Cytarabine
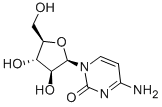
- CAS No.
- 147-94-4
- Chemical Name:
- Cytarabine
- Synonyms
- CYTOSINE ARABINOSIDE;cytarabin;DepoCyte;cytosar-u;1-β-D-Arabinofuranosylcytosine;4-AMino-1-((2R,3S,4S,5R)-3,4-dihydroxy-5-(hydroxyMethyl)tetrahydrofuran-2-yl)pyriMidin-2(1H)-one;ARA-C;Iretin;udicil;CS-193
- CBNumber:
- CB6305084
- Molecular Formula:
- C9H13N3O5
- Molecular Weight:
- 243.22
- MOL File:
- 147-94-4.mol
- MSDS File:
- SDS
- Modify Date:
- 2024/5/16 14:11:51
| Melting point | 214 °C |
|---|---|
| alpha | D24 +153° (c = 0.5 in water) |
| Boiling point | 386.09°C (rough estimate) |
| Density | 1.3686 (rough estimate) |
| refractive index | 1.5100 (estimate) |
| storage temp. | 2-8°C |
| solubility | H2O: 50 mg/mL, clear, colorless |
| form | crystalline |
| pka | pKa 4.3 (Uncertain) |
| color | Prisms from EtOH (aq) |
| Water Solubility | almost transparency |
| λmax | 272nm(H2O)(lit.) |
| Merck | 14,2784 |
| BRN | 89175 |
| Stability | Stable for 1 year as supplied from date of purchase. Solutions in DMSO or water may be stored at -20°C for up to 1 month. |
| InChIKey | UHDGCWIWMRVCDJ-STUHELBRSA-N |
| LogP | -1.808 (est) |
| CAS DataBase Reference | 147-94-4(CAS DataBase Reference) |
| EPA Substance Registry System | Cytarabine (147-94-4) |
SAFETY
Risk and Safety Statements
| Symbol(GHS) |   GHS07,GHS08 |
|||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Signal word | Warning | |||||||||
| Hazard statements | H317-H361fd | |||||||||
| Precautionary statements | P202-P261-P272-P280-P302+P352-P308+P313 | |||||||||
| Hazard Codes | Xn,Xi | |||||||||
| Risk Statements | 43-63-36/37/38-20/21/22 | |||||||||
| Safety Statements | 36/37-37/39-36-26 | |||||||||
| WGK Germany | 3 | |||||||||
| RTECS | HA5425000 | |||||||||
| F | 10-23 | |||||||||
| HS Code | 29349990 | |||||||||
| Toxicity | LD50 oral in rat: > 5gm/kg | |||||||||
| NFPA 704 |
|
Cytarabine price More Price(10)
| Manufacturer | Product number | Product description | CAS number | Packaging | Price | Updated | Buy |
|---|---|---|---|---|---|---|---|
| Sigma-Aldrich(India) | C1768 | Cytosine β-D-arabinofuranoside crystalline, ≥90% (HPLC) | 147-94-4 | 100MG | ₹4730.53 | 2022-06-14 | Buy |
| Sigma-Aldrich(India) | C1768 | Cytosine β-D-arabinofuranoside crystalline, ≥90% (HPLC) | 147-94-4 | 500MG | ₹15642.13 | 2022-06-14 | Buy |
| Sigma-Aldrich(India) | PHR1787 | Cytarabine Pharmaceutical Secondary Standard; Certified Reference Material | 147-94-4 | 500MG | ₹16053.48 | 2022-06-14 | Buy |
| Sigma-Aldrich(India) | C1768 | Cytosine β-D-arabinofuranoside crystalline, ≥90% (HPLC) | 147-94-4 | 1G | ₹21790.73 | 2022-06-14 | Buy |
| Sigma-Aldrich(India) | C1768 | Cytosine β-D-arabinofuranoside crystalline, ≥90% (HPLC) | 147-94-4 | 5G | ₹75244.58 | 2022-06-14 | Buy |
Cytarabine Chemical Properties,Uses,Production
Chemical Properties
A white or almost white, crystalline powder, freely soluble in water, very slightly soluble in alcohol and in methylene chloride.
Uses
Cytarabine USP (Cytosar)is used to treat Acute granulocytic leukemia (adults); acute lymphocytic leukemia (children); Hodgkin’s disease
Indications
Cytarabine (cytosine arabinoside, ara-C, Cytosar-U) is an analogue of the pyrimidine nucleosides cytidine and deoxycytidine. It is one of the most active agents available for the treatment of acute myelogenous leukemia. Cytarabine kills cells in the S-phase of the cycle by competitively inhibiting DNA polymerase. The drug must first be activated by pyrimidine nucleoside kinases to the triphosphate nucleotide ara-cytosine triphosphate (ara-CTP). The susceptibility of tumor cells to cytarabine is thought to be a reflection of their ability to activate the drug more rapidly (by kinases) than to inactivate it (by deaminases).
