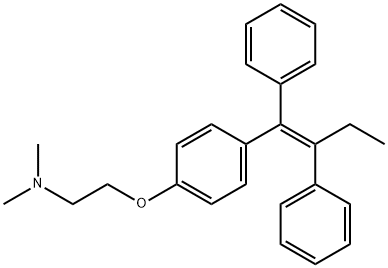
Tamoxifen
- CAS No.
- 10540-29-1
- Chemical Name:
- Tamoxifen
- Synonyms
- TAMOXIFEN BASE;Mammaton;Novaldex;amoxifen;z-tamoxifen;Genox;C07108;Tamoxen;ici47699;TAMOXIFEN
- CBNumber:
- CB9438781
- Molecular Formula:
- C26H29NO
- Molecular Weight:
- 371.51
- MOL File:
- 10540-29-1.mol
- MSDS File:
- SDS
- Modify Date:
- 2024/7/26 15:15:06
| Melting point | 97-98 °C(lit.) |
|---|---|
| Boiling point | 501.18°C (rough estimate) |
| Density | 1.0630 (rough estimate) |
| vapor pressure | 0Pa at 25℃ |
| refractive index | 1.6000 (estimate) |
| storage temp. | 2-8°C |
| solubility | H2O: insoluble <0.1% at 20°C |
| form | Solid |
| pka | pKa 8.71(H2O t = 25 I = 0.025) (Uncertain) |
| color | Crystals from pet ether |
| Water Solubility | Insoluble in water. Soluble in methanol, ethanol, propanol or propylene glycol.Soluble in dimethyl sulfoxide, dichloromethane and ethanol. Insoluble in water. |
| Merck | 13,9137 |
| Stability | Light Sensitive |
| InChIKey | NKANXQFJJICGDU-QPLCGJKRSA-N |
| LogP | 6.3 at 20℃ |
| CAS DataBase Reference | 10540-29-1(CAS DataBase Reference) |
| IARC | 1 (Vol. 66, 100A) 2012 |
| EPA Substance Registry System | Tamoxifen (10540-29-1) |
SAFETY
Risk and Safety Statements
| Symbol(GHS) |   GHS08,GHS09 |
|||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Signal word | Danger | |||||||||
| Hazard statements | H350-H360-H410 | |||||||||
| Precautionary statements | P201-P273-P308+P313 | |||||||||
| Hazard Codes | T,Xi | |||||||||
| Risk Statements | 45-60-61-64-36/37/38 | |||||||||
| Safety Statements | 53-45-36-26 | |||||||||
| WGK Germany | 3 | |||||||||
| RTECS | KR5919600 | |||||||||
| HS Code | 29221990 | |||||||||
| Toxicity | LD50 orl-rat: 4100 mg/kg DRFUD4 9,186,84 | |||||||||
| NFPA 704 |
|
Tamoxifen price More Price(6)
| Manufacturer | Product number | Product description | CAS number | Packaging | Price | Updated | Buy |
|---|---|---|---|---|---|---|---|
| Sigma-Aldrich(India) | T5648 | Tamoxifen ≥99% | 10540-29-1 | 1G | ₹28480.58 | 2022-06-14 | Buy |
| Sigma-Aldrich(India) | T5648 | Tamoxifen ≥99% | 10540-29-1 | 5G | ₹97998.73 | 2022-06-14 | Buy |
| Sigma-Aldrich(India) | T-143 | Tamoxifen solution 1?mg/mL in methanol, certified reference material, ampule of 1?mL, Cerilliant? | 10540-29-1 | 1ML | ₹12156.2 | 2022-06-14 | Buy |
| Sigma-Aldrich(India) | 85256 | Tamoxifen analytical standard | 10540-29-1 | 50MG | ₹5260.95 | 2022-06-14 | Buy |
| Sigma-Aldrich | 06734 | Tamoxifen certified reference material, TraceCERT? | 10540-29-1 | 50MG | ₹8844.03 | 2022-06-14 | Buy |
Tamoxifen Chemical Properties,Uses,Production
Description
In 1966, ICI Pharmaceuticals (now AstraZeneca) first synthesized tamoxifen in the hope of developing a morning-after contraceptive pill. The UK patent for this compound was in place in 1962, whereas the US patent was repeatedly denied until the 1980s. Tamoxifen was approved for a fertility treatment but it was not proven as useful in regulating human contraception. Even though there was a link between estrogen and breast cancer, developing a cancer treatment was not a priority at the time. In 1971, the first clinical study showed a convincing effect of tamoxifen in treating advanced breast cancer. From 1971 to 1977, this drug was neither clinically nor financially remarkable. In 1980s, however, publications first showed that tamoxifen, in addition to chemotherapy, improved survival for patients with early stage breast cancer. In 1998, the meta-analysis by the Oxford-based Early Breast Cancer Trialists’ Collaborative Group showed that tamoxifen did indeed save lives in early breast cancer. In 2001, tamoxifen sales were over $1.024 billion. Since the expiration of the patent in 2002, it is now widely available as a generic drug. By 2004, tamoxifen was the best selling hormonal drug for the treatment of breast cancer.
