Boric acid
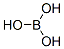
- CAS No.
- 11113-50-1
- Chemical Name:
- Boric acid
- Synonyms
- Boric acid flake;boric acid chunks;borid acid;Chemical Boric Acid Flake Powder;Boric acid;Borsrenatliche;Boric acid chunk;Boric acid Joyce;Boric acid USP/EP/BP;BoricAcid(AS),Borofax
- CBNumber:
- CB8874759
- Molecular Formula:
- BH3O3
- Molecular Weight:
- 61.83
- MOL File:
- 11113-50-1.mol
- Modify Date:
- 2024/7/24 13:33:21
| Melting point | >148oC (dec.) |
|---|---|
| storage temp. | Room Temperature |
| solubility | DMSO (Slightly), Water (Slightly) |
| form | Liquid |
| pka | 9.2(at 25℃) |
| color | Clear, dark blue |
| InChI | InChI=1S/BH3O3/c2-1(3)4/h2-4H |
| InChIKey | KGBXLFKZBHKPEV-UHFFFAOYSA-N |
| SMILES | B(O)(O)O |
| EPA Substance Registry System | Boric acid (11113-50-1) |
Boric acid Chemical Properties,Uses,Production
Chemical Properties
Boric acid is a weak acid (pKa = 9.15), existing in aqueous solutions at or below pH 7 as undissociated boric acid. In the solid there is considerable hydrogen bonding between H3BO3 molecules resulting in a layer structure, which accounts for the easy cleavage of the crystals. H3BO3 molecules also exist in dilute solutions but in more concentrated solutions polymeric acids and ions are formed (e.g. H4B2O7; pyroboric acid or tetrahydroxomonoxodiboric(III) acid). The compound is a very weak acid but also acts as a Lewis acid in accepting hydroxide ions:
B(OH)3 + H2O→B(OH)4 - + H+
If solid boric acid is heated it loses water and transforms to another acid at 300℃. This is given the formula HBO2 but is in fact a polymer (HBO2)n. It is called metaboric acid or, technically, polydioxoboric(III) acid.
Physical properties
Boric acid exists in the form of colorless crystals or as a white powder and is soluble in water. It occurs naturally in the condensate from volcanic steam vents (suffioni). Commercially, it is made by treating borate minerals (e.g. kernite, Na2B4O7.4H2O) with sulphuric acid followed by recrystallization.
Uses
Boric acid is used in the manufacture of glass (borosilicate glass), glazes and enamels, leather, paper, adhesives, and explosives. It is widely used (particularly in the USA) in detergents, and because of the ability of fused boric acid to dissolve other metal oxides it is used as a flux in brazing and welding. Because of its mild antiseptic properties it is used in the pharmaceutical industry and as a food preservative.
boric acid is an effective preservative against yeast. It is used in concentrations of 0.01 to 1.0 percent and has fair to good antiseptic properties. It may also be used as a buffer and denaturant. Boric acid is prepared from sulfuric acid and natural borax. It can cause skin rashes and irritation if used in high concentrations. The use of boric acid in cosmetic preparations is no longer very popular.
Indications
Boric Acid is a weakly acidic hydrate of boric oxide with mild antiseptic, antifungal, and antiviral properties. The exact mechanism of action of boric acid is unknown; generally cytotoxic to all cells. It is used in the treatment of yeast infections and cold sores.
Boric acid, 600 mg in a gelatin capsule, used intravaginally daily for 14 days, has been reported effective even in resistant Candida infections.
Definition
ChEBI: Boric acid is a member of boric acids. It has a role as an astringent. It is a conjugate acid of a dihydrogenborate.
Preparation
Boric acid is prepared by treating borax with nitric acid and nitric acid is chosen over hydrochloric acid because sodium nitrate is more readily soluble than sodium chloride, and sulphuric acid is hard to wash out so not employed.
In laboratory boric acid is prepared from sodium borate.
Na2B4O7 + H2SO4 + 5H2O→ Na2SO4 + 4H3BO3
Pharmaceutical Applications
Boric acid is a long-standing traditional remedy with mainly antifungal and antimicrobial effects. For medicinal uses, it has become known as sal sedativum, which was discovered by Homberg, the Dutch natural philosopher, in 1702. Diluted solutions were and sometimes still are used as antiseptics for the treatment of athletes’ foot and bacterial thrush, and in much diluted solutions as eyewash.
