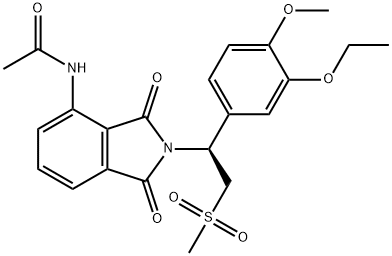
Apremilast
- CAS No.
- 608141-41-9
- Chemical Name:
- Apremilast
- Synonyms
- Apst;Apster;Apremilas;N-{2-[(1S)-1-(3-Ethoxy-4-methoxyphenyl)-2-(methylsulfonyl)ethyl]-1,3-dioxo-2,3-dihydro-1H-isoindol-4-yl}acetamide;CS-1055;Apr-im D;Apmisast;ApreMilast;Apremildst;The staples
- CBNumber:
- CB12563430
- Molecular Formula:
- C22H24N2O7S
- Molecular Weight:
- 460.5
- MOL File:
- 608141-41-9.mol
- MSDS File:
- SDS
- Modify Date:
- 2024/6/18 13:26:42
| Melting point | 152-156°C |
|---|---|
| Boiling point | 741.3±60.0 °C(Predicted) |
| Density | 1.381 |
| storage temp. | Refrigerator |
| solubility | Chloroform (Slightly), DMSO (Slightly), Methanol (Slightly, Heated) |
| pka | 14.01±0.20(Predicted) |
| form | Solid |
| color | White to Pale Yellow |
| InChIKey | IMOZEMNVLZVGJZ-QGZVFWFLSA-N |
| SMILES | C(NC1=CC=CC2=C1C(=O)N([C@@H](C1=CC=C(OC)C(OCC)=C1)CS(C)(=O)=O)C2=O)(=O)C |
| CAS DataBase Reference | 608141-41-9 |
SAFETY
Risk and Safety Statements
| Symbol(GHS) |   GHS08,GHS07 |
|||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Signal word | Warning | |||||||||
| Hazard statements | H302-H361 | |||||||||
| Precautionary statements | P201-P202-P281-P308+P313-P405-P501-P264-P270-P301+P312-P330-P501 | |||||||||
| NFPA 704 |
|
Apremilast Chemical Properties,Uses,Production
Description
Apremilast is the first and only oral phosphodiesterase IV (PDE- 4) inhibitor and anti-tumor necrosis factor alpha (TNFa) agent launched in the USA by Celgene for the treatment of active psoriatic arthritis (PsA). Apremilast was also approved for the treatment of moderate to severe plaque psoriasis in the USA. Later, this drug was also approved in the European Union (EU) for both indications. Although multiple approaches to the synthesis of apremilast have been described, the most likely process scale approach involves the construction of the challenging stereogenic benzylic carbon center by catalytic asymmetric hydrogenation.
Uses
Apremilast is an oral phosphodiesterase 4 inhibitor used in the treatment of adults with active psoriatic arthritis and adults with moderate to severe plaque psoriasis.
Definition
ChEBI: Apremilast is a member of the class of isoindoles that is isoindole-1,3-dione substituted at position 4 by an acetamido group and at position 1 by a 1-(3-ethoxy-4-methoxyphenyl)-2-(methylsulfonyl)ethyl group. Used for treatment of psoriatic arthritis. It has a role as a phosphodiesterase IV inhibitor and a non-steroidal anti-inflammatory drug. It is an aromatic ether, a N-acetylarylamine, a sulfone and a member of phthalimides.
Overdosage
Apremilast was studied in healthy subjects at a maximum total daily dose of 100 mg (given as 50 mg twice daily) for 4.5 days without evidence of dose limiting toxicities. In case of an overdose, it is recommended that the patient is monitored for any signs or symptoms of adverse effects and appropriate symptomatic treatment is instituted. In the event of overdose, symptomatic and supportive care is advised.
