N-NITROSODIMETHYLAMINE
|
- ₹0
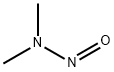
- Product name: N-NITROSODIMETHYLAMINE
- CAS: 62-75-9
- MF: C2H6N2O
- MW: 74.08
- EINECS:200-549-8
- MDL Number:MFCD00002053
- Synonyms:n-methyl-n-nitroso-methanamin ;N-Methyl-N-nitrosomethanamine;N-methyl-N-nitroso-Methanamine ;n-nitroso-dimethylamin ;N-Nitroso-N,N-Dimethylamine;Rcra waste number P082;rcrawastenumberp082 ;DIMETHYLNITROSAMINE
| Manufacturer | Product number | Product description | Packaging | Price | Updated | Buy |
|---|
Properties
Melting point :<25 °C
Boiling point :153 °C774 mm Hg(lit.)
Density :1.01 g/mL(lit.)
vapor pressure :5 mm Hg ( 20 °C)
refractive index :n20/D 1.437(lit.)
Flash point :142 °F
storage temp. :2-8°C
solubility :Soluble in solvents (U.S. EPA, 1985), including ethanol and ether (Weast, 1986)
pka :-3.63±0.70(Predicted)
form :Yellow liquid
color :Colourless to Light Yellow
Water Solubility :Miscible (Mirvish et al., 1976)
Merck :13,6671
Henry's Law Constant :0.143 at 25 °C (estimated using a solubility of 1,000 g/L)
Dielectric constant :54.0(20℃)
Stability :Stability Stable, but light-sensitive. Combustible. Incompatible with strong oxidizing agents, strong bases, strong reducing agents.
CAS DataBase Reference :62-75-9(CAS DataBase Reference)
IARC :2A (Vol. 17, Sup 7) 1987
EPA Substance Registry System :N-Nitrosodimethylamine (62-75-9)
Boiling point :153 °C774 mm Hg(lit.)
Density :1.01 g/mL(lit.)
vapor pressure :5 mm Hg ( 20 °C)
refractive index :n
Flash point :142 °F
storage temp. :2-8°C
solubility :Soluble in solvents (U.S. EPA, 1985), including ethanol and ether (Weast, 1986)
pka :-3.63±0.70(Predicted)
form :Yellow liquid
color :Colourless to Light Yellow
Water Solubility :Miscible (Mirvish et al., 1976)
Merck :13,6671
Henry's Law Constant :0.143 at 25 °C (estimated using a solubility of 1,000 g/L)
Dielectric constant :54.0(20℃)
Stability :Stability Stable, but light-sensitive. Combustible. Incompatible with strong oxidizing agents, strong bases, strong reducing agents.
CAS DataBase Reference :62-75-9(CAS DataBase Reference)
IARC :2A (Vol. 17, Sup 7) 1987
EPA Substance Registry System :N-Nitrosodimethylamine (62-75-9)
Safety Information
| Symbol(GHS): |
  
|
||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Signal word: | Danger | ||||||||||||||||||||||||||||||||||||||||||
| Hazard statements: |
|
||||||||||||||||||||||||||||||||||||||||||
| Precautionary statements: |
|
Description
Nitrosamines are chemicals that possess the general structure R1N(R2)-N=O. These chemicals have been used in the manufacture of rocket fuel, cosmetics, pesticides, and polymers. Research studies dating back to the 1950s have demonstrated that most nitrosamines (>90%) possess some degree of toxicity.Of particular interest is the nitrosamine N-nitrosodimethylamine (DMN). This semi-volatile organic compound is highly toxic and is a suspected human carcinogen. At higher doses, it has been shown to be a hepatotoxin that causes liver fibrosis and cancer in several animal species. Its toxic effects were first reported by British scientists John Barnes and Peter Magee in 1956 during their screening of chemicals that were being used as solvents in the dry cleaning industry. Since then, levels of DMN have been detected in food, drinking water, soil, and air.
The consumption of contaminated food and water accounts for the majority of the exposure to DMN. Most nonoccupational exposure to DMN is a result of chemical reactions between precursors that form DMN, rather than the industrial utilization of the chemical itself. For example, after DMN was discovered in beer in Europe in the 1970s, it was shown that the direct firedrying of the malt barley used was the DMN source. Modifications to the drying procedure were able to substantially reduce the levels of DMN found in beer today. Additionally, the formation of DMN was attributed to an outbreak of liver cancers and disorders in livestock that were fed herring meal in Norway in the 1970s. Subsequent studies showed that reaction of dimethylamine (naturally occurring in fish) with nitrosating reagents derived from sodium nitrite (a widely used preservative) formed the NMD responsible for the liver toxicities. These studies caused widespread concern over the use of sodium nitrite in many foods consumed by humans. To ameliorate the formation of DMN in food caused by sodium nitrite, manufactures now add antioxidants such as ascorbic acid (vitamin C), erythorbic acid (an isomer of ascorbic acid), and a-tocopherol (vitamin E).
DMN has been found in groundwater in many states. Major sources of DMN in groundwater include rocket fuel production, and water treatment via chlorination or chloroamination of wastewater. The removal of DMN from drinking water usually involves ultraviolet treatment or reverse osmosis.
Related product price
- Lomustine
₹3700-19614.9 - N-NITROSODIBUTYLAMINE
₹4761.4-14408.08 - Streptozocin
₹1791-158088.3






