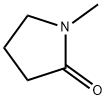
N-Methyl-2-pyrrolidone
- CAS No.
- 872-50-4
- Chemical Name:
- N-Methyl-2-pyrrolidone
- Synonyms
- NMP;N-METHYLPYRROLIDONE;1-METHYL-2-PYRROLIDINONE;1-methyl-pyrrolidin-2-one;1-METHYL-2-PYRROLIDONE;N-METHYL-2-PYRROLIDINONE;1-Methylpyrrolidin-2-one;N-METHYLPYRROLIDINONE;Methylpyrrolidone;N-Methylpyrrolidon
- CBNumber:
- CB2402488
- Molecular Formula:
- C5H9NO
- Molecular Weight:
- 99.13
- MOL File:
- 872-50-4.mol
- MSDS File:
- SDS
- Modify Date:
- 2024/6/15 22:11:33
| Melting point | -24 °C (lit.) |
|---|---|
| Boiling point | 202 °C (lit.) 81-82 °C/10 mmHg (lit.) |
| Density | 1.028 g/mL at 25 °C (lit.) |
| vapor density | 3.4 (vs air) |
| vapor pressure | 0.29 mm Hg ( 20 °C) |
| refractive index |
n |
| Flash point | 187 °F |
| storage temp. | Store at +5°C to +30°C. |
| solubility | ethanol: miscible0.1ML/mL, clear, colorless (10%, v/v) |
| form | Liquid |
| pka | -0.41±0.20(Predicted) |
| color | ≤20(APHA) |
| Odor | Slight amine odor |
| PH Range | 7.7 - 8.0 |
| PH | 8.5-10.0 (100g/l, H2O, 20℃) |
| explosive limit | 1.3-9.5%(V) |
| Water Solubility | >=10 g/100 mL at 20 ºC |
| Sensitive | Hygroscopic |
| λmax | 283nm(MeOH)(lit.) |
| Merck | 14,6117 |
| BRN | 106420 |
| Dielectric constant | 32.200000000000003 |
| Stability | Stable, but decomposes upon exposure to light. Combustible. Incompatible with strong oxidizing agents, strong acids, reducing agents, bases. |
| InChIKey | SECXISVLQFMRJM-UHFFFAOYSA-N |
| LogP | -0.46 at 25℃ |
| CAS DataBase Reference | 872-50-4(CAS DataBase Reference) |
| NIST Chemistry Reference | 2-Pyrrolidinone, 1-methyl-(872-50-4) |
| EPA Substance Registry System | N-Methyl-2-pyrrolidone (872-50-4) |
SAFETY
Risk and Safety Statements
| Symbol(GHS) |   GHS07,GHS08 |
|||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Signal word | Danger | |||||||||
| Hazard statements | H315-H319-H335-H360D | |||||||||
| Precautionary statements | P202-P261-P264-P302+P352-P305+P351+P338-P308+P313 | |||||||||
| Hazard Codes | T,Xi | |||||||||
| Risk Statements | 45-65-36/38-36/37/38-61-10-46 | |||||||||
| Safety Statements | 41-45-53-62-26 | |||||||||
| WGK Germany | 1 | |||||||||
| RTECS | UY5790000 | |||||||||
| F | 3-8-10 | |||||||||
| Autoignition Temperature | 518 °F | |||||||||
| TSCA | Y | |||||||||
| HS Code | 2933199090 | |||||||||
| Toxicity | LD50 orally in Rabbit: 3598 mg/kg LD50 dermal Rabbit 8000 mg/kg | |||||||||
| NFPA 704 |
|
N-Methyl-2-pyrrolidone price More Price(67)
| Manufacturer | Product number | Product description | CAS number | Packaging | Price | Updated | Buy |
|---|---|---|---|---|---|---|---|
| Sigma-Aldrich(India) | M79603 | 1-Methyl-2-pyrrolidinone ReagentPlus?, 99% | 872-50-4 | 1L | ₹3279.98 | 2022-06-14 | Buy |
| Sigma-Aldrich(India) | 8.06072 | 1-Methyl-2-pyrrolidone EMPLURA? | 872-50-4 | 8060720521 | ₹2290 | 2022-06-14 | Buy |
| Sigma-Aldrich(India) | M79603 | 1-Methyl-2-pyrrolidinone ReagentPlus?, 99% | 872-50-4 | 2.5L | ₹6635.73 | 2022-06-14 | Buy |
| Sigma-Aldrich(India) | 8.06072 | 1-Methyl-2-pyrrolidone EMPLURA? | 872-50-4 | 8060722521 | ₹9250 | 2022-06-14 | Buy |
| Sigma-Aldrich(India) | PHR1352 | N-Methylpyrrolidone Pharmaceutical Secondary Standard; Certified Reference Material | 872-50-4 | 2G | ₹8281.13 | 2022-06-14 | Buy |
N-Methyl-2-pyrrolidone Chemical Properties,Uses,Production
Description
N-Methyl-2-pyrrolidone is an aprotic solvent with a wide range of applications: petrochemical processing, surface coating, dyes and pigments, industrial and domestic cleaning compounds, and agricultural and pharmaceutical formulations. It is mainly an irritant, but has also caused several cases of contact dermatitis in a small electrotechnical company.
