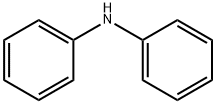
Diphenylamine
- CAS No.
- 122-39-4
- Chemical Name:
- Diphenylamine
- Synonyms
- DPA;DFA;N-PHENYLANILINE;Diphenylamin;Diphenylaniline;Diphenylamine, synthesis grade;Z-876;OA-505;CORAZA;ci10355
- CBNumber:
- CB7852949
- Molecular Formula:
- C12H11N
- Molecular Weight:
- 169.22
- MOL File:
- 122-39-4.mol
- Modify Date:
- 2024/3/14 15:18:27
| Melting point | 52 °C |
|---|---|
| Boiling point | 302 °C(lit.) |
| Density | 1.16 |
| vapor density | 5.82 (vs air) |
| vapor pressure | 1 mm Hg ( 108 °C) |
| refractive index | 1.5785 (estimate) |
| Flash point | 307 °F |
| storage temp. | Store below +30°C. |
| solubility | alcohol: passes test |
| form | crystalline |
| pka | 0.79(at 25℃) |
| color | tan |
| Odor | Floral odor |
| Water Solubility | Slightly soluble. 0.03 g/100 mL |
| Sensitive | Air & Light Sensitive |
| Merck | 14,3317 |
| BRN | 508755 |
| Exposure limits | TLV-TWA 10 mg/m3 (ACGIH and MSHA). |
| Dielectric constant | 3.3(11℃) |
| Stability | Stable; may discolour on exposure to light. Incompatible with strong acids, strong oxidizing agents. |
| LogP | 3.82 at 20.2℃ |
| CAS DataBase Reference | 122-39-4(CAS DataBase Reference) |
| NIST Chemistry Reference | Diphenylamine(122-39-4) |
| EPA Substance Registry System | Diphenylamine (122-39-4) |
SAFETY
Risk and Safety Statements
| Symbol(GHS) |    GHS06,GHS08,GHS09 |
|||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Signal word | Danger | |||||||||
| Hazard statements | H301+H311+H331-H373-H410 | |||||||||
| Precautionary statements | P273-P280-P301+P310-P302+P352+P312-P304+P340+P311-P314 | |||||||||
| Hazard Codes | T,N,F | |||||||||
| Risk Statements | 23/24/25-33-50/53-52/53-39/23/24/25-11-51/53 | |||||||||
| Safety Statements | 28-36/37-45-60-61-28A-16-7 | |||||||||
| RIDADR | UN 3077 9/PG 3 | |||||||||
| OEB | B | |||||||||
| OEL | TWA: 10 mg/m3 | |||||||||
| WGK Germany | 3 | |||||||||
| RTECS | JJ7800000 | |||||||||
| F | 8-10-23 | |||||||||
| Autoignition Temperature | 630 °C DIN 51794 | |||||||||
| TSCA | Yes | |||||||||
| HS Code | 2921 44 00 | |||||||||
| HazardClass | 6.1 | |||||||||
| PackingGroup | III | |||||||||
| Toxicity | LD50 orally in Rabbit: 1120 mg/kg LD50 dermal Rabbit > 5000 mg/kg | |||||||||
| NFPA 704 |
|
Diphenylamine price More Price(28)
| Manufacturer | Product number | Product description | CAS number | Packaging | Price | Updated | Buy |
|---|---|---|---|---|---|---|---|
| Sigma-Aldrich(India) | 8.20528 | Diphenylamine for synthesis | 122-39-4 | 1IN28205280250 | ₹1470.01 | 2022-06-14 | Buy |
| Sigma-Aldrich(India) | 8.20528 | Diphenylamine for synthesis | 122-39-4 | 100G | ₹5330 | 2022-06-14 | Buy |
| Sigma-Aldrich(India) | CRM69121 | Diphenylamine certified reference material, TraceCERT? | 122-39-4 | 100MG | ₹12427.1 | 2022-06-14 | Buy |
| Sigma-Aldrich(India) | 8.20528 | Diphenylamine for synthesis | 122-39-4 | 1KG | ₹12720 | 2022-06-14 | Buy |
| Sigma-Aldrich(India) | 8.20528 | Diphenylamine for synthesis | 122-39-4 | 50KG | ₹118150 | 2022-06-14 | Buy |
Diphenylamine Chemical Properties,Uses,Production
Description
Solutions of diphenylamine are used to treat apples a few days before harvest. Residue on apples’ surfaces of 10 ppm is permitted by regulation in Australia, Canada, and the United States.
