Arformoterol tartrate

- CAS No.
- 200815-49-2
- Chemical Name:
- Arformoterol tartrate
- Synonyms
- 200-589-5;Aphmote tartrate;Aforterol tartrate;R,R-Form L-tartrate;Afromoterol tartrate;Arformoterol tartrate;(R,R)-Formoterol tartrate;(R,R)-ForMoterol-L-(+)-Tartrate;Formoterol Impurity 23 Tartrate;Formoterol Impurity 28 Tartrate
- CBNumber:
- CB91459486
- Molecular Formula:
- C19H24N2O4.C4H6O6
- Molecular Weight:
- 494.494
- MOL File:
- 200815-49-2.mol
- Modify Date:
- 2024/4/26 14:50:00
SAFETY
Risk and Safety Statements
| Symbol(GHS) |   GHS08,GHS06 |
|||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Signal word | Danger | |||||||||
| Hazard statements | H302-H361-H334-H331-H351-H372 | |||||||||
| Precautionary statements | P264-P270-P301+P312-P330-P501-P201-P202-P281-P308+P313-P405-P501-P260-P264-P270-P314-P501-P201-P202-P281-P308+P313-P405-P501-P261-P285-P304+P341-P342+P311-P501-P261-P271-P304+P340-P311-P321-P403+P233-P405-P501 | |||||||||
| NFPA 704 |
|
Arformoterol tartrate Chemical Properties,Uses,Production
Description
Sepracor’s Brovana®, a nebulized long acting bronchodilator, was launched in the U.S. in April 2007. The β2- adrenoceptor agonist is indicated for the twice-daily, longtermmaintenance treatment of bronchoconstriction in patients with chronic obstructive pulmonary disease (COPD), which includes chronic bronchitis and emphysema. It is the first long-acting nebulized bronchodilator approved by the FDA for this indication.
Uses
Anti-asthmatic and bronchodilator.
Synthesis
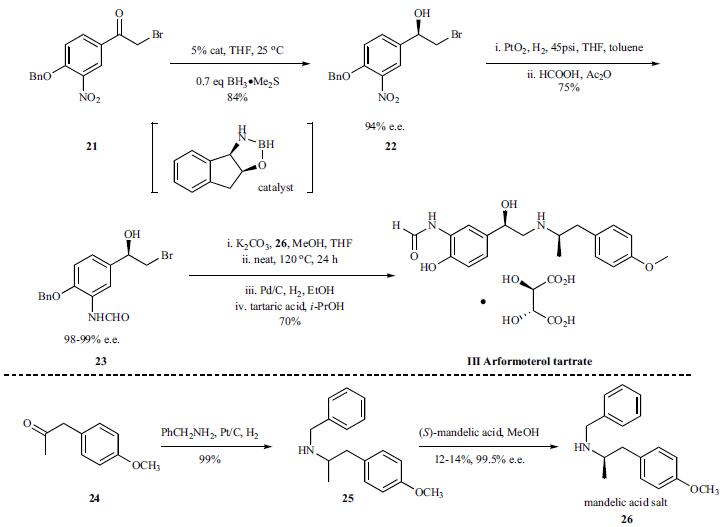
There are several reports on the synthesis of arformoterol. A large-scale synthesis of enantio/diastereomerically pure (R,R)-formoterol is cited here. Bromoalcohol 22 was synthesized in 84% yield with 94% e.e. through the catalytic enantioselective reduction of bromo ketone 21. The nitro functional group in 22 was reduced in quantitative yield by hydrogena-tion in the presence of Adams catalyst and the resulting aniline was isolated by filtration of the catalyst and removal of the solvent. In order to avoid auto-oxidation, the aniline was treated with a mixture of formic acid and acetic anhydride immediately after the removal of the platinum catalyst. Upon concentrating the reaction mixture, bromohydrin 23 crystallized and could be isolated in 75% yield with 98.6% e.e. It was further enriched to >99.5% e.e. by a single re-crystallization from ethylacetate. Next, a mixture of bromohydrin 23 and amine salt (R)-26-(S)-mandelic acid was treated with K2CO3 resulting in generation of the corresponding epoxide of 23 and liberation of the free base of (R)-26. After an aqueous work up to remove salts and mandelic acid, the reaction mixture was heated to 120??C to affect epoxide opening with the amine of 26. Removal of the benzyl protecting groups of the resulting crude product via catalytic hydrogenation followed by salt formation with tartaric acid afforded arformoterol tartrate (III) in 70% yield upon crystallization.
Arformoterol tartrate Preparation Products And Raw materials
Raw materials
Preparation Products
| Supplier | Tel | Country | ProdList | Advantage | Inquiry |
|---|---|---|---|---|---|
| Cipla Ltd | +912224826000 | Maharashtra, India | 133 | 58 | Inquiry |
| MELODY HEALTHCARE PVT LTD | +undefined2228780912 | Maharashtra, India | 20 | 58 | Inquiry |
| Ralington Pharma | +91-7948911722 +91-9687771722 | Gujarat, India | 1350 | 58 | Inquiry |
| GLENMARK LIFE SCIENCES LTD | +91 22 4018 9999 | New Delhi, India | 103 | 58 | Inquiry |
| Pharmaffiliates Analytics and Synthetics P. Ltd | +91-172-5066494 | Haryana, India | 6773 | 58 | Inquiry |
| Angle Biopharma | 08046067501 | Ahmedabad, India | 142 | 58 | Inquiry |
| SynZeal Research Pvt Ltd | +1 226-802-2078 | Gujarat, India | 6522 | 58 | Inquiry |
| Pharma Affiliates | 172-5066494 | Haryana, India | 6761 | 58 | Inquiry |
| CLEARSYNTH LABS LTD. | +91-22-45045900 | Hyderabad, India | 6351 | 58 | Inquiry |
| Henan Bao Enluo International TradeCo.,LTD | +86-17331933971 +86-17331933971 | China | 2503 | 58 | Inquiry |





