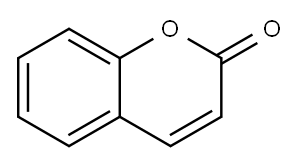
Coumarin
- CAS No.
- 91-64-5
- Chemical Name:
- Coumarin
- Synonyms
- 2H-Chromen-2-one;COUMARINE;2H-1-BENZOPYRAN-2-ONE;CUMARIN;CHROMEN-2-ONE;1,2-BENZOPYRONE;Kumarin;coumarin powder;1-BENZOPYRAN-2-ONE;Rattex
- CBNumber:
- CB3112168
- Molecular Formula:
- C9H6O2
- Molecular Weight:
- 146.14
- MOL File:
- 91-64-5.mol
- MSDS File:
- SDS
- Modify Date:
- 2024/6/19 22:55:17
| Melting point | 68-73 °C (lit.) |
|---|---|
| Boiling point | 298 °C (lit.) |
| Density | 0.935 |
| vapor pressure | 0.01 mm Hg ( 47 °C) |
| refractive index | 1.5100 (estimate) |
| Flash point | 162 °C |
| storage temp. | Store below +30°C. |
| solubility | 1.7g/l |
| form | Crystals or Crystalline Powder |
| color | White |
| PH Range | Non' uorescence (9.5) to light green ' uorescence (10.5) |
| Odor | at 10.00 % in dipropylene glycol. sweet hay tonka new mown hay |
| Odor Type | tonka |
| Water Solubility | 1.7 g/L (20 ºC) |
| λmax | 275nm |
| Merck | 14,2562 |
| BRN | 383644 |
| Major Application | color filter, organic electroluminescent devices, liquid crystal displays, field emission displays, inks, nickel plating, detergents, deodorant for shoes, petroleum products, cigarettes, personal care products, cosmetics, sunscreen cream, perfumes, nucleic acid sequencing, antiinflammatory agent, treatment of cancer, neurotransmission disorders, bleeding disorders, cerebrovascular disease, thrombosis, hemorrhoids, rheumatic disease, arthritic disease, epilepsy, vaginitis, painkiller, teeth whitening agent, skin whitening agent, wound healing promoter |
| InChIKey | ZYGHJZDHTFUPRJ-UHFFFAOYSA-N |
| LogP | 1.39 at 25℃ |
| CAS DataBase Reference | 91-64-5(CAS DataBase Reference) |
| IARC | 3 (Vol. Sup 7, 77) 2000 |
| NIST Chemistry Reference | Coumarin(91-64-5) |
| EPA Substance Registry System | Coumarin (91-64-5) |
SAFETY
Risk and Safety Statements
| Symbol(GHS) |  GHS06 |
|||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Signal word | Danger | |||||||||
| Hazard statements | H301+H311-H317 | |||||||||
| Precautionary statements | P261-P264-P270-P280-P301+P310-P302+P352+P312 | |||||||||
| Hazard Codes | Xn | |||||||||
| Risk Statements | 22-40-36/37/38-20/21/22-43 | |||||||||
| Safety Statements | 36-36/37-26 | |||||||||
| RIDADR | UN 2811 6.1/PG 3 | |||||||||
| WGK Germany | 1 | |||||||||
| RTECS | GN4200000 | |||||||||
| TSCA | Yes | |||||||||
| HazardClass | 6.1 | |||||||||
| PackingGroup | III | |||||||||
| HS Code | 29322010 | |||||||||
| Toxicity | LD50 orally in rats, guinea pigs: 680, 202 mg/kg (Jenner) | |||||||||
| NFPA 704 |
|
Coumarin price More Price(24)
| Manufacturer | Product number | Product description | CAS number | Packaging | Price | Updated | Buy |
|---|---|---|---|---|---|---|---|
| Sigma-Aldrich(India) | W526509 | Coumarin ≥98% | 91-64-5 | 1SAMPLE | ₹5141.88 | 2022-06-14 | Buy |
| Sigma-Aldrich(India) | W526509 | Coumarin ≥98% | 91-64-5 | 1KG | ₹15414.8 | 2022-06-14 | Buy |
| Sigma-Aldrich(India) | W526509 | Coumarin ≥98% | 91-64-5 | 5KG | ₹33395.13 | 2022-06-14 | Buy |
| Sigma-Aldrich(India) | W526509 | Coumarin ≥98% | 91-64-5 | 10KG | ₹38764.33 | 2022-06-14 | Buy |
| Sigma-Aldrich(India) | C4261 | Coumarin ≥99% (HPLC) | 91-64-5 | 50G | ₹3031 | 2022-06-14 | Buy |
Coumarin Chemical Properties,Uses,Production
Description
Coumarin is a naturally occurring Benzopyrone compound. It is found in a large number of plants belonging to many different families including tonka beans, woodruff, lavender oil, cassia, melilot (sweet clover), and other plants. It is found in edible plants such as strawberries, cinnamon, peppermint, green tea, carrots, and celery, as well as in partially fermented tea, red wine, beer, and other foodstuffs. Concentrations range from 87 000 ppm in cassia and 40 000 ppm in cinnamon to 20 ppm in peppermint and 5 ppb in tangerines.
