Eptifibatide Acetate
- CAS No.
- 148031-34-9
- Chemical Name:
- Eptifibatide Acetate
- Synonyms
- Eptifitide;INTEGRELIN;Eptifibatide;EFTIFIBATIDE;Human Eptifibatide;EPTIFIBATIDE ACETATE;peptide eptifibatide acetate;Eptifibatide?Acetate impurity;MAP-LYS-GLY-ASP-TRP-PRO-CYS-NH2;Mpr-Har-Gly-Asp-Trp-Pro-Cys-NH2
- CBNumber:
- CB7416565
- Molecular Formula:
- C35H49N11O9S2
- Molecular Weight:
- 831.96
- MOL File:
- 148031-34-9.mol
- Modify Date:
- 2024/8/21 22:41:43
| Melting point | >220°C (dec.) |
|---|---|
| storage temp. | Refrigerator |
| solubility | DMSO (Slightly), Methanol (Slightly), Water (Slightly) |
| form | Solid |
| color | White to Off-White |
| CAS DataBase Reference | 148031-34-9(CAS DataBase Reference) |
Eptifibatide Acetate Chemical Properties,Uses,Production
Indications
Acute Coronary Syndrome (ACS)
INTEGRILIN? is indicated to decrease the rate of a combined endpoint of death or new myocardial infarction (MI) in patients with ACS (unstable angina [UA]/non-ST-elevation myocardial infarction [NSTEMI]), including patients who are to be managed medically and those undergoing percutaneous coronary intervention (PCI).
Percutaneous Coronary Intervention (PCI)
INTEGRILIN is indicated to decrease the rate of a combined endpoint of death, new MI, or need for urgent intervention in patients undergoing PCI, including those undergoing intracoronary stenting.
Application
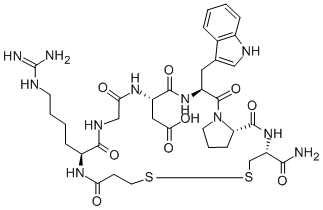
Eptifibatide is a cyclic heptapeptide containing 6 amino acids and 1 mercaptopropionyl (des-amino cysteinyl) residue. An interchain disulfide bridge is formed between the cysteine amide and the mercaptopropionyl moieties. Chemically it is N6-(aminoiminomethyl)-N2-(3-mercapto-1-oxopropyl)-Llysylglycyl-L-α-aspartyl-L-tryptophyl-L-prolyl-L-cysteinamide, cyclic (1→6)-disulfide. Eptifibatide binds to the platelet receptor glycoprotein (GP) IIb/IIIa of human platelets and inhibits platelet aggregation.
The eptifibatide peptide is produced by solution-phase peptide synthesis, and is purified by preparative reverse-phase liquid chromatography and lyophilized. The structural formula is:

Dosage forms
Before infusion of INTEGRILIN, the following laboratory tests should be performed to identify pre-existing hemostatic abnormalities: hematocrit or hemoglobin, platelet count, serum creatinine, and PT/aPTT. In patients undergoing PCI, the activated clotting time (ACT) should also be measured.
The activated partial thromboplastin time (aPTT) should be maintained between 50 and 70 seconds unless PCI is to be performed. In patients treated with heparin, bleeding can be minimized by close monitoring of the aPTT and ACT.
Eptifibatide Acetate Preparation Products And Raw materials
Raw materials
Preparation Products
| Supplier | Tel | Country | ProdList | Advantage | Inquiry |
|---|---|---|---|---|---|
| USV Pvt Ltd | +91-2267861111 +91-2225564048 | Maharashtra, India | 36 | 58 | Inquiry |
| OCEAN TRADING CORPORATION | +91(22) 24921669 | New Delhi, India | 6211 | 58 | Inquiry |
| USV PRIVATE LTD | +91-22-2556-4048 & 6786-1111 | New Delhi, India | 27 | 58 | Inquiry |
| SUN PHARMACEUTICAL INDUSTRIES LTD | (+91 22) 4324 4324 | New Delhi, India | 175 | 58 | Inquiry |
| CLEARSYNTH LABS LTD. | +91-22-45045900 | Hyderabad, India | 6351 | 58 | Inquiry |
| SynZeal Research Pvt Ltd | +1 226-802-2078 | Gujarat, India | 6522 | 58 | Inquiry |
| CAD Pharma Inc | 08048372459Ext 215 | Karnataka, India | 12 | 58 | Inquiry |
| Jigs Chemical Limited | +91 80 4290 2764 | Ahmedabad, India | 235 | 58 | Inquiry |
| Hemmo Pharmaceuticals Private Limited | 08048361298Ext 414 | Mumbai, India | 6 | 58 | Inquiry |
| Cellmano Biotech Limited | 0551-65326643 18156095617 | China | 999 | 58 | Inquiry |
| Supplier | Advantage |
|---|---|
| USV Pvt Ltd | 58 |
| OCEAN TRADING CORPORATION | 58 |
| USV PRIVATE LTD | 58 |
| SUN PHARMACEUTICAL INDUSTRIES LTD | 58 |
| CLEARSYNTH LABS LTD. | 58 |
| SynZeal Research Pvt Ltd | 58 |
| CAD Pharma Inc | 58 |
| Jigs Chemical Limited | 58 |
| Hemmo Pharmaceuticals Private Limited | 58 |
| Cellmano Biotech Limited | 58 |





