(II) iodide 99% | 10101-63-0 | 50G | ₹7231.1 | 2022-06-14 | Buy |
| Sigma-Aldrich(India) | 203602 | Lead(II) iodide 99.999% trace metals basis | 10101-63-0 | 50G | ₹17796.3 | 2022-06-14 | Buy |
| ALFA India | ALF-057103-30 | Lead(II) iodide, 98.5% | 10101-63-0 | 250g | ₹28218 | 2022-05-26 | Buy |
Lead(II) iodide Chemical Properties,Uses,Production
Uses
Lead(II) iodide is used as a detector material for high energy photons including x-rays and gamma rays. It is used in photography, printing, mosaic gold, and bronzing. It exhibits ferroelastic properties and has efficiency in stopping X-ray and gamma ray, which provides excellent environmental stability.
Preparation
Lead diiodide is prepared by mixing aqueous solutions of lead nitrate or lead acetate with an aqueous solution of potassium or sodium iodide or hydriodic acid, followed by crystallization. The product is purified by recrystallization.
Pb2+(aq) + 2Iˉ (aq) → PbI2(s).
General Description
Lead iodide appears as a yellow crystalline solid. Insoluble in water and denser than water. Primary hazard is threat to the environment. Immediate steps should be taken to limit spread to the environment. Used in printing and photography, to seed clouds and other uses.
Air & Water Reactions
Slightly water soluble.
Reactivity Profile
Lead(II) iodide has weak oxidizing or reducing powers. Redox reactions can however still occur. The majority of compounds in this class are slightly soluble or insoluble in water. If soluble in water, then the solutions are usually neither strongly acidic nor strongly basic. These compounds are not water-reactive. Light sensitive
Hazard
Lead diiodide is toxic if ingested. The symptoms are those of lead poisoning.
Health Hazard
Early symptoms of lead intoxication via inhalation or ingestion are most commonly gastrointestinal disorders, colic, constipation, etc.; weakness, which may go on to paralysis, chiefly of the extensor muscles of the wrists and less often the ankles, is noticeable in the most serious cases. Ingestion of a large amount causes local irritation of the alimentary tract. Pain, leg cramps, muscle weakness, paresthesias, depression, coma, and death may follow in 1 or 2 days. Contact with eyes causes irritation.
Potential Exposure
Lead iodide is used in bronzing, gold pencils; mosaic gold; printing, and photography
Shipping
UN3288 Toxic solids, inorganic, n.o.s., Hazard Class: 6.1; Labels: 6.1-Poisonous materials, Technical Name Required. UN3077 Environmentally hazardous substances, solid, n.o.s., Hazard class: 9; Labels: 9-Miscellaneous hazardous material, Technical Name Required
Purification Methods
It crystallises from a large volume of water. The solubility in H2O is 1.1% at ~10o, and 3.3% at ~ 100o.
Incompatibilities
Lead iodide has weak oxidizing or reducing powers. Redox reactions can however still occur. The majority of compounds in this class are slightly soluble or insoluble in water. If soluble in water, then the solutions are usually neither strongly acidic nor strongly basic. These compounds are not water-reactive. Light sensitive Contact with oxidizers or active metals may cause violent reaction
Lead(II) iodide Preparation Products And Raw materials
| Supplier | Tel | Country | ProdList | Advantage | Inquiry |
|---|---|---|---|---|---|
| GLR Innovations | +91 9891111994 | New Delhi, India | 4542 | 58 | Inquiry |
| QUALIKEMS FINE CHEM PVT LTD | +911123618475 | New Delhi, India | 51 | 58 | Inquiry |
| Infinium Pharmachem Limited | +91-2692238849 +91-2692238850 | Gujarat, India | 65 | 58 | Inquiry |
| Techno Pharmchem | 91-9818265860 | Delhi, India | 173 | 58 | Inquiry |
| Qualikems Fine Chem Pvt., Ltd. | 91-265-2841531 | Gujarat, India | 54 | 58 | Inquiry |
| Oxford laboratories | +91 250 239 0032 | Maharashtra, India | 652 | 58 | Inquiry |
| TCI Chemicals (India) Pvt. Ltd. | 1800 425 7889 | New Delhi, India | 6778 | 58 | Inquiry |
| Otto Chemie Pvt. Ltd. | +91 9820041841 | Mumbai, India | 5873 | 58 | Inquiry |
| Alfa Aesar | 1 800 209 7001 | Maharashtra, India | 6913 | 58 | Inquiry |
| Dr. J. Pharmachem India | -27472871.01 | Pune, India | 23 | 58 | Inquiry |
| Supplier | Advantage |
|---|---|
| GLR Innovations | 58 |
| QUALIKEMS FINE CHEM PVT LTD | 58 |
| Infinium Pharmachem Limited | 58 |
| Techno Pharmchem | 58 |
| Qualikems Fine Chem Pvt., Ltd. | 58 |
| Oxford laboratories | 58 |
| TCI Chemicals (India) Pvt. Ltd. | 58 |
| Otto Chemie Pvt. Ltd. | 58 |
| Alfa Aesar | 58 |
| Dr. J. Pharmachem India | 58 |
10101-63-0(Lead(II) iodide)Related Search:
1of4
chevron_right