Chemical Properties
Coumarin occurs widely in nature and determines, for example, the odor of woodruff. It forms white crystals (mp 70.6°C) with a hay-like, spicy odor. When treated with dilute alkali, coumarin is hydrolyzed to the corresponding coumarinic acid salt [(Z)-2-hydroxycinnamic acid]. Heating with concentrated alkali or with sodium ethanolate in ethanol results in the formation of o-coumaric acid salts [(E)-2-hydroxycinnamic acid]. 3,4-Dihydrocoumarin is obtained by catalytic hydrogenation, for example, with Raney nickel as a catalyst; octahydrocoumarin is obtained if hydrogenation is carried out at high temperature (200–250°C).
Occurrence
Found in many plants and essential oils such as cassia, melilot, orchid, lavender and balsam of Peru (Sp?th, 1937; Gildemeister & Hoffman, 1966).
Uses
coumarin is considered a blood thinner, it can also increase blood flow. Some sources cite anti-oxidant capacities, as well. It is a specific plant constituent and is what creates the fragrance of freshly mowed hay. Coumarin is found in such plants as cherries, lavender, licorice, and sweet clover.
Preparation
Coumarin is currently produced by Perkin synthesis from salicylaldehyde.
In the presence of sodium acetate, salicylaldehyde reacts with acetic
anhydride to produce coumarin and acetic acid. The reaction is carried out in the
liquid phase at elevated temperature.
A process for the production of coumarin from hexahydrocoumarin by dehydrogenation has also been elaborated.
Since the odor of coumarin is relatively weak, strong-smelling by-products (e.g.,
vinylphenol) must be removed. Many purification methods have been reported
and patented.
Definition
A colorless crystalline compound with a pleasant odor, used in making perfumes. On hydrolysis with sodium hydroxide it forms coumarinic acid.
General Description
Colorless crystals, flakes or colorless to white powder with a pleasant fragrant vanilla odor and a bitter aromatic burning taste.
Air & Water Reactions
Insoluble in water.
Reactivity Profile
Coumarin is sensitive to exposure to light. Coumarin is also sensitive to heat. Coumarin is incompatible with strong acids, strong bases and oxidizers. Coumarin is hydrolyzed by hot concentrated alkalis. Coumarin can be halogenated, nitrated and hydrogenated (in the presence of catalysts).
Hazard
Toxic by ingestion; carcinogenic. Use in food products prohibited (FDA). Questionable carcinogen.
Health Hazard
SYMPTOMS: Exposure to Coumarin may cause narcosis. It may also cause irritation and liver damage.
Fire Hazard
Coumarin is combustible.
