Nickel(II) oxide
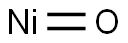
- CAS No.
- 1313-99-1
- Chemical Name:
- Nickel(II) oxide
- Synonyms
- NICKEL MONOXIDE;bunsenite;C.I. 77777;(II) oxide;OXIDICNICKEL;Nickel 0xide;nickd monoxide;CATALYST R1-10;A nickel oxide;mononickeloxide
- CBNumber:
- CB2358284
- Molecular Formula:
- NiO
- Molecular Weight:
- 74.69
- MOL File:
- 1313-99-1.mol
- MSDS File:
- SDS
- Modify Date:
- 2024/5/28 19:59:05
| Melting point | 1960 °C |
|---|---|
| Density | 6.67 g/mL at 25 °C(lit.) |
| solubility | insoluble in H2O; soluble in acid solutions |
| form | Powder/Solid |
| Specific Gravity | 6.67 |
| color | green |
| Water Solubility | insoluble |
| Merck | 14,6512 |
| Exposure limits |
ACGIH: TWA 0.2 mg/m3 NIOSH: IDLH 10 mg/m3; TWA 0.015 mg/m3 |
| Stability | Stable. Incompatible with strong acids. |
| InChI | InChI=1S/Ni.O |
| InChIKey | GNRSAWUEBMWBQH-UHFFFAOYSA-N |
| SMILES | O=[Ni] |
| CAS DataBase Reference | 1313-99-1(CAS DataBase Reference) |
| EPA Substance Registry System | Nickel(II) oxide (1313-99-1) |
SAFETY
Risk and Safety Statements
| Symbol(GHS) |   GHS07,GHS08 |
|||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Signal word | Danger | |||||||||
| Hazard statements | H317-H350i-H372-H413 | |||||||||
| Precautionary statements | P202-P260-P273-P280-P302+P352-P308+P313 | |||||||||
| Hazard Codes | T,Xi | |||||||||
| Risk Statements | 49-43-53-48/23-36/37 | |||||||||
| Safety Statements | 53-45-61-36/37-26 | |||||||||
| RIDADR | 3288 | |||||||||
| WGK Germany | 1 | |||||||||
| RTECS | QR8400000 | |||||||||
| TSCA | Yes | |||||||||
| HS Code | 28254000 | |||||||||
| NFPA 704 |
|
Nickel(II) oxide price More Price(57)
| Manufacturer | Product number | Product description | CAS number | Packaging | Price | Updated | Buy |
|---|---|---|---|---|---|---|---|
| Sigma-Aldrich(India) | 637130 | Nickel(II) oxide nanopowder, <50?nm particle size (TEM), 99.8% trace metals basis | 1313-99-1 | 25G | ₹4900 | 2022-06-14 | Buy |
| Sigma-Aldrich(India) | 921602 | NiO nanoparticle ink solution 2.5?wt. %, viscosity 1.3?cP | 1313-99-1 | 10ML | ₹10684.28 | 2022-06-14 | Buy |
| Sigma-Aldrich(India) | 637130 | Nickel(II) oxide nanopowder, <50?nm particle size (TEM), 99.8% trace metals basis | 1313-99-1 | 100G | ₹12410 | 2022-06-14 | Buy |
| Sigma-Aldrich(India) | 774545 | Nickel(II) oxide nanowires, diam. × L ~20?nm × 10?μm | 1313-99-1 | 500MG | ₹18260 | 2022-06-14 | Buy |
| Sigma-Aldrich(India) | 637130 | Nickel(II) oxide nanopowder, <50?nm particle size (TEM), 99.8% trace metals basis | 1313-99-1 | 250G | ₹23330 | 2022-06-14 | Buy |
Nickel(II) oxide Chemical Properties,Uses,Production
Chemical Properties
Nickel(II) oxide is Brownish black or black powder.
Uses
Nickel(II) oxide is used in the preparation of nickel alloys including nickel steel alloys, manufacture of glass and porcelain paints. Further nickel oxide is the main component in nickel-iron battery (Edison battery), nickel-cadmium rechargeable battery, and also used in ceramic industry for making frits and porcelain glazes. It also acts as a hydrogenation catalyst. NiO/CNTs (nickel oxide/carbon nanotubes) could be a potential cathode catalyst for oxygen reduction reaction (ORR) in microbial fuel cells (MFCs). It is blended with other high purity oxides and used in a variety of semiconductor applications such as thermistors and varistors.
