Minocycline hydrochloride
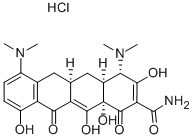
- CAS No.
- 13614-98-7
- Chemical Name:
- Minocycline hydrochloride
- Synonyms
- MINOCYCLINE HCL;Arestin;Dynacin;minocyclinechloride;[4S-(4alpha,4aalpha,5aalpha,12aalpha)]-4,7-Bis(dimethylamino)-1,4,4a,5,5a,6,11,12a-octahydro-3,10,12,12a-tetrahydroxy-1,11-dioxonaphthacene-2-carboxamide monohydrochloride;minocyn;MINOCIN;tri-mino;MYNOCINE;Minomycin
- CBNumber:
- CB6472139
- Molecular Formula:
- C23H28ClN3O7
- Molecular Weight:
- 493.94
- MOL File:
- 13614-98-7.mol
- MSDS File:
- SDS
- Modify Date:
- 2024/7/5 17:59:12
| Melting point | 205-210° (dec) |
|---|---|
| Boiling point | 813℃ |
| Flash point | >110°(230°F) |
| storage temp. | 2-8°C |
| solubility | Sparingly soluble in water, slightly soluble in ethanol (96 per cent). It dissolves in solutions of alkali hydroxides and carbonates. |
| form | crystalline |
| color | yellow |
| Water Solubility | Freely soluble in water |
| Merck | 14,6202 |
| Stability | Light Sensitive |
| InChIKey | GLMUAFMGXXHGLU-VQAITOIOSA-N |
| SMILES | [C@@]12([H])C[C@@]3([H])C(C(=O)C4C(O)=CC=C(N(C)C)C=4C3)=C(O)[C@]1(O)C(=O)C(C(=O)N)=C(O)[C@H]2N(C)C.Cl |&1:0,3,21,31,r| |
| LogP | 0.808 (est) |
| CAS DataBase Reference | 13614-98-7(CAS DataBase Reference) |
SAFETY
Risk and Safety Statements
| Symbol(GHS) |   GHS07,GHS08 |
|||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Signal word | Danger | |||||||||
| Hazard statements | H317-H360-H362-H303-H315-H319-H335 | |||||||||
| Precautionary statements | P201-P202-P260-P263-P264-P270-P272-P280-P302+P352+P333+P313+P363-P308+P313-P405-P501-P261-P280a-P304+P340-P305+P351+P338-P501a | |||||||||
| Hazard Codes | Xi | |||||||||
| Risk Statements | 36/37/38 | |||||||||
| Safety Statements | 26-36 | |||||||||
| RIDADR | 3249 | |||||||||
| WGK Germany | 3 | |||||||||
| RTECS | QI7630500 | |||||||||
| HazardClass | 6.1(b) | |||||||||
| PackingGroup | III | |||||||||
| HS Code | 29413020 | |||||||||
| Toxicity | human,TDLo,oral,14286ug/kg/10 (14.286mg/kg),LUNGS, THORAX, OR RESPIRATION: RESPIRATORY OBSTRUCTIONLUNGS, THORAX, OR RESPIRATION: DYSPNEASKIN AND APPENDAGES (SKIN): "DERMATITIS, OTHER: AFTER SYSTEMIC EXPOSURE",Archives of Internal Medicine. Vol. 154, Pg. 1633, 1994. | |||||||||
| NFPA 704 |
|
Minocycline hydrochloride price More Price(7)
| Manufacturer | Product number | Product description | CAS number | Packaging | Price | Updated | Buy |
|---|---|---|---|---|---|---|---|
| Sigma-Aldrich(India) | M9511 | Minocycline hydrochloride powder | 13614-98-7 | 25MG | ₹4026.9 | 2022-06-14 | Buy |
| Sigma-Aldrich(India) | M9511 | Minocycline hydrochloride powder | 13614-98-7 | 100MG | ₹10500.25 | 2022-06-14 | Buy |
| Sigma-Aldrich(India) | M9511 | Minocycline hydrochloride powder | 13614-98-7 | 250MG | ₹18142.7 | 2022-06-14 | Buy |
| Sigma-Aldrich(India) | PHR1801 | Minocycline Hydrochloride Pharmaceutical Secondary Standard; Certified Reference Material | 13614-98-7 | 500MG | ₹24085.63 | 2022-06-14 | Buy |
| Sigma-Aldrich(India) | M9511 | Minocycline hydrochloride powder | 13614-98-7 | 1G | ₹53940.98 | 2022-06-14 | Buy |
Minocycline hydrochloride Chemical Properties,Uses,Production
Chemical Properties
Yellow Crystalline Powder
Uses
Minocycline hydrochloride is a salt prepared from minocycline, taking advantage of the two basic dimethylamino groups which protonate and readily form a salt from hydrochloric acid solutions. The hydrochloride is the preferred formulation for pharmaceutical applications. Like all tetracyclines, minocycline shows broad spectrum antibacterial and antiprotozoan activity and acts by binding to the 30S and 50S ribosomal sub-units, blocking protein synthesis.
