Osimertinib mesylate
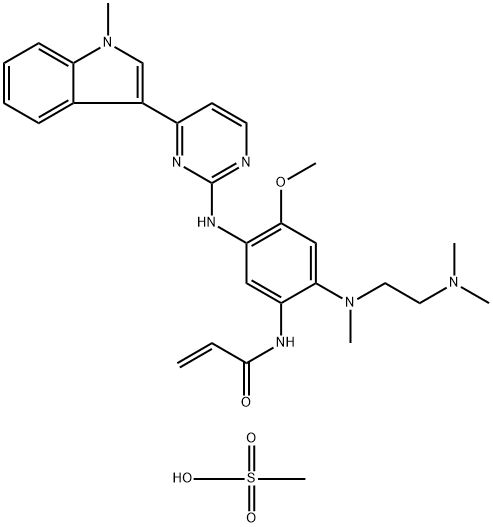
- CAS No.
- 1421373-66-1
- Chemical Name:
- Osimertinib mesylate
- Synonyms
- Osimertinib;Tagrisso;Osimertinib d6;Mereletinib mesylate;Osimertinib methanesulfonate;Osimertinib (AZD9291) mesylate;N-[2-(2-dimethylaminoethylmethylamino)-4-methoxy-5-[[4-(1-methylindol-3-yl)pyrimidin-2-yl]amino]phenyl]prop-2-enamide mesylate salt;N-(2-((2-(dimethylamino)ethyl)(methyl)amino)-4-methoxy-5-((4-(1-methyl-1H-indol-3-yl)pyrimidin-2-yl)amino)phenyl)acrylamide methanesulfonate;N-[2-[[2-(Dimethylamino)ethyl]methylamino]-4-methoxy-5-[[4-(1-methyl-1H-indol-3-yl)-2-pyrimidinyl]amino]phenyl]-2-propenamide methanesulfonate (1:1);PB1211
- CBNumber:
- CB82677060
- Molecular Formula:
- C29H37N7O5S
- Molecular Weight:
- 595.72
- MOL File:
- 1421373-66-1.mol
- Modify Date:
- 2024/7/26 15:15:06
| Melting point | >232°C (dec.) |
|---|---|
| storage temp. | -20°C Freezer, Under inert atmosphere |
| solubility | DMSO (Slightly), Methanol (Slightly) |
| form | Solid |
| color | Off-White to Yellow |
| InChIKey | FUKSNUHSJBTCFJ-UHFFFAOYSA-N |
| SMILES | S(O)(=O)(=O)C.CN1C2=CC=CC=C2C(C2C=CN=C(NC3C=C(NC(=O)C=C)C(N(C)CCN(C)C)=CC=3OC)N=2)=C1 |
SAFETY
Risk and Safety Statements
| Symbol(GHS) |    GHS08,GHS07,GHS09 |
|---|---|
| Signal word | Danger |
| Hazard statements | H317-H372-H410-H400-H361-H302 |
| Precautionary statements | P201-P202-P281-P308+P313-P405-P501-P261-P272-P280-P302+P352-P333+P313-P321-P363-P501-P260-P264-P270-P314-P501-P264-P270-P301+P312-P330-P501-P273-P391-P501-P273-P391-P501 |
| HS Code | 29339900 |
Osimertinib mesylate Chemical Properties,Uses,Production
Description
Osimertinib is active against exon 19 deletions, exon 21 mutations, and also the exon 20 T790M mutations. It is preferentially selective for mutated EGFR, and therefore toxicity at therapeutic doses is lower than for first- and second-generation agents. Notably, osimertinib is able to cross the blood-brain barrier, making it active against disease in the CNS.
Uses
Osimertinib mesylate (AZD9291) is the mesylate form of osimertinib, which is an oral, third-generation epidermal growth factor receptor (EGFR) tyrosine kinase inhibitor (TKI) drug.
Indications
The collection of ibrutinib (Imbruvica(R), Pharmacyclics Inc.), afatinib, and osimertinib represents the small, yet expanding, group of covalent SMKIs. Ibrutinib is a non-receptor Bruton’s tyrosine kinase inhibitor approved for the treatment of relapsed chronic lymphocytic leukemia. Afatinib, approved for NSCLC in 2013 and squamous NSCLC in 2016, is a second-generation irreversible EGFR inhibitor that targets wild-type EGFR, the mutant T790M EGFR, and HER2. Osimertinib (AZD9291), which was approved by FDA in November 2015, is a third-generation irreversible EGFR inhibitor that selectively targets the mutant T790M EGFR. Rociletinib, which shares a high degree of structural similarity with that of osimertinib, is a promising covalent EGFR inhibitor developed by Clovis Oncology aimed for the treatment of patients with EGFR T790M-mutated NSCLC, until the company terminated its development in May 2016 following a negative vote fromthe FDA’sOncologic Drugs Advisory Committee.
Definition
ChEBI: A methanesulfonate (mesylate) salt prepared from equimolar amounts of osimertinib and methanesulfonic acid. Used for treatment of EGFR T790M mutation positive non-small cell lung cancer.
Application
Osimertinib mesylate is approved to treat: Non-small cell lung cancer that has certain EGFR gene mutations. It is used in adults: As adjuvant therapy after surgery to remove the cancer; As the first therapy for cancer that has spread to other parts of the body, or in patients whose cancer has spread to other parts of the body and got worse during or after treatment with another EGFR tyrosine kinase inhibitor.
brand name
TagrissoTM
General Description
Class: receptor tyrosine kinase
Treatment: NSCLC
Oral bioavailability = 70%
Elimination half-life = 48 h
Protein binding = 94.7%
Side effects
Osimertinib toxicity is dose-dependent and is associated with fewer gastrointestinal and dermatologic adverse events than with other approved EGFR TKIs.
Osimertinib mesylate Preparation Products And Raw materials
Raw materials
Preparation Products
| Supplier | Tel | Country | ProdList | Advantage | Inquiry |
|---|---|---|---|---|---|
| Jigs Chemical ltd | +919099003427 | Gujarat, India | 239 | 58 | Inquiry |
| Bulat Pharmaceutical Pvt Ltd | +91-8448085659 +91-8448085660 | Haryana, India | 123 | 58 | Inquiry |
| Alembic Pharmaceuticals Limited | +91-2652280880 +91-2652280550 | Gujarat, India | 115 | 58 | Inquiry |
| MSN LABORATORIES PRIVATE LTD | +91 40 30438600 | New Delhi, India | 230 | 58 | Inquiry |
| ALEMBIC PHARMACEUTICALS LTD | +91 40 23704923 / 24 / 25 | New Delhi, India | 108 | 58 | Inquiry |
| CLEARSYNTH LABS LTD. | +91-22-45045900 | Hyderabad, India | 6351 | 58 | Inquiry |
| Bulat Pharmaceutical Private Limited | 08048372574Ext 902 | Haryana, India | 37 | 58 | Inquiry |
| Pharmaffiliates Analytics and Synthetics P. Ltd | +91-172-5066494 | Haryana, India | 6773 | 58 | Inquiry |
| SynZeal Research Pvt Ltd | +1 226-802-2078 | Gujarat, India | 6522 | 58 | Inquiry |
| Pharma Affiliates | 172-5066494 | Haryana, India | 6761 | 58 | Inquiry |
Related articles
- Osimertinib mesylate: Mechanism of action
- Being third-generation EGFR TKIs, osimertinib mesylate (AZD9291) are designed to target T790M and EGFR TKI–sensitizing mutatio....
- Oct 18,2019