Chemical Properties
N-Methyl-2-pyrrolidone is a colourless or light yellow liquid with an amine odour. It can undergo a number of chemical reactions even though it is accepted as a stable solvent. It is resistant to hydrolysis under neutral conditions, but strong acid or base treatment results in ring opening to 4-methyl aminobutyric acid. N-Methyl-2-pyrrolidone can be reduced to 1-methyl pyrrolidine with borohydride. Treatment with chlorinating agents results in amide formation,an intermediate which can undergo further substitution, while treatment with amyl nitrate yields the nitrate. Olefins can be added to the 3 position by treatment first with oxalic esters, then with appropriate aldehyes (Hort and Anderson 1982).
Uses
N-Methyl-2-pyrrolidone is a polar solvent that is used in organic chemistry and polymer chemistry. Large scale applications include the recovery and purification of acetylenes, olefins, and diolefins, gas purification, and aromatics extraction from feedstocks.N-Methyl-2-pyrrolidone is a versatile industrial solvent. NMP is currently approved for use only in veterinary pharmaceuticals. The determination of the disposition and metabolism of NMP in the rat will contribute toward understanding the toxicology of this exogenous chemical which man may likely be exposed to in increasing amounts.
Uses
N-Methyl-2-pyrrolidone, is useful for spectrophotometry, chromatography and ICP-MS detection.
Production Methods
N-Methyl-2-pyrrolidone is manufactured by the reaction of buytrolactone with methylamine (Hawley 1977). Other processes include preparation by hydrogenation of solutions of maleic or succinic acids with methylamine (Hort and Anderson 1982). Manufacturers of this chemical include Lachat Chemical, Inc, Mequon, Wisconsin and GAF Corporation, Covert City, California.
Definition
ChEBI: N-methylpyrrolidin-2-one is a member of the class of pyrrolidine-2-ones that is pyrrolidin-2-one in which the hydrogen attached to the nitrogen is replaced by a methyl group. It has a role as a polar aprotic solvent. It is a N-alkylpyrrolidine, a lactam and a member of pyrrolidin-2-ones.
General Description
N-Methyl-2-Pyrrolidone (NMP) is a powerful, aprotic solvent with high solvency, and low volatility. This colorless, high boiling, high flash point and low vapor pressure liquid carries a mild amine-like odor. NMP has high chemical and thermal stability and is completely miscible with water at all temperatures. NMP can serve as a co-solvent with water, alcohols, glycol ethers, ketones, and aromatic/chlorinated hydrocarbons. NMP is both recyclable by distillation and readily biodegradable. NMP is not found on the Hazardous Air Pollutants (HAPs) list of the 1990 Clean Air Act Amendments.
Air & Water Reactions
Soluble in water.
