Abacavir
- CAS No.
- 136470-78-5
- Chemical Name:
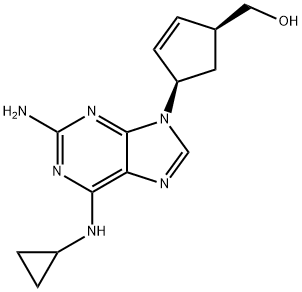
- Abacavir
- Synonyms
- CS-107;1592u89;Abacavir;Abacavir >Abacavir(base);Abacavir Tablet;Abacavir, ≥ 98.0%;Abacavir USP/EP/BP;Abacavir Standard;Abacavir (see A105000)
- CBNumber:
- CB5698138
- Molecular Formula:
- C14H18N6O
- Molecular Weight:
- 286.33
- MOL File:
- 136470-78-5.mol
- MSDS File:
- SDS
- Modify Date:
- 2024/4/16 18:29:06
| Melting point | 165° |
|---|---|
| alpha | D20 -59.7°; 43620 -127.8°; 36520 -218.1° (c = 0.15 in methanol) |
| Boiling point | 636.0±65.0 °C(Predicted) |
| Density | 1.70±0.1 g/cm3(Predicted) |
| storage temp. | Sealed in dry,2-8°C |
| solubility | DMSO (Slightly, Heated), Methanol (Slightly) |
| pka | 5.01(at 25℃) |
| form | Solid |
| color | Off-White to Pale Beige |
| Merck | 14,1 |
| BCS Class | 3 |
| CAS DataBase Reference | 136470-78-5(CAS DataBase Reference) |
| EPA Substance Registry System | 2-Cyclopentene-1-methanol, 4-[2-amino-6-(cyclopropylamino)-9H-purin-9-yl]-, (1S,4R)- (136470-78-5) |
SAFETY
Risk and Safety Statements
| Symbol(GHS) |  GHS08 |
|||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Signal word | Danger | |||||||||
| Hazard statements | H361-H317-H334-H341 | |||||||||
| Precautionary statements | P501-P261-P272-P202-P201-P280-P284-P302+P352-P308+P313-P362+P364-P304+P340-P333+P313-P405 | |||||||||
| RTECS | GY5979200 | |||||||||
| HS Code | 2933.99.7500 | |||||||||
| NFPA 704 |
|
Abacavir price More Price(2)
Abacavir Chemical Properties,Uses,Production
Description
The drug is extensively metabolized via stepwise phosphorylation to 5′-mono-, di-, and triphosphate. Abacavir is well absorbed (>75%) and penetrates the CNS. The drug can be taken without regard to meals. The drug does not show any clinically significant drug–drug interactions. Abacavir has been reported to produce life-threatening hypersensitivity reactions. The major use of abacavir appears to be in combination with other nucleoside RT inhibitors. A fixed-combination product has recently been approved by the U.S. FDA consisting of 300 mg of ABC, 150 mg of 3TC, and 300 mg of ZDV (Trizivar). The combination has been shown to be superior to other combinations in reducing viral load as well as to show improvement in CD4 cell count.
Uses
Abacavir is a commonly used nucleoside analogue with potent antiviral activity against HIV-1. - See more at: http://www.selleckchem.com/products/abacavir-sulfate.html#sthash.lApvcTNO.dpuf
Indications
Abacavir (Ziagen) is a guanosine nucleoside analogue indicated for the therapy of HIV-1 infection in adults and children. It is used as part of a multidrug regimen and is available in a fixed-dose combination with zidovudine and lamivudine (Trizivir). It is also used for postexposure HIV infection prophylaxis.
Antimicrobial activity
Abacavir has activity against HIV-1, HIV-2 and human T-cell lymphotrophic virus type-1 (HTLV-1).
Acquired resistance
Resistance is associated with specific changes in codons 184 with 65, 74 or 115 in the HIV reverse transcriptase codon region.
General Description
Abacavir is a nucleoside reverse transcriptase inhibitorNRTI that has been approved for use in combination therapiesfor the treatment of HIV and AIDS. Once in the tissues,it is metabolized by stepwise phosphorylation to themonophosphate, diphosphate, and triphosphate. Abacavir ishighly bioavailable (>75%) and is effective by the oralroute. It penetrates the blood-brain barrier efficiently.Abacavir has been reported to produce life-threatening hypersensitivityreactions in some patients.
