Adefovir
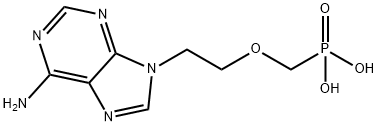
- CAS No.
- 106941-25-7
- Chemical Name:
- Adefovir
- Synonyms
- PMEA;GS-393;gs0393;Adefovi;ADEFOVIR;A Duff Vee;Adefovir98%;Adefovir API;adefovir ,97%;Adefovir(PMEA)
- CBNumber:
- CB2698137
- Molecular Formula:
- C8H12N5O4P
- Molecular Weight:
- 273.19
- MOL File:
- 106941-25-7.mol
- MSDS File:
- SDS
- Modify Date:
- 2024/3/29 23:06:43
| Melting point | >260°C |
|---|---|
| Boiling point | 632.5±65.0 °C(Predicted) |
| Density | 1.88 |
| storage temp. | -20°C |
| solubility | 0.1 M NaOH: ≥5mg/mL |
| pka | pKa1 2.0, pKa2 6.8(at 25℃) |
| form | powder |
| color | white to beige |
| InChIKey | SUPKOOSCJHTBAH-UHFFFAOYSA-N |
| CAS DataBase Reference | 106941-25-7(CAS DataBase Reference) |
Adefovir price More Price(2)
Adefovir Chemical Properties,Uses,Production
Chemical Properties
Pale Brown Solid
Uses
Adefovir has been used to study its anti-retro viral effect on porcine endogenous retrovirus (PERV) activity.
Acquired resistance
It has a lower propensity to induce drug resistance than lamivudine. Clinical trials of patients receiving 48 weeks of therapy did not identify any cases of resistance. Longer courses yield resistant strains of HBV with mutations in the DNA polymerase gene; other rare variants of resistant strains have been identified. Lamivudine-resistant strains of HBV retain susceptibility to adefovir.
Pharmaceutical Applications
A nucleotide analog of adenosine monophosphate, administered orally as its prodrug, adefovir dipivoxil.
Pharmacokinetics
Oral absorption: c. 60%
Cmax 10 mg/kg oral: 18.4 ng/mL
Plasma half-life: c. 7.5 h.
Volume of distribution: 392 mL/kg
Plasma protein binding: Not known
The prodrug is metabolized to adefovir, which is excreted by the kidneys and therefore requires dose adjustment in patients with impaired renal function. It does not induce cytochrome P450 at standard doses and does not influence the metabolism or plasma concentrations of the other licensed medications used in the treatment of hepatitis B.
Clinical Use
Treatment of chronic hepatitis B virus infection in patients >12 years of age
Side effects
It is generally well tolerated, with headache, pharyngitis, abdominal pain and peripheral neuropathy being the most common side effects. Nephrotoxicity has been observed in some patients, with those receiving higher doses and longer courses of therapy at greater risk. Exacerbation of hepatitis has been reported in patients immediately following discontinuation of treatment. Most exacerbations occur within 12 weeks of stopping therapy, and elevations of alanine aminotransferase (ALT) up to 10 times the upper limit of normal can be observed in over 25% of patients. Lactic acidosis has been reported in a few patients and is an indication for immediate discontinuation.
Adefovir Preparation Products And Raw materials
| Supplier | Tel | Country | ProdList | Advantage | Inquiry |
|---|---|---|---|---|---|
| KPS Chemicals And Pharmaceuticals | +91-9870076629 +91-8469484608 | Gujarat, India | 33 | 58 | Inquiry |
| CLEARSYNTH LABS LTD. | +91-22-45045900 | Hyderabad, India | 6351 | 58 | Inquiry |
| A.J Chemicals | 91-9810153283 | New Delhi, India | 6124 | 58 | Inquiry |
| Luna Chemical Industry Private Limited | 08048372457Ext 777 | Mumbai, India | 3 | 58 | Inquiry |
| Luna Chemical Industries Private Limited | 08048372754Ext 585 | Vadodara, India | 22 | 58 | Inquiry |
| Pharmaffiliates Analytics and Synthetics P. Ltd | +91-172-5066494 | Haryana, India | 6773 | 58 | Inquiry |
| SynZeal Research Pvt Ltd | +1 226-802-2078 | Gujarat, India | 6522 | 58 | Inquiry |
| Pharma Affiliates | 172-5066494 | Haryana, India | 6761 | 58 | Inquiry |
| Trignokem International | +91 (40) 2717-0709 | New Delhi, India | 380 | 50 | Inquiry |
| Hebei Yanxi Chemical Co., Ltd. | +86-17531190177; +8617531190177 | China | 6011 | 58 | Inquiry |






