Vanadyl sulfate
- CAS No.
- 27774-13-6
- Chemical Name:
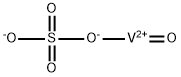
- Vanadyl sulfate
- Synonyms
- ci77940;vanadyl;Vandanyl;c.i.77940;VANDIN(IV);Chebi:33146;Vanadyl sulfat;Vanadyl sulfate;VVanadyl Sulfate;oxysulfato-vanadiu
- CBNumber:
- CB3252402
- Molecular Formula:
- O5SV
- Molecular Weight:
- 163
- MOL File:
- 27774-13-6.mol
- MSDS File:
- SDS
- Modify Date:
- 2024/3/13 13:40:15
| Melting point | 105°C |
|---|---|
| Density | 2.06[at 20℃] |
| solubility | soluble in H2O |
| form | Solid |
| color | Blue |
| Water Solubility | soluble |
| Stability | Stable. Incompatible with strong oxidizing agents. |
| LogP | -1.031 (est) |
| CAS DataBase Reference | 27774-13-6(CAS DataBase Reference) |
| EPA Substance Registry System | Vanadyl sulfate (27774-13-6) |
SAFETY
Risk and Safety Statements
| Symbol(GHS) |  GHS07 |
|||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Signal word | Warning | |||||||||
| Hazard statements | H315-H319 | |||||||||
| Precautionary statements | P264-P280-P302+P352-P337+P313-P305+P351+P338-P362+P364-P332+P313 | |||||||||
| Risk Statements | 22-36/38 | |||||||||
| Safety Statements | 26-36/37/39 | |||||||||
| RIDADR | UN 2931/9152 | |||||||||
| NFPA 704 |
|
Vanadyl sulfate price More Price(2)
Vanadyl sulfate Chemical Properties,Uses,Production
Chemical Properties
Vanadyl sulfate is a pale blue crystalline powder. It is odorless and very soluble in water. It is denser than water. Contact may irritate skin, eyes, and mucous membranes. It may be toxic if ingested, inhaled, or absorbed through the skin.
Physical properties
The dihydrate, VOSO4.2H2O is a blue black crystalline powder, soluble in water.
Uses
The dihydrate is a mordant in dyeing and printing fabrics; used in preparing aniline black; a colorant in ceramics to form blue and green glazes; used in making colored glass; and a reducing agent.
Preparation
Vanadyl sulfate is prepared by passing sulfur dioxide through a cold solution of vanadium pentoxide in sulfuric acid, followed by crystallization:
V2O5 + H2SO4 + H2O + SO2 → 2VOSO4 + 2H2O
General Description
Vanadyl sulfate is a sulfate of vanadium that occurs naturally as the mineral pauflerite. It is a common source of vanadium in the laboratory. Vanadyl sulfate is an intermediate in the extraction of vanadium from petroleum residues and is also a component of some food supplements and drugs. Vanadium is a transition metal with the chemical symbol V and atomic number 23. The element usually combines with other elements such as oxygen, sodium, sulfur, or chloride, and occurs naturally in about 65 different minerals and in fossil fuel deposits. Vanadium is found in many organisms, and is used by some life forms as an active center of enzymes. (L837, L838, L852)
Reactivity Profile
Vanadyl sulfate has weak oxidizing or reducing powers. Redox reactions can however still occur.
Health Hazard
Highly toxic, may be fatal if inhaled, swallowed or absorbed through skin. Avoid any skin contact. Effects of contact or inhalation may be delayed. Fire may produce irritating, corrosive and/or toxic gases. Runoff from fire control or dilution water may be corrosive and/or toxic and cause pollution.
Fire Hazard
Non-combustible, substance itself does not burn but may decompose upon heating to produce corrosive and/or toxic fumes. Containers may explode when heated. Runoff may pollute waterways.
Safety Profile
A poison and an inhalation hazard. Poison by intravenous, intraperitoneal, and subcutaneous routes. Mutation data reported. When heated to decomposition it emits toxic fumes of VOx and SOx. See also SULFATES and VANADIUM COMPOUNDS.
Potential Exposure
Vanadyl sulfate is used as a fixative for textile dyes, a colorant for glass and ceramics; a reducing agent and a catalyst.
Shipping
UN2931 Vanadyl sulfate, Hazard Class: 6.1; Labels: 6.1-Poisonous materials.
Incompatibilities
Vanadyl sulfate has weak oxidizing or reducing powers. Redox reactions can however still occur. Incompatible with oxidizers (chlorates, nitrates, peroxides, permanganates, perchlorates, chlorine, bromine, fluorine, etc.); contact may cause fires or explosions. Keep away from alkaline materials, strong bases, strong acids, oxoacids, epoxides. Vanadyl sulfate may attack metals; sulfates react with aluminum, magnesium.
Vanadyl sulfate Preparation Products And Raw materials
| Supplier | Tel | Country | ProdList | Advantage | Inquiry |
|---|---|---|---|---|---|
| ShanPar Industries Pvt Ltd | +91-2652638973 +91-9909901206 | Gujarat, India | 115 | 58 | Inquiry |
| Svaks Biotech India Pvt Ltd | +91-7709222999 +91-7767806001 | Maharashtra, India | 81 | 58 | Inquiry |
| Otto Chemie Pvt. Ltd. | +91 9820041841 | Mumbai, India | 5873 | 58 | Inquiry |
| Omkar Speciality Chemicals Ltd | + 91-251-2697347 | Maharashtra, India | 75 | 58 | Inquiry |
| Triveni chemicals | 08048762458 | New Delhi, India | 6093 | 58 | Inquiry |
| Veera Chemicals | 08048372076Ext 176 | Maharashtra, India | 2 | 58 | Inquiry |
| Shanghai Daken Advanced Materials Co.,Ltd | +86-371-66670886 | China | 16209 | 58 | Inquiry |
| Henan Tianfu Chemical Co.,Ltd. | +86-0371-55170693 +86-19937530512 | China | 21675 | 55 | Inquiry |
| Hangzhou FandaChem Co.,Ltd. | 008657128800458; +8615858145714 | China | 9338 | 55 | Inquiry |
| Hefei TNJ Chemical Industry Co.,Ltd. | +86-0551-65418679 +86-18949832763 | China | 2989 | 55 | Inquiry |
27774-13-6(Vanadyl sulfate )Related Search:
1of4
chevron_right




