Sodium chlorite
- CAS No.
- 7758-19-2
- Chemical Name:
- Sodium chlorite
- Synonyms
- CHLORITE;textile;orite;UN1496;TEXTONE;alcideld;neosiloxd;ALCIDEALLAY;CHLORITE ION;Natriumchlorit
- CBNumber:
- CB8854304
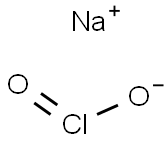
- Molecular Formula:
- ClNaO2
- Molecular Weight:
- 90.44
- MOL File:
- 7758-19-2.mol
- Modify Date:
- 2024/3/16 16:59:00
| Melting point | 190 °C (dec.) |
|---|---|
| Density | 2.5 g/cm3 |
| vapor pressure | 0Pa at 25℃ |
| solubility | Methanol (Slightly), Water (Slightly) |
| form | Powder |
| color | White |
| Odor | odorless |
| PH Range | 10 - 11 |
| explosive limit | 7% |
| Water Solubility | 39 g/100 mL (17 ºC) |
| Sensitive | Hygroscopic |
| Merck | 14,8600 |
| Dielectric constant | 6.1(Ambient) |
| Stability | Stable. Incompatible with phosphorus, sulphur, zinc, ammonia, finely powdered metals, strong reducing agents, acids, organic materials. |
| LogP | -2.7 at 25℃ |
| CAS DataBase Reference | 7758-19-2(CAS DataBase Reference) |
| IARC | 3 (Vol. 52) 1991 |
| EPA Substance Registry System | Sodium chlorite (7758-19-2) |
SAFETY
Risk and Safety Statements
| Symbol(GHS) |      GHS03,GHS05,GHS06,GHS08,GHS09 |
|||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Signal word | Danger | |||||||||
| Hazard statements | H271-H302-H310-H314-H373-H400 | |||||||||
| Precautionary statements | P210-P260-P273-P280-P303+P361+P353-P305+P351+P338 | |||||||||
| Hazard Codes | O,Xn,T+,T,N | |||||||||
| Risk Statements | 8-22-24-32-34-9-26-25-14-36/37/38-21-50 | |||||||||
| Safety Statements | 17-26-36/37/39-45-50A-38-36-61-28 | |||||||||
| RIDADR | UN 2813 4.3/PG 3 | |||||||||
| WGK Germany | 2 | |||||||||
| RTECS | VZ4800000 | |||||||||
| TSCA | Yes | |||||||||
| HazardClass | 5.1 | |||||||||
| PackingGroup | II | |||||||||
| HS Code | 28289000 | |||||||||
| NFPA 704 |
|
Sodium chlorite price More Price(11)
| Manufacturer | Product number | Product description | CAS number | Packaging | Price | Updated | Buy |
|---|---|---|---|---|---|---|---|
| Sigma-Aldrich(India) | 244155 | Sodium chlorite technical grade, 80% | 7758-19-2 | 5G | ₹2944.4 | 2022-06-14 | Buy |
| Sigma-Aldrich(India) | 71388 | Sodium chlorite puriss. p.a., 80% (RT) | 7758-19-2 | 250G | ₹4622.28 | 2022-06-14 | Buy |
| Sigma-Aldrich(India) | 244155 | Sodium chlorite technical grade, 80% | 7758-19-2 | 100G | ₹4503.2 | 2022-06-14 | Buy |
| Sigma-Aldrich(India) | 71388 | Sodium chlorite puriss. p.a., 80% (RT) | 7758-19-2 | 1KG | ₹14743.65 | 2022-06-14 | Buy |
| Sigma-Aldrich(India) | 244155 | Sodium chlorite technical grade, 80% | 7758-19-2 | 1KG | ₹8833.2 | 2022-06-14 | Buy |
Sodium chlorite Chemical Properties,Uses,Production
Chemical Properties
It is a white crystalline powder or flakes that is readily soluble in water. It is slightly hygroscopic.
Uses
Sodium chlorite is used in the in situ generation of chlorine dioxide for stripping of textiles, bleaching, pulp and paper industries. It acts as a disinfectant in water treatment plant and as a preservative in eye drops. It is also used in contact lens cleaning solution and for sanitizing air ducts. It is associated with zinc chloride and used as a component in therapeutic rinses, toothpastes, mouth sprays and gels. It is utilized for the synthesis of 4-oxo-2-alkenoic acids from alkyl furans. Further, it is involved as a reagent in Pinnick oxidation reaction to prepare carboxylic acid from aldehydes.
Definition
ChEBI: Sodium chlorite is an inorganic sodium salt in which chlorite is the counterion. It has a role as an oxidising agent. It is an inorganic sodium salt and a chlorite salt.
General Description
Sodium chlorite appears as a white crystalline solid. Difficult to burn, but accelerates the burning of organic substances. Forms explosive mixtures with certain combustible materials. May explode under prolonged exposure to heat or fire. Used in water purification, to bleach wood pulp, textile, fats, oils; and for many other uses.
Air & Water Reactions
Soluble in water.
Reactivity Profile
SODIUM CHLORITE SOLUTION is an oxidizing agent. Can react with acids to form spontaneously explosive chlorine dioxide gas (ClO2). Reacts with ammonia to produce ammonium chlorite, which is shock-sensitive. Finely divided metallic or organic substances in dry mixture with chlorites are highly flammable and may be ignited on friction (Lab. Gov. Chemist 1965). A mixture of organic matter and solid sodium chlorite can be extremely sensitive to heat, impact, or friction (Diox Process 1949). Sodium chlorite reacts very violently with organic materials containing divalent sulfur or with free sulfur (may ignite).