Agricultural Uses
Boric acid is used as an insecticide for controlling house holding pests like termites, ants, and small insects.
Boric acid (H3BO3) is one of the boron-containing nutrients added to fertilizers. It contains around 17% boron. A solution of boric acid and water is used as a foliar spray to overcome boron deficiency.
Potential Exposure
Boric acid is a fireproofing agent for wood; a preservative, and an antiseptic. It is used in the manufacture of glass, pottery, enamels, glazes, cosmetics, cements, porcelain, borates, leather, carpets, hats, soaps; artificial gems; in tanning leather; printing, dyeing, painting, and photography.
Shipping
UN 3077 Environmentally hazardous substances, solid, n.o.s., Hazard class: 9; Labels: 9—Miscellaneous hazardous material, Technical Name Required.
Incompatibilities
Boric acid decomposes in heat above 100 C, forming boric anhydride and water. Boric acid is hygroscopic; it will absorb moisture from the air. Boric acid aqueous solution is a weak acid; incompatible with strong reducing agents including alkali metals and metal hydrides (may generate explosive hydrogen gas); acetic anhydride, alkali carbonates, and hydroxides. Violent reaction with powdered potassium metal, especially if impacted. Attacks iron in the presence of moisture.
Waste Disposal
Boric acids may be recovered from organic process wastes as an alternative to disposal.
Boric acid Preparation Products And Raw materials
Raw materials
1of2
chevron_rightPreparation Products
1of3
chevron_right| Supplier | Tel | Country | ProdList | Advantage | Inquiry |
|---|---|---|---|---|---|
| LABDHI CHEMICAL INDUSTRIES | +91-9322229293 +91-9322229293 | Mumbai, India | 21 | 58 | Inquiry |
| Indiana ChemPort | +91-91-2652638667 +91-7574886711 | Gujarat, India | 85 | 58 | Inquiry |
| QUALIKEMS FINE CHEM PVT LTD | +911123618475 | New Delhi, India | 51 | 58 | Inquiry |
| PERFECT CHEMICAL | +91-9820465461 +91-9820465461 | Mumbai, India | 391 | 58 | Inquiry |
| VITRAG CHEMICALS PVT LTD | +91-7490053194 +91-7490053194 | Gujarat, India | 47 | 58 | Inquiry |
| JSK Chemicals | +919879767970 | Gujarat, India | 3756 | 58 | Inquiry |
| Organic Industries Pvt Ltd | +91-9890854479 +91-9890854479 | Mumbai, India | 4 | 58 | Inquiry |
| Kronox Lab Sciences Pvt Ltd | +91-9313231074 +91-9313231074 | Gujarat, India | 240 | 58 | Inquiry |
| Fluobor India Pvt Ltd | +91-8691012149 +91-8691012149 | Maharashtra, India | 7 | 58 | Inquiry |
| Sujata Chemicals | +91-9898025496 +91-9898025496 | Gujarat, India | 73 | 58 | Inquiry |
| Supplier | Advantage |
|---|---|
| LABDHI CHEMICAL INDUSTRIES | 58 |
| Indiana ChemPort | 58 |
| QUALIKEMS FINE CHEM PVT LTD | 58 |
| PERFECT CHEMICAL | 58 |
| VITRAG CHEMICALS PVT LTD | 58 |
| JSK Chemicals | 58 |
| Organic Industries Pvt Ltd | 58 |
| Kronox Lab Sciences Pvt Ltd | 58 |
| Fluobor India Pvt Ltd | 58 |
| Sujata Chemicals | 58 |
Related articles
- How long does it take for boric acid to dissolve?
- Understanding the dissolution process is crucial to ensure the suppositories are effective and provide relief. Boric acids tak....
- Mar 6,2024
- The uses of Boric acid
- Boric acid is a water-soluble white compound and occurs naturally. Its molecular formula is H3BO3.
- Mar 5,2024
- What Color Does Boric Acid Burn?
- A very pale green color is imparted to the flame by boron in boric acid. Because, Boric Acid is a boron containing compound An....
- Jan 25,2024
11113-50-1(Boric acid)Related Search:
1of4
chevron_right





