Fondaparinux sodium
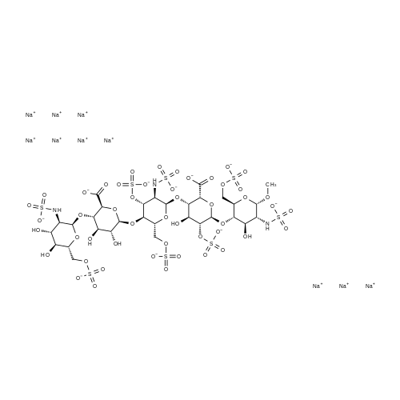
- CAS No.
- 114870-03-0
- Chemical Name:
- Fondaparinux sodium
- Synonyms
- Fondaparin SodiuM;Arixtra;Fondaparinux and intermediates;decasodium 6-[[6-[[2-carboxylato-4-hydroxy-6-[[4-hydroxy-6-methoxy-5-(sulfonatoamino)-2-(sulfonatooxymethyl)-3-oxanyl]oxy]-5-sulfonatooxy-3-oxanyl]oxy]-5-(sulfonatoamino)-4-sulfonatooxy-2-(sulfonatoox;Methyl O-2-Deoxy-6-O-sulfo-2-(sulfoamino)-α-D-glucopyranosyl-(14)-O-β- D-glucopyranuronosyl-(14)-O-2-deoxy-3,6-di-O-sulfo-2-(sulfoamino)-α-D-glucopyranosyl-(14)-O-2-O-sulfo-α-L-idopyranuronosyl-(14)-2-deoxy-2-(sulfoamino)-α-D-glucopyra;a-D-Glucopyranoside, methylO-2-deoxy-6-O-sulfo-2-(sulfoamino)-a-D-glucopyranosyl-(14)-O-b-D-glucopyranuronosyl-(14)-O-2-deoxy-3,6-di-O-sulfo-2-(sulfoamino)-a-D-glucopyranosyl-(14)-O-2-O-sulfo-a-L-idopyranuronosyl-(14)-2-deoxy-2-(sulfoamino)-,6-(hydrog;decasodiuM (2R,3S,4S,5R,6R)-3-{[(2R,3R,4R,5R,6R)-5-{[(2R,3R,4R,5S,6S)-6-carboxylato-5-{[(2R,3R,4R,5S,6R)-4,5-dihydroxy-3-(sulfonatoaMino)-6-[(sulfonatooxy)Methyl]oxan-2-yl]oxy}-3,4-dihydroxyoxan-2-yl]oxy}-3-(sulfonatoaMino)-4-(sulfonatooxy)-6-[(sulfonatoo;(2S,3S,4R,5R,6R)-6-(((2R,3R,4R,5R,6R)-6-(((2R,3S,4S,5R,6R)-2-Carboxy-4-hydroxy-6-(((2R,3S,4R,5R,6S)-4-hydroxy-6-methoxy-5-(sulfoamino)-2-((sulfooxy)methyl)tetrahydro-2H-pyran-3-yl)oxy)-5-(sulfooxy)tetrahydro-2H-pyran-3-yl)oxy)-5-(sulfoamino)-4-(sulfooxy)-;D01844;Arixtra (tn)
- CBNumber:
- CB41456602
- Molecular Formula:
- C31H43N3O49S8.10Na
- Molecular Weight:
- 1728.08
- MOL File:
- 114870-03-0.mol
- MSDS File:
- SDS
- Modify Date:
- 2024/3/14 18:29:10
SAFETY
Risk and Safety Statements
| Symbol(GHS) |  GHS07 |
|||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Signal word | Warning | |||||||||
| Hazard statements | H332 | |||||||||
| Precautionary statements | P261-P271-P304+P340-P312 | |||||||||
| HS Code | 3822000002 | |||||||||
| NFPA 704 |
|
Fondaparinux sodium Chemical Properties,Uses,Production
Description
Fondaparinux sodium was first introduced in the US for prophylaxis of deep vein thrombosis which may lead to pulmonary embolism following major orthopaedic surgery. Fondaparinux is the first of a new class of antithrombic agents distinct from low molecular weight heparin (LMWH) and heparin. This entirely synthetic molecule is a copy of the heparin pentasaccharide sequence, the shortest fragment able to catalyze antithrombin lllmediated inhibition of factor Xa thereby inhibiting thrombin generation without antithrombin action. Fondaparinux does not display significant effects on coagulation tests (such as activated partial thromboplastin time and prothrombin time), does not bind to platelet factor 4 or promote heparin-induced thrombocytopenia. In phase III studies, fondaparinux significantly reduced the incidence of thromboembolism following orthopedic surgery, with an overall risk reduction of 50% in comparison to the LMWH, enoxaparin. Following subcutaneous administration, fondaparinux has a nearly complete bioavailability, a rapid onset of action, a prolonged half-life (17.2 h) enabling once daily dosing and is not metabolized preceeding renal excretion. The drug appears to be generally safe, with haemoragic complications either comparable to or higher than those for LMWH.
