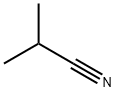
Isobutyronitrile
- CAS No.
- 78-82-0
- Chemical Name:
- Isobutyronitrile
- Synonyms
- 2-METHYLPROPIONITRILE;2-METHYLPROPANENITRILE;isiobutyronitrile;ISOPROPYL CYANIDE;Azobis (2-methylpropionitrile);iso-C3H7CN;2-Cyanopropane;Isobutyronitril;Isopropylcyanid;Isopropylkyanid
- CBNumber:
- CB9854381
- Molecular Formula:
- C4H7N
- Molecular Weight:
- 69.11
- MOL File:
- 78-82-0.mol
- Modify Date:
- 2023/11/28 16:31:44
| Melting point | -72 °C (lit.) |
|---|---|
| Boiling point | 107-108 °C (lit.) |
| Density | 0.770 g/mL at 20 °C (lit.) |
| vapor density | 2.38 (vs air) |
| vapor pressure | 100 mm Hg ( 54.4 °C) |
| refractive index |
n |
| Flash point | 39 °F |
| storage temp. | Flammables area |
| solubility | slightly soluble in water and acetone, very soluble in alcohol and ether |
| form | Liquid |
| color | Clear colorless to light yellow |
| Water Solubility | 35 g/L (20 ºC) |
| Merck | 14,5156 |
| BRN | 1340512 |
| Dielectric constant | 23.9(Ambient) |
| Stability | Stable. Flammable. May form explosive mixtures with air. Incompatible with strong oxidizing agents. |
| CAS DataBase Reference | 78-82-0(CAS DataBase Reference) |
| NIST Chemistry Reference | Propanenitrile, 2-methyl-(78-82-0) |
| EPA Substance Registry System | Isobutyronitrile (78-82-0) |
SAFETY
Risk and Safety Statements
| Symbol(GHS) |   GHS02,GHS06 |
|||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Signal word | Danger | |||||||||
| Hazard statements | H225-H300+H310+H330 | |||||||||
| Precautionary statements | P210-P233-P240-P280-P303+P361+P353-P304+P340+P310 | |||||||||
| Hazard Codes | F,T | |||||||||
| Risk Statements | 11-23/24/25-36/37/38 | |||||||||
| Safety Statements | 9-16-24/25-26-36/37/39-45-23 | |||||||||
| OEB | A | |||||||||
| OEL | TWA: 8 ppm (22 mg/m3) | |||||||||
| RIDADR | UN 2284 3/PG 2 | |||||||||
| WGK Germany | 2 | |||||||||
| RTECS | TZ4900000 | |||||||||
| Hazard Note | Highly Flammable/Toxic | |||||||||
| TSCA | Yes | |||||||||
| HazardClass | 3 | |||||||||
| PackingGroup | II | |||||||||
| HS Code | 29269095 | |||||||||
| Toxicity | LD50 (mg/kg): 25 i.p. in mice; 200 orally in rats (Zeller); LD50 orally in male mice: 0.3652 mmol/kg (Tanii) | |||||||||
| NFPA 704 |
|
Isobutyronitrile price More Price(8)
| Manufacturer | Product number | Product description | CAS number | Packaging | Price | Updated | Buy |
|---|---|---|---|---|---|---|---|
| Sigma-Aldrich(India) | I15602 | Isobutyronitrile 99% | 78-82-0 | 100ML | ₹1948.5 | 2022-06-14 | Buy |
| Sigma-Aldrich(India) | I15602 | Isobutyronitrile 99% | 78-82-0 | 1L | ₹3712.98 | 2022-06-14 | Buy |
| Sigma-Aldrich(India) | 538248 | Isobutyronitrile 99.6% | 78-82-0 | 100ML | ₹3745.45 | 2022-06-14 | Buy |
| Sigma-Aldrich(India) | 538248 | Isobutyronitrile 99.6% | 78-82-0 | 1L | ₹6170.25 | 2022-06-14 | Buy |
| TCI Chemicals (India) | I0112 | Isobutyronitrile min. 98.0 % | 78-82-0 | 25ML | ₹1900 | 2022-05-26 | Buy |
Isobutyronitrile Chemical Properties,Uses,Production
Chemical Properties
Colorless liquid with a foul odor. Insoluble in water, easily soluble in ethanol and ether.
Uses
Isobutyronitrile can be derived from isobutyraldehyde. It is used in organic synthesis, as a catalyst in the polymerization of ethylene and in the petroleum industry as a gasoline additive. Isobutyronitrile is also used to synthesize the intermediate 2-isopropyl-4-methyl-6-hydroxypyrimidine of the organophosphorus insecticide diazinon.
Production Methods
Isobutyronitrile is prepared from isobutyraldehyde by cyanation with ammonia.
Production Methods
Isobutyronitrile is usually obtained by the catalytic gas-phase reaction of isobutyraldehyde or isobutanol with ammonia. Its major use is the synthesis of the insecticide diazinon.
General Description
A clear colorless liquid. Flash point 47°F. Less dense than water. Vapors heavier than air. Toxic oxides of nitrogen produced during combustion.
Air & Water Reactions
Highly flammable. Slightly soluble in water.
Reactivity Profile
Isobutyronitrile is incompatible with the following: Oxidizers, reducing agents, strong acids & bases .
Hazard
Toxic by ingestion, inhalation, and skin absorption.
Health Hazard
Isobutyronitrile is considered highly hazardous and full precautions should be taken to prevent skin contact or inhalation of vapor. Inhaled isobutyronitrile is about 2.4 times as toxic as acetonitrile in rats. may be fatal if inhaled, swallowed, or absorbed through skin. Contact may cause burns to skin and eyes. (Non-Specific -- Nitriles) Primarily, they are skin and eye irritants. Large doses cause collapse and stop breathing. In order to protect workers, the recommended TWA limit is obtained by dividing that for acetonitrile by the factor 2.4. NIOSH has therefore recommended that employee exposure should not exceed 8 p.p.m. (22 mg/m3) for either compound as a TLV-TWA.
Fire Hazard
Vapor may explode if ignited in an enclosed area. Toxic oxides of nitrogen are produced during combustion. Isobutyronitrile is a flammable/combustible material and may be ignited by heat, sparks, or flames. Vapors may travel to a source of ignition and flash back. Container may explode in heat of fire. Vapor explosion and poison hazard indoors, outdoors or in sewers. Runoff to sewer may create fire or explosion hazard. Hazardous polymerization may not occur.
Metabolism
Thiocyanate was present in the urine of rats dosed orally with isobutyronitrile.
Purification Methods
Shake the nitrile with conc HCl (to remove isonitriles), then with water and aqueous NaHCO3. After a preliminary drying with silica gel or Linde type 4A molecular sieves, it is shaken or stirred with CaH2 until hydrogen evolution ceases, then decanted and distilled from P2O5 (not more than 5g/L, to minimize gel formation) or Drierite (b 101-103o/760mm). Finally it is refluxed with, and slowly distilled from CaH2 (5g/L), taking precautions to exclude moisture. [Beilstein 2 H 294, 2 I 129, 2 II 263, 2 III 655, 2 IV 853.]
Isobutyronitrile Preparation Products And Raw materials
Raw materials
1of2
chevron_rightPreparation Products
1of3
chevron_right78-82-0(Isobutyronitrile)Related Search:
1of4
chevron_right