Enzyme inhibitor
This thalidomide-like psoriasis drug (FW = 460.50 g/mol; CAS 608141-41-9; Solubility: 90 mg/mL DMSO; <1 mg/mL H2O), also known as CC-10004, Otezla? and N-[2-[(1S)-1-(3-ethoxy-4-methoxyphenyl)-2-(methyl-sulfonyl)ethyl]-2,3-dihydro-1,3-dioxo-1H-isoindol-4-yl]acetamide, is a potent, orally active phosphodiesterase inhibitor that targets phosphodiesterase PDE4 (IC50 = 74 nM), itself a proinflammatory mediator, and TNF-α, IC50 = 77 nM. Apremilast inhibits PBMC production of the chemokines CXCL9 and CXCL10, cytokines interferon-γ and tumor necrosis factor-α (TNF-α), and interleukins IL-2, IL-12 and IL- 23 from human rheumatoid synovial membrane cultures. Apremilast significantly reduces clinical scores in both murine models of arthritis over a ten-day treatment period and maintains healthy joint architecture in a dose-dependent manner. Unlike rolipram, however, apremilast demonstrated no adverse behavioral effects in na?ve mice. Otezla is specifically indicated by the Food & Drug Administration for the treatment of patients with moderate to severe plaque psoriasis who are typically candidates for phototherapy or systemic therapy. That said, the specific mechanism(s) for therapeutic action in psoriatic arthritis patients and psoriasis patients is unclear.
Mode of action
Apremilast, an oral small-molecule inhibitor of phosphodiesterase 4 (PDE4), works intracellularly to modulate a network of pro-inflammatory and anti-inflammatory mediators. PDE4 is a cyclic adenosine monophosphate (cAMP)-specific PDE and the dominant PDE in inflammatory cells. PDE4 inhibition elevates intracellular cAMP levels, which in turn down-regulates the inflammatory response by modulating the expression of TNF-α, IL-23, IL-17 and other inflammatory cytokines. Cyclic AMP also modulates levels of anti-inflammatory cytokines such as IL-10. These pro- and anti-inflammatory mediators have been implicated in psoriatic arthritis and psoriasis.
Apremilast Preparation Products And Raw materials
Raw materials
Preparation Products
| Supplier | Tel | Country | ProdList | Advantage | Inquiry |
|---|---|---|---|---|---|
| Jigs Chemical ltd | +919099003427 | Gujarat, India | 239 | 58 | Inquiry |
| Saptagir Laboratories PVT.LTD. | +91-9059978049 +91-9059978049 | Telangana, India | 31 | 58 | Inquiry |
| Anant Pharmaceuticals Pvt Ltd | +91-8550986868 +91-9485998001 | Haryana, India | 461 | 58 | Inquiry |
| Bulat Pharmaceutical Pvt Ltd | +91-8448085659 +91-8448085660 | Haryana, India | 123 | 58 | Inquiry |
| GLP Pharma Standards | +91 9866074638 | Hyderabad, India | 1644 | 58 | Inquiry |
| Lewens Labs | +917043057100 | Gujarat, India | 30 | 58 | Inquiry |
| KARPSCHEM LABORATORIES | +91-7249203006 +91-7249203006 | Maharashtra, India | 786 | 58 | Inquiry |
| Varanous Labs Pvt Ltd | +91-7036248882 +91-7036248882 | Hyderabad, India | 1541 | 58 | Inquiry |
| Aurobindo Pharma Limited | +914066725000 | Telangana, India | 112 | 58 | Inquiry |
| Lupin Ltd | +91-8019896181 +91-8019896181 | Maharashtra, India | 93 | 58 | Inquiry |
| Supplier | Advantage |
|---|---|
| Jigs Chemical ltd | 58 |
| Saptagir Laboratories PVT.LTD. | 58 |
| Anant Pharmaceuticals Pvt Ltd | 58 |
| Bulat Pharmaceutical Pvt Ltd | 58 |
| GLP Pharma Standards | 58 |
| Lewens Labs | 58 |
| KARPSCHEM LABORATORIES | 58 |
| Varanous Labs Pvt Ltd | 58 |
| Aurobindo Pharma Limited | 58 |
| Lupin Ltd | 58 |
Related articles
- Apremilast Uses and Relevance to the Military
- Apremilast effectively treats psoriasis and psoriatic arthritis, offering advantages for military personnel in austere conditi....
- Jan 29,2024
- Apremilast: A Review in Psoriasis and Psoriatic Arthritis
- Apremilast (Otezla) is an orally administered, small molecule inhibitor of phosphodiesterase 4 (PDE4). Apremilast 30 mg twice ....
- Feb 24,2023
608141-41-9(Apremilast)Related Search:
1of4
chevron_right