Reactivity Profile
This amine is a very mild chemical base. N-Methyl-2-pyrrolidone does tend to neutralize acids to form salts plus water. The amount of heat that is evolved per mole of amine in a neutralization is largely independent of the strength of the amine as a base. Amines may be incompatible with isocyanates, halogenated organics, peroxides, phenols (acidic), epoxides, anhydrides, and acid halides. Flammable gaseous hydrogen is generated by amines in combination with strong reducing agents, such as hydrides.
Hazard
Severe skin and eye irritant. Explosive lim-its 2.2–12.2%.
Health Hazard
Inhalation of hot vapors can irritate nose and throat. Ingestion causes irritation of mouth and stomach. Contact with eyes causes irritation. Repeated and prolonged skin contact produces a mild, transient irritation.
Fire Hazard
Special Hazards of Combustion Products: Toxic oxides of nitrogen may be formed in fire.
Industrial uses
1) N-Methyl-2-pyrrolidone is used as a general dipolar aprotic solvent, stable and unreactive;
2) for extraction of aromatic hydrocarbons from lubricating oils;
3) for carbon dioxide removal in ammonia generators;
4) as a solvent for polymerization reactions and polymers;
5) as a paint stripper;
6) for pesticide formulations (USEPA 1985).
Other non-industrial uses of N-Methyl-2-pyrrolidone are based on its properties as a dissociating solvent suitable for electrochemical and physical chemical studies (Langan and Salman 1987). Pharmaceutical applications make use of the properties of N-Methyl-2-pyrrolidone as a penetration enhancer for a more rapid transfer of substances through the skin (Kydoniieus 1987; Barry and Bennett 1987; Akhter and Barry 1987). N-Methyl-2-pyrrolidone has been approved as a solvent for slimicide application to food packaging materials (USDA 1986).
Contact allergens
N-Methyl-2-pyrrolidone is an aprotic solvent with a wide range of applications: petrochemical processing, surface coating, dyes and pigments, industrial and domestic cleaning compounds, and agricultural and pharmaceutical formulations. It is mainly an irritant, but it can cause severe contact dermatitis due to prolonged contact.
Safety Profile
Poison by intravenous route. Moderately toxic by ingestion and intraperitoneal routes. Mildly toxic by skin contact. An experimental teratogen. Experimental reproductive effects. Mutation data reported. Combustible when exposed to heat, open flame, or powerful oxidizers. To fight fire, use foam, CO2, dry chemical. When heated to decomposition it emits toxic fumes of NOx.
Carcinogenicity
Rats were exposed to N-Methyl-2-pyrrolidone vapor at 0, 0.04, or 0.4 mg/L for 6 h/day, 5 days/week for 2 years.Male rats at 0.4 mg/L showed slightly reduced mean body weight. No life-shortening toxic or carcinogenic effects were observed in rats exposed for 2 years to either 0.04 or 0.4mg/L of N-Methyl-2-pyrrolidone. By the dermal route, a group of 32 mice received an initiation dose of 25mg of N-Methyl-2-pyrrolidone followed 2 weeks later by applications of the tumor promoter phorbol myristate acetate, three times a week, for more than 25 weeks. Dimethylcarbamoyl chloride and dimethylbenzanthracene served as positive controls. Although the N-Methyl-2-pyrrolidone group had three skin tumors, this response was not considered significant when compared with that of the positive controls.
Metabolic pathway
Rats are administered radio-labeled N-methyl-2- pyrrolidinone (NMP), and the major route of excretion by rats is via the urine. The major metabolite, representing 70-75% of the administered dose, is 4-(methylamino)butenoic acid. This unsaturated intact product may be formed from the elimination of water, and a hydroxyl group may be present on the metabolite prior to acid hydrolysis.