Pharmaceutical Applications
A synthetic analog of guanine formulated for oral use.
Pharmacokinetics
Oral absorption: 83%
Cmax 300 mg oral, twice daily: 3.0 ± 0.89 mg/L
600 mg once daily: 4.26 mg/L
Plasma half-life: 1.5 h
Volume of distribution: 0.8 L/kg
Plasma protein binding: c. 49%
Absorption
After oral administration abacavir sulfate undergoes rapid and extensive absorption unaffected by food.
Distribution
It penetrates well into the cerebrospinal fluid (CSF) and is an NRTI of choice if this characteristic is thought desirable. Good penetration into the male genital tract has been observed. The drug is secreted into human breast milk.
Metabolism
It is primarily metabolized in the liver, mainly by alcohol dehydrogenase and glucuronidation.
Excretion
Around 83% of the dose is eliminated in the urine, <2% as unchanged drug; the remainder is excreted in the feces. Dose adjustment is unnecessary in renal impairment. It can be used in moderate hepatic impairment, but is contraindicated if dysfunction is severe.
Clinical Use
Treatment of HIV infection in adults and children (in combination with other antiretroviral drugs)
Side effects
Life-threatening hypersensitivity reactions occur in 5–8%
of all individuals, necessitating discontinuation of the drug.
Typically patients present within the first 6 weeks of starting
treatment with fever, rash or other symptoms that worsen
in severity with continued drug exposure. Hypersensitivity
is associated with carriage of the major histocompatibility
complex class I allele HLA-B57*01 and screening for this
allele can significantly reduce the incidence
of this effect.
Current or recent (within the preceding 6 months) use of
abacavir has been associated with a risk of myocardial infarction,
but studies have yielded conflicting data.
Abacavir Preparation Products And Raw materials
Raw materials
1of2
chevron_rightPreparation Products
| Supplier | Tel | Country | ProdList | Advantage | Inquiry |
|---|---|---|---|---|---|
| J S LABS | +91-7330612784 +91-7330612784 | Tamil Nadu, India | 160 | 58 | Inquiry |
| GLP Pharma Standards | +91 9866074638 | Hyderabad, India | 1644 | 58 | Inquiry |
| HRV Global Life Sciences | +91-9820219686 +91-9820219686 | Telangana, India | 379 | 58 | Inquiry |
| Svak Life Sciences | +91-9963463333 +91-9705383777 | Hyderabad, India | 101 | 58 | Inquiry |
| Rivashaa Agrotech Biopharma Pvt. Ltd. | +91-26463395 +91-7926462688 | Gujarat, India | 1615 | 58 | Inquiry |
| Raising Sun Pharma | +91-9399941155 +91-9399941155 | Telangana, India | 127 | 58 | Inquiry |
| Laurus Labs Ltd | +91-4066594333 +91-4039804333 | Telangana, India | 50 | 58 | Inquiry |
| Arene Lifesciences Limited | +91-9849847907 +91-8455241148 | Telangana, India | 55 | 58 | Inquiry |
| Vishrudh laboratories pvt ltd | +91-9666132889 +91-8097369002 | Telangana, India | 117 | 58 | Inquiry |
| Vijaya Pharma And Life Science | +91-8939866271 +91-8939866271 | Tamil Nadu, India | 320 | 58 | Inquiry |
| Supplier | Advantage |
|---|---|
| J S LABS | 58 |
| GLP Pharma Standards | 58 |
| HRV Global Life Sciences | 58 |
| Svak Life Sciences | 58 |
| Rivashaa Agrotech Biopharma Pvt. Ltd. | 58 |
| Raising Sun Pharma | 58 |
| Laurus Labs Ltd | 58 |
| Arene Lifesciences Limited | 58 |
| Vishrudh laboratories pvt ltd | 58 |
| Vijaya Pharma And Life Science | 58 |
Related articles
- What is Abacavir sulfate?
- Abacavir sulfate (also known as 1592U89) is a carbocyclic nucleotide analog that received US Food and Drug Administration (FDA....
- Apr 15,2022
136470-78-5(Abacavir)Related Search:
1of4
chevron_right