Preparation
Nickel oxide is prepared by heating pure nickel powder with oxygen at a temperature above 400°C. In some commercial processes, green nickel oxide is made by heating a mixture of nickel powder and water in air at 1,000°C. Adding some nickel oxide to the above mixture enhances the rate of reaction. An alternative method of preparation of the green oxide involves thermal decomposition of an oxo acid salt of nickel at elevated temperatures. Thus, nickel nitrate, nickel sulfate or, more conveniently, nickel carbonate when heated at 1,000°C, yields the green oxide. The black oxide, on the other hand, is produced at a lower temperature from incomplete calcination of the carbonate or nitrate salt at 600°C. The oxygen content of the black form is slight-ly greater than its green counterpart.
Reactions
Nickel(II) oxide is insoluble in water but soluble in acids as long as it has not been ignited at a high temperature (under these latter conditions it is converted into grey-black octahedra having a metallic lustre). It reacts reversibly with hydrogen, the reaction NiO+H2 ? Ni+H20 proceeding from left to right at relatively low temperatures in a stream of hydrogen.
General Description
Nickel(II) oxide (NiO) is a metal oxide based nanomaterial with a good semiconducting property. Nanosized nickel oxide can be found in a variety of morphologies which include nanoflowers, spheres, wires, and tubes. It exhibits high performance in applications which require charge transfer and charge transport based processes. It can be prepared by a variety of physical and thermal methods such as sol-gel, hydrothermal and solvothermal techniques.
Hazard
Confirmed carcinogen.
Safety Profile
Confirmed carcinogen with experimental carcinogenic and tumorigenic data. Poison by intratracheal, intravenous, and subcutaneous routes. Mutation data reported. Can react violently with fluorine, hydrogen peroxide, hydrogen sulfide, iodine, barium oxide + air. See also NICKEL COMPOUNDS.
Nickel(II) oxide Preparation Products And Raw materials
Raw materials
1of2
chevron_rightPreparation Products
| Supplier | Tel | Country | ProdList | Advantage | Inquiry |
|---|---|---|---|---|---|
| GLR Innovations | +91 9891111994 | New Delhi, India | 4542 | 58 | Inquiry |
| Himalaya Industries | +91-9011992000 +91-9011882000 | Maharashtra, India | 10 | 58 | Inquiry |
| Metal Chem India | +91-8999856889 +91-9011882000 | Maharashtra, India | 46 | 58 | Inquiry |
| Eastmen Chemicals | +91-2228727766 +91-2228730294 | Maharashtra, India | 19 | 58 | Inquiry |
| Monopoly Innovations Pvt. Ltd. | +undefined-9321425897 +undefined-9819745149 | Maharashtra, India | 45 | 58 | Inquiry |
| Euroasia Trans Continental | +91 22 56349035-36 | New Delhi, India | 519 | 47 | Inquiry |
| ALPHA CHEMIKA | +91-22-22061123 +91-22-66382501 | Mumbai, India | 1681 | 43 | Inquiry |
| Scientific OEM | +91-22- 2343 7546 / 2341 3094 | New Delhi, India | 1996 | 38 | Inquiry |
| S.K. CHEMICAL INDUSTRIES | +91-22-66370299 Ext 21 / 91-22-23437380 Ext 25 | New Delhi, India | 262 | 30 | Inquiry |
| FINAR LTD | +91 - 2717 - 616717 | New Delhi, India | 660 | 58 | Inquiry |
| Supplier | Advantage |
|---|---|
| GLR Innovations | 58 |
| Himalaya Industries | 58 |
| Metal Chem India | 58 |
| Eastmen Chemicals | 58 |
| Monopoly Innovations Pvt. Ltd. | 58 |
| Euroasia Trans Continental | 47 |
| ALPHA CHEMIKA | 43 |
| Scientific OEM | 38 |
| S.K. CHEMICAL INDUSTRIES | 30 |
| FINAR LTD | 58 |
Related articles
- Uses of Nickel Monoxide
- Nickel monoxide (synonyms: Nickel(II) oxide, Nickel protoxide or Green nickel oxide) is a green to black colored, inorganic co....
- Oct 10,2019
1313-99-1(Nickel(II) oxide)Related Search:
1of4
chevron_right