Chemical Properties
White Powder (after lyophilisation)
Uses
Fondaparinux sodium has been used to test its neutralizing effect towards enterovirus D68-947 infection. It may be used in ultraviolet photodissociation (UVPD) measurements.
Definition
ChEBI: An organic sodium salt, being the decasodium salt of fondaparinux.
Indications
Fondaparinux sodium injection is a Factor Xa inhibitor (anticoagulant) indicated for:
Prophylaxis of deep vein thrombosis (DVT) in patients undergoing hip fracture surgery (including extended prophylaxis), hip replacement surgery, knee replacement surgery, or abdominal surgery.
Treatment of DVT or acute pulmonary embolism (PE) when administered in conjunction with warfarin.
Side effects
Fondaparinux sodium is a prescription medication and as such, most people do not have serious side effects. However, pain, bruising, redness, and swelling at the injection site may occur, as well as headache, nausea, vomiting, swelling of the hands and feet, or fever. A few serious side effects may occur: easy bleeding or bruising; dark urine, yellowing of the eyes or skin; shortness of breath, coughing up blood, chest pain, unusual dizziness, fainting, fast or irregular heartbeat; joint or muscle pain; confusion. Very few patients may experience allergic reactions (e.g. rash, itching or swelling (especially on face/tongue/throat); severe dizziness, difficulty breathing).
Fondaparinux sodium Preparation Products And Raw materials
Raw materials
Preparation Products
| Supplier | Tel | Country | ProdList | Advantage | Inquiry |
|---|---|---|---|---|---|
| Dr. Reddy's Laboratories Ltd | +91-4049002900 +91-4049002900 | Hyderabad, India | 165 | 58 | Inquiry |
| EAGLE CHEMICAL WORKS | +91-22263303652226330365 +91-2226330365 | Maharashtra, India | 61 | 58 | Inquiry |
| Emmennar Pharma Pvt Ltd | +91-7680064455 +91-9966766666 | Telangana, India | 31 | 58 | Inquiry |
| Manus Aktteva | +91 (79) 6512-3395 | New Delhi, India | 581 | 34 | Inquiry |
| AUROBINDO PHARMA LTD | +91 (40) 6672 1200 | New Delhi, India | 209 | 58 | Inquiry |
| MYLAN API US LLC | 080 6672 8000 | New Delhi, India | 44 | 58 | Inquiry |
| DR REDDYS LABORATORIES LTD | +91-40-49002900 | New Delhi, India | 201 | 58 | Inquiry |
| CLEARSYNTH LABS LTD. | +91-22-45045900 | Hyderabad, India | 6351 | 58 | Inquiry |
| A.J Chemicals | 91-9810153283 | New Delhi, India | 6124 | 58 | Inquiry |
| Pharmaffiliates Analytics and Synthetics P. Ltd | +91-172-5066494 | Haryana, India | 6773 | 58 | Inquiry |
| Supplier | Advantage |
|---|---|
| Dr. Reddy's Laboratories Ltd | 58 |
| EAGLE CHEMICAL WORKS | 58 |
| Emmennar Pharma Pvt Ltd | 58 |
| Manus Aktteva | 34 |
| AUROBINDO PHARMA LTD | 58 |
| MYLAN API US LLC | 58 |
| DR REDDYS LABORATORIES LTD | 58 |
| CLEARSYNTH LABS LTD. | 58 |
| A.J Chemicals | 58 |
| Pharmaffiliates Analytics and Synthetics P. Ltd | 58 |
Related articles
- Fondaparinux sodium: Pharmacokinetic Profile and Drug Interaction
- Fondaparinux sodium exhibits favorable pharmacokinetics, minimal drug interactions, and reliable antithrombotic efficacy, maki....
- Feb 19,2024
- Mechanism of action of Fondaparinux sodium
- rixtra, also known as Fondaparinux sodium, is a drug that inhibits Factor X, a protein involved in blood clotting.
- Jun 7,2023





