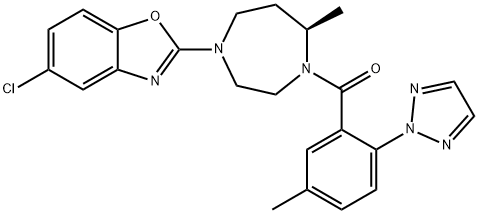
Suvorexant
|
- ₹0
- Product name: Suvorexant
- CAS: 1030377-33-3
- MF: C23H23ClN6O2
- MW: 450.92
- EINECS:685-109-7
- MDL Number:MFCD22377755
- Synonyms:5-Chloro-2-[(5R)-5-methyl-4-[5-methyl-2-(2H-1,2,3-triazol-2-yl)benzoyl]-1,4-diazepan-1-yl]-1,3-benzoxazole;MK 4305;Suvorexant;MK-4305 (Suvorexant);Methanone, [(7R)-4-(5-chloro-2-benzoxazolyl)hexahydro-7-Methyl-1H-1,4-diazepin-1-yl][5-Methyl-2-(2H-1,2,3-triazol-2-yl)phenyl]-;Suvorexant (MK-4305);Suvorexant (with 4 ints.);SUVOREXABT
| Manufacturer | Product number | Product description | Packaging | Price | Updated | Buy |
|---|
Properties
Melting point :153℃
Boiling point :669.8±65.0 °C(Predicted)
Density :1.41
storage temp. :Room Temperature
solubility :Acetonitrile (Slightly), Chloroform (Slightly), DMSO (Slightly, Heated)
form :Solid
pka :1.47±0.40(Predicted)
color :White to Pale Beige
InChIKey :JYTNQNCOQXFQPK-MRXNPFEDSA-N
SMILES :C(N1[C@H](C)CCN(C2=NC3=CC(Cl)=CC=C3O2)CC1)(C1=CC(C)=CC=C1N1N=CC=N1)=O
Boiling point :669.8±65.0 °C(Predicted)
Density :1.41
storage temp. :Room Temperature
solubility :Acetonitrile (Slightly), Chloroform (Slightly), DMSO (Slightly, Heated)
form :Solid
pka :1.47±0.40(Predicted)
color :White to Pale Beige
InChIKey :JYTNQNCOQXFQPK-MRXNPFEDSA-N
SMILES :C(N1[C@H](C)CCN(C2=NC3=CC(Cl)=CC=C3O2)CC1)(C1=CC(C)=CC=C1N1N=CC=N1)=O
Safety Information
| Symbol(GHS): |
  
|
|||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Signal word: | Warning | |||||||||||||||||||||||||||||||||||
| Hazard statements: |
|
|||||||||||||||||||||||||||||||||||
| Precautionary statements: |
|
Description
Suvorexant (Belsomra® ) is a novel sedative hypnotic drug that is prescribed to promote sleep in patients with insomnia. It is the first of a new class of drugs classified as dual orexin receptor antagonists (DORAs) and is currently approved for the treatment of insomnia in the United States and Japan. It is a CNS depressant and blocks the binding of wake-promoting neuropeptides orexin A and orexin B to the two orexin receptors (OX1R and OX2R) thus, altering the signaling (action) of orexin in the brain and suppressing the sleep-wake drive.Suvorexant is a central nervous system (CNS) depressant and can impair daytime wakefulness even when used as prescribed. Prescribers should monitor for somnolence and CNS depressant effects, but impairment can occur in the absence of symptoms, and may not be reliably detected by ordinary clinical exam. CNS depressant effects may persist in some patients for up to several days after discontinuing Suvorexant.
Belsomra (Suvorexant) can impair driving skills and may increase the risk of falling asleep while driving. Discontinue or decrease the dose in patients who drive if daytime somnolence develops. Caution patients taking Belsomra 20 mg against next-day driving and other activities requiring full mental alertness. Caution patients taking lower doses of Belsomra as well, because there is individual variation in sensitivity to Belsomra.
More related product prices
Olaparib Crizotinib Ibrutinib Bosutinib Nintedanib Ramelteon AGOMELATINE Obeticholic Acid ILOPERIDONE Olopatadine Tafluprost Sunitinib Malate SUN11602Related product price
- Crizotinib
₹13174.03 - Bosutinib
₹10800-12730.2 - Ramelteon
₹5100-53854.38
Suppliers and manufacturers
CLEARSYNTH LABS LTD.
Pharmaffiliates Analytics and Synthetics P. Ltd
SynZeal Research Pvt Ltd
Pharma Affiliates
Seasons Biotechnology Co., Ltd.
Wuhan Topule Biopharmaceutical Co., Ltd
Shanghai Daken Advanced Materials Co.,Ltd
Shanghai Time Chemicals CO., Ltd.
Hangzhou FandaChem Co.,Ltd.