Chemical Properties
Diphenylamine is a colorless monoclinic leafl et substance. It is used in the manufacture of a variety of substances, i.e., dyestuffs and their intermediates, pesticides, antihelmintic drugs, and as reagents in analytical chemistry laboratories.
Uses
Diphenylamine is an aromatic amine that was shown to exhibit antioxidant activities and is now used as an anti-scald agent. It is also used in the manufacture of a variety of substances, for instance, dye stuffs and their intermediates, pesticides, anthelmintic drugs, and as reagents in analytical chemistry laboratories.
Definition
diphenylamine: A colourless crystallinearomatic compound,(C6H5)2NH; m.p. 54°C. It is made byheating phenylamine (aniline) withphenylamine hydrochloride. It is asecondary amine and is both slightlyacidic (forming an N-potassium salt)and slightly basic (forming salts withmineral acids). Its derivatives are employedas stabilizers for syntheticrubber and rocket fuels.
General Description
Light tan to brown solid with a pleasant odor. Sinks in water.
Air & Water Reactions
Dust may be explosive if mixed with air in critical proportions and in the presence of a source of ignition [USCG, 1999]. Insoluble in water.
Reactivity Profile
Diphenylamine discolors in light. Diphenylamine can react violently with hexachloromelamine and trichloromelamine. Diphenylamine is incompatible with strong oxidizing agents and strong acids. Diphenylamine is also incompatible with iron and silver salts. Diphenylamine reacts with nitrogen oxides.
Health Hazard
Diphenylamine is highly toxic and is rapidly absorbed by the skin and through inhalation. It has caused anorexia, hypertension, eczema, and bladder symptoms. Experimental animals exposed to diphenylamine demonstrated cystic lesions but failed to demonstrate cancerous growth. Inhalation of diphenylamine dust may cause systemic poisoning. The symptoms of toxicity include, but are not limited to, anoxia, headache, fatigue, anorexia, cyanosis, vomiting, diarrhea, emaciation, hypothermia, bladder irritation, kidney, heart, and liver damage.
Fire Hazard
Noncombustible solid; autoignition temperature 634°C (1173°F); low reactivity.
Agricultural Uses
Insecticide, Fungicide, Herbicide, Plant growth regulator: Topically in anti-screwworm mixtures, foliar application in a modified growth chamber to decrease ozone injury to leaves of apple, bean, muskmelon, petunia, and tobacco plants. To control weather fleck in tobacco and inhibit algae formation. To prolong the fresh appearance of snapdragons. Protect rice from the toxic effects of thiolcarbamate herbicides [83] . Not currently approved for use in EU countries (resubmitted) . Registered for use in the U.S. and other countries.
Trade name
NOSCALD DPA 31; NOSCALD DPA 283; SCALDIP; Z-876
Safety Profile
Poison by ingestion. Experimental teratogenic effects. Action similar to anhne but less severe. Combustible when exposed to heat or flame. Can react violently with hexachloromelamine or trichloromelamine. Can react with oxilzing materials. To fight fire, use CO2, dry chemical. When heated to decomposition it emits highly toxic fumes of NOx,. See also ANILINE, AMINES, and AROMATIC AMINES.
Environmental Fate
Diphenylamine is present in waste water from industrial
processes. Diphenylamine has been detected in milk of
animals (cow, sheep, goat, water buffalo) raised in Italy and
France.
