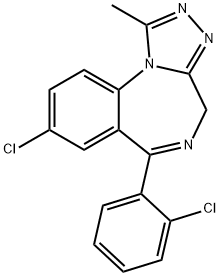
Triazolam
- CAS No.
- 28911-01-5
- Chemical Name:
- Triazolam
- Synonyms
- Songar;Halcyon;Asasion;Halcion;U-33,030;novidorm;Novodorm;D II-18-2;TRIAZOLAM;clorazolam
- CBNumber:
- CB2218405
- Molecular Formula:
- C17H12Cl2N4
- Molecular Weight:
- 343.21
- MOL File:
- 28911-01-5.mol
- Modify Date:
- 2023/5/4 17:34:37
| Melting point | 209-212°C |
|---|---|
| Boiling point | 499.51°C (rough estimate) |
| Density | 1.2835 (rough estimate) |
| refractive index | 1.6300 (estimate) |
| Flash point | 11 °C |
| storage temp. | 2-8°C |
| solubility | DMF: 30 mg/ml; DMF:PBS(pH 7.2)(1:1): 0.5 mg/ml; DMSO: 20 mg/ml; Ethanol: 10 mg/ml |
| form | A crystalline solid |
| pka | pKa 1.52(H2O) (Uncertain);6.5(H2O) (Uncertain) |
| Water Solubility | 30mg/L(ambient temperature) |
| CAS DataBase Reference | 28911-01-5(CAS DataBase Reference) |
| NIST Chemistry Reference | Triazolam(28911-01-5) |
| EPA Substance Registry System | Triazolam (28911-01-5) |
SAFETY
Risk and Safety Statements
| Symbol(GHS) |    GHS02,GHS06,GHS08 |
|---|---|
| Signal word | Danger |
| Hazard statements | H225-H301+H311+H331-H370 |
| Precautionary statements | P210-P260-P280-P301+P310-P311 |
| Hazard Codes | F,T |
| Risk Statements | 11-23/24/25-36/38-39/23/24/25 |
| Safety Statements | 22-24/25-26-36-45-36/37-16-7 |
| RIDADR | 3249 |
| WGK Germany | 2 |
| RTECS | XZ5472500 |
| HazardClass | 6.1(b) |
| PackingGroup | III |
| HS Code | 2933910000 |
| Toxicity | LD50 in mice, rats (mg/kg): >100, >5000 orally (Pharm. Weekblad.) |
Triazolam Chemical Properties,Uses,Production
Chemical Properties
Yellow Solid
Uses
Triazolam
World Health Organization (WHO)
Triazolam, a benzodiazepine derivative with sedative and hypnotic activity, was introduced in 1978 for themanagement of insomnia. It is controlled under Schedule IV of the 1971 Convention of Psychotropic Substances. Concern regarding the psychotropic effects of triazolam was first raised in the Netherlands in 1979 when this compound was suspended for sale and subsequently withdrawn by the Committee for the Evaluation of Medicines on the basis of reports of a reversible complex of symptoms including paranoia, depersonalization, nightmares, suicidal tendency and hyperaesthesia in patients receiving the drug. The basis for this decision was later successfully contested by the manufacturer and the drug was reregistered in early 1990 with a revised product information. However, concern was regenerated elsewhere that higher doses are associated with an unacceptable incidence of unwanted effects and the manufacturer has eventually withdrawn 0.5 mg tablets on a worldwide basis. In 1991 the issue of the safety of triazolam was again reopened by reports of retrograde amnesia and depression among patients taking the decreased recommended dosages. The product information has been revised by the United States FDA to include more rigorous cautions regarding dosage. In the Member States of the European Communities the products have been suspended pending further review by the EC Committee on Proprietary Medicinal Products.
General Description
Triazolam, 8-chloro-6-(o-chlorophenyl)-1-methyl-4H-s-triazolo[4,3-a][1,4] benzodiazepine(Halcion), has all of the characteristic benzodiazepine pharmacologicalactions. It is an ultra–short-acting hypnoticbecause it is rapidly α-hydroxylated to the 1-methyl alcohol,which is then rapidly conjugated and excreted.Consequently, it has gained popularity as sleep inducers, especiallyin elderly patients, because it causes less daytimesedation. It is metabolically inactivated primarily by hepaticand intestinal CYP3A4; therefore, coadministration withgrapefruit juice increases its peak plasma concentration by30%, leading to increased drowsiness.
Triazolam Preparation Products And Raw materials
28911-01-5(Triazolam)Related Search:
1of4
chevron_right




