Misoprostol
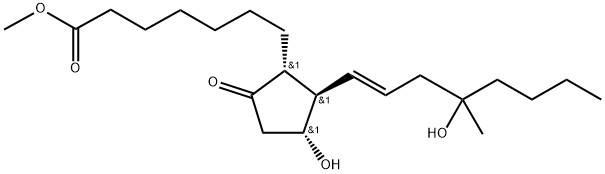
- CAS No.
- 59122-46-2
- Chemical Name:
- Misoprostol
- Synonyms
- Misoprosto;(11α,13E)-(-)-11,16-Dihydroxy-16-methyl-9-oxo-prost-13-en-1-oicacidmethylester;SC 2933;CYTOTEC;Misogon;SC-29333;Misoprostil;MISOPROSTOL;Mesoprostal;DL-MISOPROSTOL
- CBNumber:
- CB7713293
- Molecular Formula:
- C22H38O5
- Molecular Weight:
- 382.54
- MOL File:
- 59122-46-2.mol
- MSDS File:
- SDS
- Modify Date:
- 2024/3/6 17:09:24
| Melting point | 261-263°C |
|---|---|
| Boiling point | 429.67°C (rough estimate) |
| Density | 1.0323 (rough estimate) |
| refractive index | 1.6120 (estimate) |
| storage temp. | Sealed in dry,Store in freezer, under -20°C |
| solubility | Practically insoluble in water, soluble in ethanol (96 per cent), sparingly soluble in acetonitrile. |
| pka | 13.92±0.60(Predicted) |
| CAS DataBase Reference | 59122-46-2(CAS DataBase Reference) |
| EPA Substance Registry System | Misoprostol (59122-46-2) |
SAFETY
Risk and Safety Statements
| Symbol(GHS) |   GHS06,GHS08 |
|||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Signal word | Danger | |||||||||
| Hazard statements | H301-H360 | |||||||||
| Precautionary statements | P201-P280-P301+P330+P331+P310-P308+P313 | |||||||||
| Hazard Codes | T | |||||||||
| Risk Statements | 45-60-61-25-36/37/38 | |||||||||
| Safety Statements | 53-22-36/37/39-45 | |||||||||
| RIDADR | UN 2810 6.1/PG 3 | |||||||||
| WGK Germany | 2 | |||||||||
| RTECS | UK8390000 | |||||||||
| HS Code | 2937500000 | |||||||||
| Toxicity | LD50 in rats, mice (mg/kg): 40-62, 70-160 i.p.; 81-100, 27-138 orally (Kotsonis) | |||||||||
| NFPA 704 |
|
Misoprostol Chemical Properties,Uses,Production
Description
Misoprostol is an orally-active PGEl analog useful in the treatment of gastric and duodenal ulcers. As with other agents of this type, misoprostol may prove useful as a gastric cytoprotective when administered concurrently with irritating drugs such as nonsteroidal antiinflammatories.
Chemical Properties
Misoprostol is a yellow, viscous liquid with a musty odour. Miscible with ethanol, ether or chloroform, extremely insoluble in water or n-hexane. Very unstable at room temperature and undergoes thermal epimerization to the 8-isomer. It is also extremely sensitive to the Ph value. It removes the hydroxyl group at the 11α position in acidity and alkalinity, converts it into A-type prostaglandins, and isomerizes into B-type prostaglandins. However, it is relatively stable in the dispersion system of hydroxypropyl methylcellulose and can be stored at room temperature.
Uses
Misoprostol is a cytoprotective prostaglandin PGE1 analogue that inhibits gastric acid secretion. It has many medical uses, including nine obstetric indications. The drug was originally produced by Pfizer for the treatment of gastric ulcers. Misoprostol is commonly used to prevent NSAID-induced gastric ulcers in patients at high risk of complications from a gastric ulcer, such as elderly patients and patients with a history of ulcer. It has also been used in treating duodenal ulcers unresponsive to histamine H2-antagonists; the drug does not prevent duodenal ulcers,however, in patients taking NSAIDS. Misoprostol can cause miscarriage, often associated with potentially dangerous bleeding.
Indications
Misoprostol (Cytotec), which is an analogue of prostaglandin E1, has been approved for use in the prevention of nonsteroidal antiinflammatory drug–induced ulceration. It also is approved in other countries for the treatment of peptic ulcer disease.