Definition
ChEBI: A pyrimidine nucleoside in which cytosine is attached to D-arabinofuranose via a beta-N1-glycosidic bond.
General Description
Cytarabine is a pyrimidine nucleoside drug that is related toidoxuridine. This agent is primarily used as an anticanceragent for Burkitt lymphoma and myeloid and lymphaticleukemias. Cytarabine blocks the cellular utilization of deoxycytidine,hence inhibiting the replication of viral DNA.Before it becomes active, the drug is converted to monophosphates,diphosphates, and triphosphates, which block DNApolymerase and the C-2 reductase that converts cytidinediphosphate into the deoxy derivative.
The antiviral use of cytarabine is in the treatment of herpeszoster (shingles), herpetic keratitis, and viral infectionsthat resist idoxuridine. Cytarabine is usually administeredtopically. Toxicity occurs on bone marrow, the gastrointestinal(GI) tract, and the kidneys.
Air & Water Reactions
Water soluble.
Health Hazard
ACUTE/CHRONIC HAZARDS: Very toxic. Hazardous decomposition products. May cause irritation on contact. Teratogen. Mutagen. Central nervous system effects.
Mechanism of action
Cytarabine is rapidly metabolized in the liver, kidney, intestinal mucosa, and red blood cells and has a half-life in plasma of only 10 minutes after intravenous bolus injection. The major metabolite, uracil arabinoside (ara-U), can be detected in the blood shortly after cytarabine administration. About 80% of a given dose is excreted in the urine within 24 hours, with less than 10% appearing as cytarabine; the remainder is ara-U.When the drug is given by continuous infusion, cytarabine levels in CSF approach 40% of those in plasma.
Clinical Use
Cytarabine is used in the chemotherapy of acute myelogenous leukemia, usually in combination with an anthracycline agent, thioguanine, or both. It is less useful in acute lymphoblastic leukemia and the lymphomas and has no known activity against other tumors. It has been used intrathecally in the treatment of meningeal leukemias and lymphomas as an alternative to methotrexate.
Safety Profile
Moderate to low toxicity byingestion. Human systemic effects: allergic dermatitis,ataxia, blood changes, central nervous system effectsconjunctive irritation, degenerative brain changes, hearingacuity change, lachrymation, peripheral nervefasciculati
Purification Methods
Purify cytarabin by recrystallisation from aqueous EtOH or a large volume of H2O (it solubility at ~20o is 5%). It has max 212 and 279nm at pH 2 and 272nm at pH 12. It is an acute leukaemic agent. [Walwick et al. Proc Chem Soc (London) 84 1959, Beilstein 25 III/IV 3669.]
Cytarabine Preparation Products And Raw materials
Raw materials
Preparation Products
| Supplier | Tel | Country | ProdList | Advantage | Inquiry |
|---|---|---|---|---|---|
| Basil Drugs AND Pharmaceuticals Pvt Ltd | +91-2249700250 +91-9619320820 | Mumbai, India | 108 | 58 | Inquiry |
| Kekule Pharma Limited | +919849798889 | Hyderabad, India | 59 | 58 | Inquiry |
| Ralington Pharma | +91-7948911722 +91-9687771722 | Gujarat, India | 1350 | 58 | Inquiry |
| Kekule Life Sciences Limited | +91-9963474090 +91-9963474090 | AndhraPradesh, India | 19 | 58 | Inquiry |
| Mac Chem Products (India) Pvt Ltd (Naprod Group) | +91-9727753234 +91-7770016606 | Maharashtra, India | 37 | 58 | Inquiry |
| Surajlok Chemicals Pvt Ltd (Neon Laboratories Ltd) | +91-2268107000 +91-9987232509 | Maharashtra, India | 30 | 58 | Inquiry |
| Myosynth Laboratories Pvt Ltd | +91-8913266227 +91-9885891884 | AndhraPradesh, India | 49 | 58 | Inquiry |
| AKASH PHARMA EXPORTS | +91-9388123451 +91-9846039283 | Kerela, India | 470 | 58 | Inquiry |
| Innovative Synthetic Molecules | +91 40-24111684 | Hyderabad, India | 527 | 58 | Inquiry |
| FRESENIUS KABI ONCOLOGY LIMITED | +91 124 4885000 / +91 124 3325000 | New Delhi, India | 24 | 58 | Inquiry |
147-94-4(Cytarabine)Related Search:
1of4
chevron_right




