Nevirapine
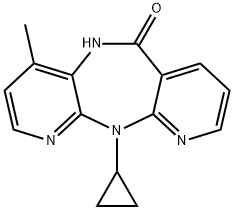
- CAS No.
- 129618-40-2
- Chemical Name:
- Nevirapine
- Synonyms
- D00435;Nevirne;VIRAMUNE;BI-RG-587;Viramnune;Nevirapil;Weilaping;BIRG 0587;HSDB 7164;AIDS005653
- CBNumber:
- CB9743074
- Molecular Formula:
- C15H14N4O
- Molecular Weight:
- 266.3
- MOL File:
- 129618-40-2.mol
- MSDS File:
- SDS
- Modify Date:
- 2024/5/24 18:18:52
| Melting point | 247°C |
|---|---|
| Boiling point | 409.5°C (rough estimate) |
| Density | 1.1300 (rough estimate) |
| refractive index | 1.6200 (estimate) |
| Flash point | 9℃ |
| storage temp. | 2-8°C |
| solubility | DMSO: ≥22mg/mL |
| form | powder |
| pka | 2.8(at 25℃) |
| color | white to tan |
| Water Solubility | 0.1g/L(temperature not stated) |
| Merck | 14,6490 |
| BCS Class | 2 |
| CAS DataBase Reference | 129618-40-2(CAS DataBase Reference) |
| EPA Substance Registry System | 6H-Dipyrido[3,2-b:2',3'-e][1,4]diazepin-6-one, 11-cyclopropyl-5,11-dihydro-4-methyl- (129618-40-2) |
SAFETY
Risk and Safety Statements
| Symbol(GHS) |    GHS02,GHS06,GHS08 |
|||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Signal word | Danger | |||||||||
| Hazard statements | H225-H301+H311+H331-H370 | |||||||||
| Precautionary statements | P210-P260-P280-P301+P310-P311 | |||||||||
| Hazard Codes | Xi | |||||||||
| Risk Statements | 36/37/38 | |||||||||
| Safety Statements | 26-36-37/39 | |||||||||
| RIDADR | UN1230 - class 3 - PG 2 - Methanol, solution | |||||||||
| WGK Germany | 2 | |||||||||
| RTECS | JM5562500 | |||||||||
| HS Code | 29339900 | |||||||||
| NFPA 704 |
|
Nevirapine price More Price(4)
| Manufacturer | Product number | Product description | CAS number | Packaging | Price | Updated | Buy |
|---|---|---|---|---|---|---|---|
| Sigma-Aldrich(India) | SML0097 | Nevirapine | 129618-40-2 | 10MG | ₹9536.83 | 2022-06-14 | Buy |
| Sigma-Aldrich(India) | SML0097 | Nevirapine | 129618-40-2 | 50MG | ₹38829.28 | 2022-06-14 | Buy |
| Sigma-Aldrich(India) | PHR1757 | Nevirapine Pharmaceutical Secondary Standard; Certified Reference Material | 129618-40-2 | 1G | ₹12080.7 | 2022-06-14 | Buy |
| TCI Chemicals (India) | N0922 | Nevirapine min. 98.0 % | 129618-40-2 | 200MG | ₹11500 | 2022-05-26 | Buy |
Nevirapine Chemical Properties,Uses,Production
Description
Nevirapine and its analogues exhibit antiretroviral effect against azothymidine-resistant HIV strains. Nevirapine in combination with ZDV and ddI produced approximately 18% higher CD4 cell counts and a decrease in viral load compared with patients who took ZDV and ddI. Nevirapine is recommended with nucleosides for patients infected with HIV-1 who have experienced clinical or immunologic deterioration. The significant side effects of nevirapine are liver dysfunction and skin rashes.
Chemical Properties
Crystalline Solid
Uses
Labelled Nevirapine , a potent (IC50=84nM) and selective non-nucleoside inhibitor of HIV-1 reverse transcriptase. Antiviral.;Labeled Nevirapine, intended for use as an internal standard for the quantification of Nevirapine by GC- or LC-mass spectrometry.
Indications
Nevirapine (Viramune) is approved for the treatment of HIV infection in adults and children as part of a combination therapy. During the first 12 weeks of treatment, patients must be closely monitored for the development of potentially fatal hepatic toxicity (i.e., hepatitis, hepatic necrosis, and hepatic failure) and skin reactions (i.e., Stevens-Johnson syndrome, toxic epidermal necrolysis, and hypersensitivity reactions). Although these toxicities are rare, common side effects include mild to moderate rash, fever, nausea, fatigue, headache, and elevated liver enzymes.
Definition
ChEBI: A dipyridodiazepine that is 5,11-dihydro-6H-dipyrido[3,2-b:2',3'-e][1,4]diazepine which is substituted by methyl, oxo, and cyclopropyl groups at positions 4, 6, and 11, respectively. A non-nucleoside reverse tr nscriptase inhibitor with activity against HIV-1, it is used in combination with other antiretrovirals for the treatment of HIV infection.
Acquired resistance
One or more changes within the HIV reverse transcriptase at amino acid positions 100, 103, 106, 108, 181, 188 and 190 are associated with resistance. These point mutations have also been implicated, either alone or in combination, in HIV resistance to other non-nucleoside reverse transcriptase inhibitors.