Metabolism
Male Sprague-Dawley rats were given a single intraperitoneal injection (45 mg/kg) of radiolabeled 1-methyl-2-pyrrolidone. Plasma levels of radioactivity and compound were monitored for six hours and the results suggested a rapid distribution phase which was followed by a slow elimination phase. The major amount of label was excreted in the urine within 12 hours and accounted for approximately 75% of the labelled dose. Twenty-four hours after dosage, cumulative excretion (urine) was approximately 80% of the dose. Both ring- and methyl-labeled species were used, as well as both [14C]- and [3H]-labeled l-methyl-2-pyrrolidone. The initial labeled ratios were maintained during the first 6 hours after dosage. After 6 hours, the liver and intestines were found to contain the highest accumulations of radioactivity, approximately 2-4% of the dose. Little radioactivity was noted in the bile or respired air. High performance liquid chromatography of urine showed the presence of one major and two minor metabolites. The major metabolite (70-75% of the administered radioactive dose) was analyzed by liquid chromatography-mass spectrometry and gas chromatography-mass spectrometry and was proposed to be a 3- or 5-hydroxy-l-methyl-2-pyrrolidone (Wells 1987).
Purification Methods
Dry the pyrrolidone by removing water as the *benzene azeotrope. Fractionally distil at 10 torr through a 100-cm column packed with glass helices. [Adelman J Org Chem 29 1837 1964, McElvain & Vozza J Am Chem Soc 71 896 1949.] The hydrochloride has m 86-88o (from EtOH or Me2CO/EtOH) [Reppe et al. Justus Liebigs Ann Chem 596 1 1955]. [Beilstein 21 II 213, 21 III/IV 3145, 21/6 V 321.]
N-Methyl-2-pyrrolidone Preparation Products And Raw materials
Raw materials
Preparation Products
1of6
chevron_right| Supplier | Tel | Country | ProdList | Advantage | Inquiry |
|---|---|---|---|---|---|
| Alkyl Amines Chemicals Limited | +91-91-22-24930710 +91-9075727921 | Mumbai, India | 65 | 58 | Inquiry |
| Vardhaman P Golechha | 9704422000 | Telangana, India | 294 | 58 | Inquiry |
| JIVICHEM SYNTHESIS PVT LTD | +91-7028392790 +91-7028392790 | Maharashtra, India | 337 | 58 | Inquiry |
| JSK Chemicals | +919879767970 | Gujarat, India | 3756 | 58 | Inquiry |
| BASF India Limited | +91-2262785600 +91-2262785600 | Maharashtra, India | 209 | 58 | Inquiry |
| Manas Petro Chem | +91-9004912487 +91-9004912487 | Maharashtra, India | 17 | 58 | Inquiry |
| Kairav Chemofarbe Industries Ltd. KCIL | +91-222596836 +91-8879003729 | Maharashtra, India | 9 | 58 | Inquiry |
| S D Fine Chem Limited | +91-9323715856 +91-9323715856 | Maharashtra, India | 368 | 58 | Inquiry |
| Cognizant Chemical Pvt Ltd | +91-9819500500 +91-9819500500 | Maharashtra, India | 14 | 58 | Inquiry |
| AKASH PHARMA EXPORTS | +91-9388123451 +91-9846039283 | Kerela, India | 470 | 58 | Inquiry |
| Supplier | Advantage |
|---|---|
| Alkyl Amines Chemicals Limited | 58 |
| Vardhaman P Golechha | 58 |
| JIVICHEM SYNTHESIS PVT LTD | 58 |
| JSK Chemicals | 58 |
| BASF India Limited | 58 |
| Manas Petro Chem | 58 |
| Kairav Chemofarbe Industries Ltd. KCIL | 58 |
| S D Fine Chem Limited | 58 |
| Cognizant Chemical Pvt Ltd | 58 |
| AKASH PHARMA EXPORTS | 58 |
Related articles
- How to synthesis N-Methyl-2-pyrrolidone
- Based on the raw materials used, several processes such as succinonitrile route, condensation of γ-butyrolactone and methylami....
- Mar 12,2024
- Toxic hygroscopic contact reaction to N-methyl-2-pyrrolidone
- Exposure to organic solvents such as NMP may result in neurological toxicity, inhalation in high concentrations causing headac....
- Nov 23,2023
- N-Methyl-2-pyrrolidone: Application, absorption, metabolism and toxicity
- N-Methyl-2-pyrrolidone is a colorless liquid, and is used in the petrochemical and plastics industries as a solvent. It is eas....
- Apr 11,2023
872-50-4(N-Methyl-2-pyrrolidone)Related Search:
1of4
chevron_right