Biological Activity
Oral anticoagulants can be prepared from compounds with coumarin as a base. Coumarin has been known for well over a century and, in addition to its use pharmaceutically, it is also an excellent odor-enhancing agent. However, because of its toxicity, it is not permitted in food products in the United States (Food and Drug Administration). One commercial drug is 3-(alpha-acetonyl-4-nitrobenzyl)- 4-hydroxycoumarin. This drug reduces the concentration of prothrombin in the blood and increases the prothrombin time by inhibiting the formation of prothrombin in the liver. The drug also interferes with the production of factors VII, IX, and X, so that their concentration in the blood is lowered during therapy. The inhibition of prothrombin involves interference with the action of vitamin K, and it has been postulated that the drug competes with vitamin K for an enzyme essential for prothrombin synthesis. Another commercial drug is bis-hydroxy-coumarin, C19H12O6. The actions of this drug are similar to those just described.
Contact allergens
Coumarin is an aromatic lactone naturally occurring in Tonka beans and other plants. As a fragrance allergen, it has to be mentioned by name in cosmetics within the EU
Safety Profile
Poison by ingestion, intraperitoneal, and subcutaneous routes. Questionable carcinogen with experimental tumorigenic data. Experimental teratogenic effects. Mutation data reported. Combustible when exposed to heat or flame. When heated to decomposition it emits acrid smoke and fumes. See also KETONES and ANHYDRIDES.
Environmental Fate
Coumarin toxicity is a function of blood and target tissue levels of coumarin relative to the metabolic capacity of the target organ. Cellular toxicity results when the formation of the toxic moieties exceeds the capacity of the cell to detoxify. This can have significant impact when comparing dosing by gavage to dietary exposure.
Purification Methods
Coumarin crystallises from ethanol or water and sublimes in vacuo at 43o [Srinivasan & deLevie J Phys Chem 91 2904 1987]. [Beilstein 17/10 V 143.]
Coumarin Preparation Products And Raw materials
Raw materials
1of2
chevron_rightPreparation Products
1of3
chevron_right| Supplier | Tel | Country | ProdList | Advantage | Inquiry |
|---|---|---|---|---|---|
| GLR Innovations | +91 9891111994 | New Delhi, India | 4542 | 58 | Inquiry |
| JSK Chemicals | +919879767970 | Gujarat, India | 3756 | 58 | Inquiry |
| DeliCare LifeSciences Pvt Ltd | +91-7977363866 +91-7977363866 | Maharashtra, India | 25 | 58 | Inquiry |
| Neeru Menthol Pvt Ltd | +91-8884114465 +91-9837322285 | New Delhi, India | 70 | 58 | Inquiry |
| HRV Global Life Sciences | +91-9820219686 +91-9820219686 | Telangana, India | 379 | 58 | Inquiry |
| Indenta Chemicals (India) Pvt Ltd | +91-2226849600 +91-9326627752 | Maharashtra, India | 48 | 58 | Inquiry |
| Eternis Fine Chemicals Ltd | +91-2266513400 +91-2266513400 | Maharashtra, India | 9 | 58 | Inquiry |
| Aamirav Ingredients And Specialties Pvt Ltd | +91-2224980102 +91-9820077171 | Maharashtra, India | 138 | 58 | Inquiry |
| Eiko LifeSciences Ltd | +91-02225438095 +91-02225438095 | Mumbai, India | 26 | 58 | Inquiry |
| S D Fine Chem Limited | +91-9323715856 +91-9323715856 | Maharashtra, India | 368 | 58 | Inquiry |
Related articles
- Uses of Coumarin
- Coumarin is a naturally occurring Benzopyrone compound. It is found in a large number of plants belonging to many different fa....
- Jan 12,2022
- Coumarin Metabolism, Toxicity and Carcinogenicity
- Coumarin is a natural product which exhibits marked species di?erences in both metabolism and toxicity. The majority of tests ....
- Nov 18,2019
- Coumarins — An Important Class of Phytochemicals
- Among them, coumarins are a family of benzopyrones (1,2-benzopyrones or 2H-1-benzopyran-2-ones) widely distributed in the natu....
- Nov 18,2019
91-64-5(Coumarin)Related Search:
1of4
chevron_right




