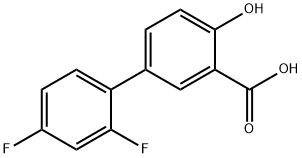
Diflunisal
- CAS No.
- 22494-42-4
- Chemical Name:
- Diflunisal
- Synonyms
- mk647;Adomal;MK 647;Dolisal;Dolobid;Dolobil;Dolobis;Flustar;Difludol;Flovacil
- CBNumber:
- CB9674965
- Molecular Formula:
- C13H8F2O3
- Molecular Weight:
- 250.2
- MOL File:
- 22494-42-4.mol
- MSDS File:
- SDS
- Modify Date:
- 2024/7/2 8:55:42
| Melting point | 207-209?C |
|---|---|
| Boiling point | 386.9±42.0 °C(Predicted) |
| Density | 1.3505 (estimate) |
| storage temp. | Keep in dark place,Sealed in dry,Room Temperature |
| solubility | Practically insoluble in water, soluble in ethanol (96 per cent). It dissolves in dilute solutions of alkali hydroxides. |
| form | Solid |
| pka | pKa 3.3 (H2O I=0.1) (Uncertain) |
| color | White |
| Water Solubility | 6.186mg/L(24.99 ºC) |
| CAS DataBase Reference | 22494-42-4(CAS DataBase Reference) |
| NIST Chemistry Reference | Diflunisal(22494-42-4) |
SAFETY
Risk and Safety Statements
| Symbol(GHS) |   GHS07,GHS08 |
|||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Signal word | Warning | |||||||||
| Hazard statements | H302-H315-H319-H335-H361 | |||||||||
| Precautionary statements | P201-P301+P312+P330-P302+P352-P305+P351+P338-P308+P313 | |||||||||
| Hazard Codes | Xn,Xi | |||||||||
| Risk Statements | 22-36/37/38-63 | |||||||||
| Safety Statements | 22-26-36 | |||||||||
| WGK Germany | 3 | |||||||||
| RTECS | DV2030000 | |||||||||
| Hazard Note | Irritant | |||||||||
| HS Code | 2918290000 | |||||||||
| Toxicity | LD50 orally in female mice: 439 mg/kg (Stone) | |||||||||
| NFPA 704 |
|
Diflunisal Chemical Properties,Uses,Production
Description
Diflunisal is more potent than aspirin but produces fewer side effects and has a biological half-life three to four times greater than that of aspirin. It is rapidly and completely absorbed on oral administration, with peak plasma levels being achieved within 2 to 3 hours of administration. It is highly bound (99%) to plasma proteins after absorption. Its elimination half-life is 8 to 12 hours, and it is excreted into urine primarily as glucuronide conjugates. The most frequently reported side effects include disturbances of the GI system (e.g., nausea, dyspepsia, and diarrhea), dermatological reactions, and CNS effects (e.g., dizziness and headache).
Chemical Properties
White Solid
Uses
As a prostaglandin synthetase inhibitor, diflunisal exhibits analgesic, fever-reducing, and anti-inflammatory action. It is used for long- and short-lasting symptomatic relief of low to moderate pain in osteoarthritis and rheumatoid arthritis.
brand name
Biartac (Merck Sharp & Dohme, Belgium), Diflonid (Dumex, Denmark, Sweden), Diflusal (Merck Sharp & Dohme, Belgium), Dolobid (Frosst, Canada; Logos, South Africa; Merck, USA; Morson, UK), Dolobis (M.S.D.-Chibret, France), Fluniget (Merck Sharp & Dohme, Austria).
General Description
Diflunisal (Dolobid), is a longer acting and more potent drugthan aspirin because of its hydrophobic, 2,4-difluorophenylgroup attached to the 5-position of the salicyclic acid. In alarge-scale comparative study with aspirin, it was also bettertolerated with less GI complications than aspirin. It ismarketed in tablet form for treating mild to moderate postoperativepain as well as RA and OA.
Diflunisal is highly protein bound. Its metabolism is subjectto a dose-dependent, saturable, and capacity-limitedglucuronide formation. This unusual pharmacokineticprofile is a result of an enterohepatic circulation and the reabsorptionof 65% of the drug and its glucuronides, followedby cleavage of its unstable, acyl glucuronide back tothe active drug. Thus, diflunisal usage in patients with renalimpairment should be closely monitored.
General Description
Diflunisal is a salicylate with greater anti-inflammatory and analgesic potency and with a long plasma half-life that allows for twice-a-day dosage. Without an O-acetyl group it is a reversible inhibitor of cyclooxygenase. It is comparable in efficacy to aspirin in the treatment of osteoarthritis and rheumatoid arthritis but is much better tolerated.
Pharmacokinetics
Diflunisal is more potent than aspirin but produces fewer side effects and has a biological half-life three to four times greater than that of aspirin. It is rapidly and completely absorbed on oral administration, with peak plasma levels being achieved within 2 to 3 hours of administration. It is highly bound (99%) to plasma proteins after absorption. Its elimination half-life is 8 to 12 hours, and it is excreted into urine primarily as glucuronide conjugates. The most frequently reported side effects include disturbances of the GI system (e.g., nausea, dyspepsia, and diarrhea), dermatological reactions, and CNS effects (e.g., dizziness and headache).
Clinical Use
Diflunisal (pKa 3.3) was introduced in the United States in 1982 and has gained considerable acceptance as an analgetic and as a treatment of rheumatoid arthritis and osteoarthritis. Diflunisal is metabolized primarily to ether and ester glucuronide conjugates.
Safety Profile
Poison by ingestion, subcutaneous, and intraperitoneal routes. Human systemic effects by ingestion: tolerance, and cholestatic jaundce (due to the stoppage of the flow of bile), agranulocytosis, increased body temperature. An experimental teratogen. Other experimental reproductive effects. An analgesic and anti-inflammatory agent. When heated to decomposition it emits toxic fumes of F-. See also FLUORIDES.
Diflunisal Preparation Products And Raw materials
Raw materials
Preparation Products
| Supplier | Tel | Country | ProdList | Advantage | Inquiry |
|---|---|---|---|---|---|
| Aarti Industries Limited (AIL) | +91-84596144931 +91-9920899935 | Maharashtra, India | 235 | 58 | Inquiry |
| Rivashaa Agrotech Biopharma Pvt. Ltd. | +91-26463395 +91-7926462688 | Gujarat, India | 1615 | 58 | Inquiry |
| Apicore Pharmaceuticals Pvt Ltd | +91-2662267177 +91-2662267166 | Gujarat, India | 181 | 58 | Inquiry |
| Cohance Lifesciences (Previously RA Chem Pharma Ltd) | +91-4044758595 +91-4044758595 | Telangana, India | 47 | 58 | Inquiry |
| TCI Chemicals (India) Pvt. Ltd. | 1800 425 7889 | New Delhi, India | 6778 | 58 | Inquiry |
| AARTI INDUSTRIES LTD | +91 22 6797 6666 | New Delhi, India | 32 | 58 | Inquiry |
| RA CHEM PHARMA LTD | +91-40-4475 8595 | New Delhi, India | 37 | 58 | Inquiry |
| MYLAN API US LLC | 080 6672 8000 | New Delhi, India | 44 | 58 | Inquiry |
| SynZeal Research Pvt Ltd | +1 226-802-2078 | Gujarat, India | 6522 | 58 | Inquiry |
| Pharmaffiliates Analytics and Synthetics P. Ltd | +91-172-5066494 | Haryana, India | 6773 | 58 | Inquiry |
22494-42-4(Diflunisal)Related Search:
1of4
chevron_right