General Description
Minocycline, 7-dimethylamino-6-demethyl-6-deoxytetracycline(Minocin, Vectrin), the most potent tetracycline currentlyused in therapy, is obtained by reductive methylationof 7-nitro-6-demethyl-6-deoxytetracycline. It was releasedfor use in the United States in 1971. Because minocycline,like doxycycline, lacks the 6-hydroxyl group, it is stablein acids and does not dehydrate or rearrange to anhydroor lactone forms. Minocycline is well absorbed orally togive high plasma and tissue levels. It has a very long serumhalf-life, resulting from slow urinary excretion and moderateprotein binding. Doxycycline and minocycline, alongwith oxytetracycline, show the least in vitro calcium bindingof the clinically available tetracyclines. The improved distributionproperties of the 6-deoxytetracyclines have been attributedto greater lipid solubility.
Perhaps the most outstanding property of minocyclineis its activity toward Gram-positive bacteria, especiallystaphylococci and streptococci. In fact, minocycline hasbeen effective against staphylococcal strains that are resistantto methicillin and all other tetracyclines, includingdoxycycline. Although it is doubtful that minocyclinewill replace bactericidal agents for the treatment of lifethreateningstaphylococcal infections, it may become auseful alternative for the treatment of less serious tissueinfections. Minocycline has been recommended for thetreatment of chronic bronchitis and other upper respiratorytract infections. Despite its relatively low renal clearance,partially compensated for by high serum and tissuelevels, it has been recommended for the treatment of urinary tract infections. It has been effective in the eradicationof N. meningitidis in asymptomatic carriers.
Side effects
Common side effects of Minocycline hydrochloride include: nausea, vomiting, diarrhoea, dizziness, lightheadedness or spinning sensation. Individuals may experience symptoms of serious adverse reactions such as gingival hyperpigmentation, pain/difficulty swallowing, tinnitus or hearing loss, joint stiffness/pain/swelling, nephrotoxicity (elevated urea nitrogen, interstitial nephritis), hepatotoxicity (hyperbilirubinaemia, hepatic cholestasis, elevated liver enzymes, fatal hepatic failure, and jaundice), and hypersensitivity reactions. It rarely causes elevated pressure around the brain (intracranial hypertension - IH). The risk of this side effect is greater in women of childbearing age who are overweight or who have had IH in the past. Serious intestinal disorders including: non-stop diarrhoea, abdominal or stomach pain/cramps, and blood/mucus in the stool are less common.
Minocycline hydrochloride Preparation Products And Raw materials
Raw materials
Preparation Products
| Supplier | Tel | Country | ProdList | Advantage | Inquiry |
|---|---|---|---|---|---|
| A.J Chemicals | 91-9810153283 | New Delhi, India | 6124 | 58 | Inquiry |
| CLEARSYNTH LABS LTD. | +91-22-45045900 | Hyderabad, India | 6351 | 58 | Inquiry |
| CHEMSWORTH | +91-261-2397244 | New Delhi, India | 6707 | 30 | Inquiry |
| TCI Chemicals (India) Pvt. Ltd. | 1800 425 7889 | New Delhi, India | 6778 | 58 | Inquiry |
| Pharmaffiliates Analytics and Synthetics P. Ltd | +91-172-5066494 | Haryana, India | 6773 | 58 | Inquiry |
| Euroasia Trans Continental | +91 22 56349035-36 | New Delhi, India | 519 | 47 | Inquiry |
| Clearsynth Labs | 91-22-45045900 | Maharashtra, India | 3889 | 58 | Inquiry |
| SynZeal Research Pvt Ltd | +1 226-802-2078 | Gujarat, India | 6522 | 58 | Inquiry |
| RA Chem Pharma Limited | 07093802565 | Hyderabad, India | 2 | 58 | Inquiry |
| Sign Pharma Trade Services Pvt., Ltd.(SPTS) | 91-22-23481235 | Mumbai, India | 160 | 58 | Inquiry |
13614-98-7(Minocycline hydrochloride)Related Search:
1of4
chevron_right