Pseudokirchneriella subcapitata (Algae) growth was inhibited
with a dose of 0.30 mg l-1. Aquatic invertebrates Daphnia
magna showed an acute 48 h EC50 dose of 1.2 mg l-1.
Metabolic pathway
The major metabolite of diphenylamine (DPA) identified in stored apples is a glucose conjugate of 4-hydroxydiphenylamine, and additional metabolites, characterized as glycosyl conjugates of 2-hydroxy- DPA, 3-hydroxy-DPA, 4-hydroxy-DPA, or dihydroxy- DPA, are also detected along with their intact (i.e. non-conjugated) forms in apple pulp.
storage
Diphenylamine should be protected from physical damage. Storage of diphenylamine outside or a detached area is preferred. Inside storage should be in a standard flammable liquids storage room or cabinet. Diphenylamine should be kept separately from oxidizing materials and incompatible chemical substances. Storage and work areas should be no smoking areas. Diphenylamine should be kept protected from light.
Purification Methods
Crystallise diphenylamine from pet ether, MeOH, or EtOH/water. Dry it under vacuum. [Beilstein 12 H 174, 12 IV 271.]
Precautions
Students and occupational workers should be careful during use and handling of diphenylamine. Workers should wear impervious protective clothing, including boots, gloves, a laboratory coat, apron or coveralls, as appropriate, to prevent skin contact. Finely dispersed particles of diphenylamine form explosive mixtures in air. Diphenylamine is very harmful on exposures by swallowing, inhalation, and/or skin absorption. Diphenylamine causes irritation to the skin, eyes, and respiratory tract, and causes blood vascular changes leading to methemoglobinemia.
Diphenylamine Preparation Products And Raw materials
Raw materials
Preparation Products
1of8
chevron_right| Supplier | Tel | Country | ProdList | Advantage | Inquiry |
|---|---|---|---|---|---|
| JSK Chemicals | +919879767970 | Gujarat, India | 3756 | 58 | Inquiry |
| Kronox Lab Sciences Pvt Ltd | +91-9313231074 +91-9313231074 | Gujarat, India | 240 | 58 | Inquiry |
| NOCIL Limited | +91-2266364062 +91-9892241417 | Maharashtra, India | 7 | 58 | Inquiry |
| DeFINE CHEMICALS | +91-8010769889 +91-9320057099 | Maharashtra, India | 120 | 58 | Inquiry |
| Nikita Transphase Adducts Private Limited | +91-2224440141 +91-2266158844 | Maharashtra, India | 3 | 58 | Inquiry |
| LANXESS India Pvt. Ltd. | +91-8356878763 +91-8356878763 | Maharashtra, India | 190 | 58 | Inquiry |
| S D Fine Chem Limited | +91-9323715856 +91-9323715856 | Maharashtra, India | 368 | 58 | Inquiry |
| CLEARSYNTH LABS LTD. | +91-22-45045900 | Hyderabad, India | 6351 | 58 | Inquiry |
| FINAR LTD | +91 - 2717 - 616717 | New Delhi, India | 660 | 58 | Inquiry |
| Otto Chemie Pvt. Ltd. | +91 9820041841 | Mumbai, India | 5873 | 58 | Inquiry |
| Supplier | Advantage |
|---|---|
| JSK Chemicals | 58 |
| Kronox Lab Sciences Pvt Ltd | 58 |
| NOCIL Limited | 58 |
| DeFINE CHEMICALS | 58 |
| Nikita Transphase Adducts Private Limited | 58 |
| LANXESS India Pvt. Ltd. | 58 |
| S D Fine Chem Limited | 58 |
| CLEARSYNTH LABS LTD. | 58 |
| FINAR LTD | 58 |
| Otto Chemie Pvt. Ltd. | 58 |
Related articles
- The biodegradability of diphenylamine
- Diphenylamine (DPA) is a compound from the third European Union (EU) list of priority pollutants.
- May 19,2022
122-39-4(Diphenylamine)Related Search:
1of4
chevron_right