Preparation
synthesis of Misoprostol
To a 1000 ml dried flask under a nitrogen atmosphere was added 74.6 g of(E)-trimethyl-[[1-methyl-1-[3-(tributylstannyl)-2-propenyl]pentyl]oxy]silane,125 ml anhydrous THF and 24.2 g of copper (I) iodide. The mixture wasstirred at room temperature for 30 minutes and then it was cooled to -25 to -30°C. 98.8 ml of methyllithium (2.86 M) in DEM was added dropwise and theresultant solution was stirred at -15°C for 2 hours. Then the reaction mixturewas cooled to -78°C and 25 g of methyl-5-oxo-3-[(triethylsilyl)oxy]-1-cyclopentene-1-heptanoate in 100 ml of THF was added rapidly. After stirringthe mixture for 5 min at -78°C, it was quenched into a mixture of 750 ml ofaqueous ammonium chloride solution and 200 ml of ammonium hydroxide.The resulting mixture was warmed to room temperature and stirred until adeep blue aqueous layer was obtained. Ethyl acetate (250 ml) was used forextraction. Then the combined organic layers were washed with brine andsubsequently dried over magnesium sulfate. After a filtration andconcentration under reduced pressure, an oil (105 g) was obtained. This oilcontaining the protected prostaglandin was subjected to acidic deprotection(cat. PPTS, acetone and water) and purification (chromatography on silica gel)to provide 15.8 g (60%) of misoprostol was identical.
Biological Activity
Cytoprotective prostaglandin E 1 analog that displays agonist activity at EP receptors. K i values are 120, 250, 67 and 67 nM at cloned mouse EP 1 , EP 2 , EP 3 and EP 4 receptors respectively. Prevents NSAID-induced gastric ulceration.
Pharmacology
Misoprostol is absorbed rapidly after oral administration and is hydrolyzed to the active compound. It is metabolized by the liver and excreted mainly in the urine. Adverse effects include crampy abdominal pain, dose-related diarrhea, and uterine contractions. The last-named effect has led to its use in the control of postpartum bleeding.
Mode of action
Misoprostol is a synthetic prostaglandin E1 analogue which has ulcer healing, gastric acid antisecretory and mucosal protective properties. The antisecretory activity is mediated by direct action on specific prostaglandin receptors on the surface of gastric parietal cells. In dogs with innervated Pavlov pouches, inhibition of secretion is achieved at a lower dosage by intrapouch injection than by intravenous or intragastric administration, suggesting that the local effect may predominate. The mucosal protective effect against various damaging agents has been demonstrated in humans with doses that inhibit and doses which minimally affect acid secretion.
Misoprostol Preparation Products And Raw materials
Raw materials
1of3
chevron_rightPreparation Products
| Supplier | Tel | Country | ProdList | Advantage | Inquiry |
|---|---|---|---|---|---|
| BTC pharm India | +918790379245 | AndhraPradesh, India | 1269 | 58 | Inquiry |
| Aptus Therapeutics | +91-9966349855 +91-9966349855 | Telangana, India | 19 | 58 | Inquiry |
| Sonia Organics | +91-9538454135 +91-9538454135 | Karnataka, India | 28 | 58 | Inquiry |
| Honour Lab Limited | +919845977466 | Telangana, India | 164 | 58 | Inquiry |
| Avra Laboratories Pvt Ltd | +91-8008572391 +91-8008572391 | Hyderabad, India | 43 | 58 | Inquiry |
| ANVI Pharma | +91-2228995761 +91-2228995761 | Maharashtra, India | 10 | 58 | Inquiry |
| Chempifine Chemicals | +91-2225667766 +91-2225667766 | Punjab, India | 144 | 58 | Inquiry |
| Fleming Laboratories Ltd | +91-9666407333 +91-9666022445 | Hyderabad, India | 22 | 58 | Inquiry |
| Besil Chem LLP | +91-7760252666 +91-9880731835 | Bangalore, India | 163 | 58 | Inquiry |
| Alfa Omega Pharma | +91-8050045945 +91-9972665399 | Maharashtra, India | 126 | 58 | Inquiry |
| Supplier | Advantage |
|---|---|
| BTC pharm India | 58 |
| Aptus Therapeutics | 58 |
| Sonia Organics | 58 |
| Honour Lab Limited | 58 |
| Avra Laboratories Pvt Ltd | 58 |
| ANVI Pharma | 58 |
| Chempifine Chemicals | 58 |
| Fleming Laboratories Ltd | 58 |
| Besil Chem LLP | 58 |
| Alfa Omega Pharma | 58 |