Hazard
Flammable, strong oxidizing agent, dan- gerous fire and moderate explosion risk. (Solution) Strong irritant to skin and tissue.
Health Hazard
TOXIC; inhalation, ingestion or skin contact with material may cause severe injury or death. Contact with molten substance may cause severe burns to skin and eyes. Avoid any skin contact. Effects of contact or inhalation may be delayed. Fire may produce irritating, corrosive and/or toxic gases. Runoff from fire control or dilution water may be corrosive and/or toxic and cause pollution.
Fire Hazard
Non-combustible, substance itself does not burn but may decompose upon heating to produce corrosive and/or toxic fumes. Some are oxidizers and may ignite combustibles (wood, paper, oil, clothing, etc.). Contact with metals may evolve flammable hydrogen gas. Containers may explode when heated.
Agricultural Uses
Chlorite is a group of greenish clay minerals of variable composition (similar to mica in structure), which crystallizes in the monoclinic system. The term chlorite is derived from 'chloros', the Greek word for green.
Chlorites are composed of complex silicates of aluminum, magnesium and iron in combination with water.
These are often called 2:2 type clays because they are similar to the unit lattice of vermiculite. But strictly speaking, they are 2:1:1 type clays. A layer of chlorite has 2 silicate tetrahedral units, one alumina octahedral unit and one magnesium octahedral sheet. It has a low cation exchange capacity. Chlorites are most commonly found in low-grade metamorphic rocks. They also occur as secondary minerals in igneous rocks as alteration products of pyroxenes, amphiboles and micas.
Chlorites are infrequent in soils and when present, make up a small fraction of clay minerals. Chlorites are primary minerals and form vermiculites and smectites. Chlorites do not swell on wetting.
Safety Profile
Poison by ingestion. An experimental teratogen. Experimental reproductive effects. Questionable carcinogen with experimental carcinogenic data. Mutation data reported. May act as an irritant due to its oxidizing power. A powerful oxidzing agent; ignited by friction, heat, or shock. An explosive sensitive to impact or heating to 200'. Potentially explosive reaction with acids, oils, organic matter, oxahc acid + water, zinc. Violent reaction or iption with carbon (above 60'), ethylene glycol (at loo'), phosphorus (above SO0), sodum dithionate, sulfur-containing materials. Can react vigorously on contact with reducing materials. When heated to decomposition it emits highly toxic fumes of Cland NazO. Used as a bleachmg agent. See also CHLORITES.
Purification Methods
Crystallise the chlorite from hot water and store it in a cool place. It has also been crystallised from MeOH by counter-current extraction with liquid ammonia [Curti & Locchi Anal Chem 29 534 1957]. A major impurity is chloride ion which can be removed by recrystallisation from 0.001M NaOH. [Schmeisser in Handbook of Preparative Inorganic Chemistry (Ed. Brauer) Academic Press Vol I p 312 1963.]
Sodium chlorite Preparation Products And Raw materials
Raw materials
1of3
chevron_rightPreparation Products
1of2
chevron_right| Supplier | Tel | Country | ProdList | Advantage | Inquiry |
|---|---|---|---|---|---|
| UNILOSA INTERNATINAL PRIVATE LIMITED | +91-9999069917 +91-9999069917 | New Delhi, India | 152 | 58 | Inquiry |
| Zed Chem Private Ltd | +91-9327003226 +91-9327003226 | Maharashtra, India | 1 | 58 | Inquiry |
| Shree Chlorates Pvt Ltd | +91-9826047243 +91-7312490172 | Madhya Pradesh, India | 3 | 58 | Inquiry |
| Maulikem Products Pvt Ltd | +91-9824023069 +91-7926463625 | Gujarat, India | 2 | 58 | Inquiry |
| Scientific OEM | +91-22- 2343 7546 / 2341 3094 | New Delhi, India | 1996 | 38 | Inquiry |
| ALPHA CHEMIKA | +91-22-22061123 +91-22-66382501 | Mumbai, India | 1681 | 43 | Inquiry |
| CHEMSWORTH | +91-261-2397244 | New Delhi, India | 6707 | 30 | Inquiry |
| Maas Pharma Chemicals | +91-9958666224 | Delhi, India | 1607 | 58 | Inquiry |
| Otto Chemie Pvt. Ltd. | +91 9820041841 | Mumbai, India | 5873 | 58 | Inquiry |
| Alfa Aesar | 1 800 209 7001 | Maharashtra, India | 6913 | 58 | Inquiry |
| Supplier | Advantage |
|---|---|
| UNILOSA INTERNATINAL PRIVATE LIMITED | 58 |
| Zed Chem Private Ltd | 58 |
| Shree Chlorates Pvt Ltd | 58 |
| Maulikem Products Pvt Ltd | 58 |
| Scientific OEM | 38 |
| ALPHA CHEMIKA | 43 |
| CHEMSWORTH | 30 |
| Maas Pharma Chemicals | 58 |
| Otto Chemie Pvt. Ltd. | 58 |
| Alfa Aesar | 58 |
Related articles
- What is sodium chlorite used for?
- Sodium chlorite is a very efficient bleaching agent. Its oxidation potential allows a controlled bleaching that is not attaina....
- Mar 16,2024
- The wide range of applications of Sodium chlorite
- Sodium chlorite is a highly efficient bleaching agent and oxidative disinfectant, widely applied in bleaching of textile, fibe....
- Dec 18,2023
- Sodium Chlorite: Can It Be Used as Medicine?
- Don’t confuse sodium chlorite with sodium chloride. Sodium chloride (NaCl) is also called table salt. Sodium chloride is used ....
- Nov 20,2019
7758-19-2(Sodium chlorite)Related Search:
1of4
chevron_right