General Description
Nevirapine (Viramune) is more than 90% absorbed by theoral route and is widely distributed throughout the body. Itdistributes well into breast milk and crosses the placenta.Transplacental concentrations are about 50% those ofserum. The drug is extensively transformed by cytochromeP450 (CYP) to inactive hydroxylated metabolites; it mayundergo enterohepatic recycling.
Pharmaceutical Applications
A synthetic heterocyclic compound formulated for oral use as anhydrous compound or as the hemihydrate in a liquid oral suspension.
Mechanism of action
Nevirapine is a dipyridodiazepinone derivative that binds directly to RT . Thus, it blocks RNA- and DNA-dependent polymerase activities by causing a disruption of the enzyme's catalytic site. The activity of nevirapine does not compete with template or nucleoside triphosphate. The HIV-2 RT and human DNA polymerases are not inhibited by nevirapine. The 50% inhibitory concentration ranged within 10 to 100 nM against HIV-1.
Pharmacokinetics
Oral absorption: c. 93%
Cmax 200 mg twice daily: c. 5.74 mg/L
Cmin 200 mg twice daily: c. 2.88 mg/L
Plasma half-life: c. 36 h
Volume of distribution: c. 1.21 L/kg
Plasma protein binding: c. 60%
Absorption and distribution
Nevirapine is orally very well absorbed and widely distributed. CNS penetration is good and the semen:plasma ratio is in the range of 0.6–1. It is distributed into breast milk.
Metabolism and excretion
It is extensively metabolized by cytochrome P450 enzymes into a number of hydroxylated intermediates that are subsequently conjugated with glucuronide. Around 81% of the dose is excreted in urine (<5% as unchanged compound) and 10% in feces. There is no significant change in the pharmacokinetics in renal impairment. It is contraindicated in patients with severe hepatic impairment; caution should be exercised in patients with moderate hepatic dysfunction.
Clinical Use
Treatment of HIV-1 infection in adults and children over 2 months old (in
combination with other antiretroviral therapies)
Reduction of maternal transmission of HIV to the fetus (recommended
only for use in HIV-infected treatment-naive women in labor who have
had no prior HIV therapy)
Side effects
Life-threatening hepatic events, including fulminant hepatitis, have been observed in treatment-naive patients, generally within the first few weeks of treatment, but sometimes later. Approximately half the patients also develop skin rash, with or without fever or constitutional symptoms. Women with elevated CD4 counts (>250 cells/mm3) appear to be at highest risk. Men with pretreatment CD4 counts >400 cells/mm3 are also at increased risk. These risks exist in the absence of underlying hepatic abnormalities and, in some cases, hepatic injury continues to progress despite discontinuation of treatment. Treatment should stop, and not be restarted, in patients with clinical evidence of hepatitis. A starting dose of 200 mg per day, with escalation to full dose if no adverse reaction occurs, reduces the frequency of reaction. Single doses given to mothers or infants for prevention of perinatal HIV infection appear safe.
Nevirapine Preparation Products And Raw materials
Raw materials
1of4
chevron_rightPreparation Products
| Supplier | Tel | Country | ProdList | Advantage | Inquiry |
|---|---|---|---|---|---|
| Varanous Labs Pvt Ltd | +91-7036248882 +91-7036248882 | Hyderabad, India | 1541 | 58 | Inquiry |
| Dodhia Group | +91-9619867345 +91-9619867345 | Mumbai, India | 129 | 58 | Inquiry |
| Aurobindo Pharma Limited | +914066725000 | Telangana, India | 112 | 58 | Inquiry |
| Cipla Ltd | +912224826000 | Maharashtra, India | 133 | 58 | Inquiry |
| Laurus Labs Ltd | +91-4066594333 +91-4039804333 | Telangana, India | 50 | 58 | Inquiry |
| Ralington Pharma | +91-7948911722 +91-9687771722 | Gujarat, India | 1350 | 58 | Inquiry |
| Arene Lifesciences Limited | +91-9849847907 +91-8455241148 | Telangana, India | 55 | 58 | Inquiry |
| Alfa Omega Pharma | +91-8050045945 +91-9972665399 | Maharashtra, India | 126 | 58 | Inquiry |
| Vishrudh laboratories pvt ltd | +91-9666132889 +91-8097369002 | Telangana, India | 117 | 58 | Inquiry |
| Hetero Drugs Limited | +91-4023704923 +91-4023704923 | Telangana, India | 296 | 58 | Inquiry |
| Supplier | Advantage |
|---|---|
| Varanous Labs Pvt Ltd | 58 |
| Dodhia Group | 58 |
| Aurobindo Pharma Limited | 58 |
| Cipla Ltd | 58 |
| Laurus Labs Ltd | 58 |
| Ralington Pharma | 58 |
| Arene Lifesciences Limited | 58 |
| Alfa Omega Pharma | 58 |
| Vishrudh laboratories pvt ltd | 58 |
| Hetero Drugs Limited | 58 |
Related articles
- The reverse transcriptase of HIV-1---Nevirapine
- Nevirapine (previously known as BI-RG-587) is a dipyridodiazepinone derivative and a potent and highly specific inhibitor of t....
- Apr 19,2022
- Application of Nevirapine as the Non-Nucleoside Reverse Transcriptase Inhibitor
- Nevirapine is a non-Nucleoside Reverse Transcriptase Inhibitor (NNRTI) that blocks RNA-dependence and the activity of DNA-depe....
- Oct 15,2019