Chemical Properties
White Crystalline Solid
Uses
A nonsteroidal estrogen antagonist of interest in the treatment of some forms of breast cancer. Tamoxifen is a Protein Kinase C inhibitor, and induces apoptosis in human malignant glioma cell lines
Indications
Tamoxifen (Nolvadex) is a synthetic antiestrogen used in the treatment of breast cancer. Normally, estrogens act by binding to a cytoplasmic protein receptor, and the resulting hormone–receptor complex is then translocated into the nucleus, where it induces the synthesis of ribosomal RNA (rRNA) and messenger RNA (mRNA) at specific sites on the DNA of the target cell. Tamoxifen also avidly binds to estrogen receptors and competes with endogenous estrogens for these critical sites. The drug–receptor complex has little or no estrogen agonist activity.Tamoxifen directly inhibits growth of human breast cancer cells that contain estrogen receptors but has little effect on cells without such receptors.
World Health Organization (WHO)
Tamoxifen is an anti-estrogen agent used mainly to treat breast cancer. Tamoxifen is listed in the WHO Model List of Essential Drugs.
General Description
Tamoxifen is a selective estrogen response modifier (SERM), protein kinase C inhibitor and anti-angiogenetic factor. Tamoxifen is a prodrug that is metabolized to active metabolites 4-hydroxytamoxifen (4-OHT) and endoxifen by cytochrome P450 isoforms CYP2D6 and CYP3A4. In breast cancer, the gene repressor activity of tamoxifen against ERBB2 is dependent upon PAX2. Blocks estradiol-stimulated VEGF production in breast tumor cells.
Mechanism of action
Tamoxifen is slowly absorbed, and maximum serum levels are achieved 4 to 7 hours after oral administration. The drug is concentrated in estrogen target tissues, such as the ovaries, uterus, vaginal epithelium, and breasts. Hydroxylation and glucuronidation of the aromatic rings are the major pathways of metabolism; excretion occurs primarily in the feces.
Pharmacokinetics
Circulating levels of the demethylated metabolite at steady state are up to twice the level of the parent drug, because the elimination half-life of N-demethyl tamoxifen is 14 days, compared with 7 days for tamoxifen. Tamoxifen demonstrates only weak estrogenic effects at several sites, including the endometrium and bone, and on the lipid profile. Tamoxifen undergoes rapid N-dem ethylation to its major metabolite, N-dem ethyltamoxifen, by CYP3A4 and via CYP2D6 to its minor metabolite, 4-hydroxytam oxifen. Evidence suggests that 4-hydroxytamoxifen is the active metabolite of tamoxifen, with a higher binding affinity than the parent drug for the ER
Clinical Use
Tamoxifen is a SERM that is used as an antiestrogen in the treatment of estrogen-dependent breas Tcancer following prim ary treatment (c hemotherapy and/or surgery).
Side effects
Tamoxifen administration is associated with few toxic side effects, most frequently hot flashes (in 10–20% of patients) and occasionally vaginal dryness or discharge. Mild nausea, exacerbation of bone pain, and hypercalcemia may occur.
Safety Profile
Confirmed human carcinogen. Moderately toxic by ingestion and intraperitoneal routes. Human systemic effects by an unspecified route: nausea or vomiting, leukopenia, thrombocytopenia, and skin changes. An experimental teratogen. Other experimental reproductive effects. Human mutation data reported. When heated to decomposition it emits toxic fumes of NOx.
Carcinogenicity
Tamoxifen is known to be a human carcinogen based on sufficient evidence of carcinogenicity from studies in humans.
Tamoxifen Preparation Products And Raw materials
| Supplier | Tel | Country | ProdList | Advantage | Inquiry |
|---|---|---|---|---|---|
| Pharmaffiliates Analytics and Synthetics P. Ltd | +91-172-5066494 | Haryana, India | 6773 | 58 | Inquiry |
| MANGALAM DRUGS AND ORGANICS LIMITED | 08147509329 | Uttar Pradesh, India | 50 | 58 | Inquiry |
| A.J Chemicals | 91-9810153283 | New Delhi, India | 6124 | 58 | Inquiry |
| CLEARSYNTH LABS LTD. | +91-22-45045900 | Hyderabad, India | 6351 | 58 | Inquiry |
| Hebei Bonster Technology Co.,Limited | +8613315996897 | China | 796 | 58 | Inquiry |
| Hong Kong Excellence Biotechnology Co., Ltd. | +86-86-18838029171 +8618126314766 | China | 887 | 58 | Inquiry |
| Hebei Mojin Biotechnology Co., Ltd | +8613288715578 | China | 12458 | 58 | Inquiry |
| Hebei Guanlang Biotechnology Co,.LTD | +86-19930503253; +8619930503252 | China | 5835 | 58 | Inquiry |
| Wuhan Cell Pharmaceutical Co., Ltd | +86-13129979210 +86-13129979210 | China | 376 | 58 | Inquiry |
| Hebei Anlijie Biotechnology Co., Ltd | +8619031013551 | China | 145 | 58 | Inquiry |
Related articles
- Tamoxifen: Origins, Clinical Trials and Dose
- Tamoxifen revolutionized early breast cancer treatment with established 5-year adjuvant therapy. Lower doses like 5 mg/day sho....
- Feb 28,2024
- The introduction of Tamoxifen
- Tamoxifen is an antiestrogenic trans isomer of a substituted triphenyl ethylene. This drug is commonly used for treating pre- ....
- Oct 13,2023





