Chromium(VI) oxide
- CAS No.
- 1333-82-0
- Chemical Name:
- Chromium(VI) oxide
- Synonyms
- CrO3;CHROMIUM TRIOXIDE;CHROMIC ANHYDRIDE;trioxochromium;Chromium(Ⅵ) oxide;chromic acid solution;Chromic;Chromic acid (H2CrO4);CHROMIC ACID ANHYDRIDE;NA 1463
- CBNumber:
- CB5304358
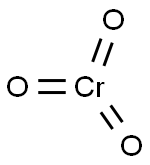
- Molecular Formula:
- CrO3
- Molecular Weight:
- 99.99
- MOL File:
- 1333-82-0.mol
- MSDS File:
- SDS
- Modify Date:
- 2024/3/14 15:18:26
| Melting point | 196 °C (dec.)(lit.) |
|---|---|
| Boiling point | 330 °C |
| Density | 2.7 |
| vapor pressure | 0Pa at 25℃ |
| Flash point | 250°C |
| storage temp. | Store below +30°C. |
| solubility | 1.667g/l |
| form | macroporous |
| pka | -0.61[at 20 ℃] |
| Specific Gravity | 2.7 |
| color | Reddish-violet |
| PH | <1 (100g/l, H2O, 20℃) |
| Water Solubility | Highly soluble |
| FreezingPoint | 170~172℃ |
| Sensitive | Hygroscopic |
| Merck | 14,2235 |
| Exposure limits |
ACGIH: TWA 0.0002 mg/m3; STEL 0.0005 mg/m3 (Skin) OSHA: Ceiling 0.1 mg/m3 NIOSH: IDLH 15 mg/m3; TWA 0.0002 mg/m3 |
| Stability | Stable. Strong oxidizer. Reacts with most organic material in a violent and often explosive fashion. Moisture sensitive. |
| InChIKey | WGLPBDUCMAPZCE-UHFFFAOYSA-N |
| CAS DataBase Reference | 1333-82-0(CAS DataBase Reference) |
| NIST Chemistry Reference | Chromium trioxide(1333-82-0) |
| EPA Substance Registry System | Chromium(VI) trioxide (1333-82-0) |
SAFETY
Risk and Safety Statements
| Symbol(GHS) |      GHS03,GHS05,GHS06,GHS08,GHS09 |
|||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Signal word | Danger | |||||||||
| Hazard statements | H271-H301+H311-H314-H317-H330-H334-H335-H340-H350-H361f-H372-H410 | |||||||||
| Precautionary statements | P210-P260-P280-P303+P361+P353-P304+P340+P310-P305+P351+P338 | |||||||||
| Hazard Codes | O,T+,N,T | |||||||||
| Risk Statements | 45-46-9-24/25-26-35-42/43-48/23-50/53-62-8 | |||||||||
| Safety Statements | 53-45-60-61-36/37/39-28-26-22 | |||||||||
| RIDADR | UN 1463 5.1/PG 2 | |||||||||
| OEB | E | |||||||||
| OEL | TWA: 0.0002 mg/m3 (8-hours) | |||||||||
| WGK Germany | 3 | |||||||||
| RTECS | GB6650000 | |||||||||
| TSCA | Yes | |||||||||
| HazardClass | 5.1 | |||||||||
| PackingGroup | II | |||||||||
| HS Code | 28191000 | |||||||||
| Toxicity | LD50 oral (rat) 80 mg/kg PEL (OSHA) 0.1 mg (CrO3)/m3 (ceiling) TLV-TWA (ACGIH) 0.05 mg (Cr)/m3 |
|||||||||
| IDLA | 15 mg Cr(VI)/m3 | |||||||||
| NFPA 704 |
|
Chromium(VI) oxide price More Price(26)
| Manufacturer | Product number | Product description | CAS number | Packaging | Price | Updated | Buy |
|---|---|---|---|---|---|---|---|
| Sigma-Aldrich(India) | 8.22266 | Chromium(VI) oxide for synthesis | 1333-82-0 | 100G | ₹2420.01 | 2022-06-14 | Buy |
| Sigma-Aldrich(India) | 8.22266 | Chromium(VI) oxide for synthesis | 1333-82-0 | 1KG | ₹7680 | 2022-06-14 | Buy |
| Sigma-Aldrich(India) | 8.22266 | Chromium(VI) oxide for synthesis | 1333-82-0 | 2.5KG | ₹21909.99 | 2022-06-14 | Buy |
| Sigma-Aldrich(India) | 675644 | Chromium(VI) oxide 99.99% trace metals basis | 1333-82-0 | 5G | ₹1688.7 | 2022-06-14 | Buy |
| Sigma-Aldrich(India) | 675644 | Chromium(VI) oxide 99.99% trace metals basis | 1333-82-0 | 25G | ₹5542.4 | 2022-06-14 | Buy |
Chromium(VI) oxide Chemical Properties,Uses,Production
Description
Chromium hydroxide (Cr2O(OH)4) is a bright bluish-green pigment prepared by the calcinations of bichromate with boric acid at 500°C. The mass during cooling is hydrolyzed with water, which yields the hydrate.
Chemical Properties
Chromium trioxide is a dark-red crystalline substance. It is odorless
Physical properties
Dark-red crystals, flakes or granular powder; bipyramidal prismatic system; density 2.70 g/cm3; melts at 197°C; decomposes on further heating; highly soluble in water, 61.7 g and 67 g/100 mL at 0°C and 100°C, respectively; soluble in sulfuric and nitric acids.
Uses
Chromium(VI) oxide is widely used in chromium electroplating, and as protective coatings in corrosion/oxidation resistance of metals. It is also employed in CDs and DVDs. It is useful as an oxidant in Jones oxidation in acetic acid or acetone. It is used in the oxidation of primary alcohols into carboxylic acids, and secondary alcohols into ketones. With phosphoric acid, it is used as a stripping agent for anodic coatings of all types and in the production of syntheric rubies.
Definition
chromium trioxide: A redcompound, CrO3; rhombic; r.d. 2.70;m.p. 196°C. It can be made by carefuladdition of concentrated sulphuricacid to an ice-cooled concentratedaqueous solution of sodium dichromatewith stirring. The mixture isthen filtered through sintered glass,washed with nitric acid, then dried at120°C in a desiccator.
Chromium(VI) oxide is an extremelypowerful oxidizing agent,especially to organic matter; it immediatelyinflames ethanol. It is anacidic oxide and dissolves in water toform ‘chromic acid’, a powerful oxidizingagent and cleansing fluid forglassware. At 400°C, chromium(VI)oxide loses oxygen to givechromium(III) oxide.
Preparation
Chromium(VI) oxide is prepared by heating sodium dichromate dihydrate with a slight excess of sulfuric acid in a steel tank or cast iron container: Na2Cr2O7 + 2H2SO4 → 2CrO3 + 2NaHSO4 + H2O The temperature of the mixture is kept above the melting point of chromium(VI) oxide to evaporate water and separate the top layer of sodium bisulfate from the molten chromium(VI) oxide at the bottom. Temperature control and duration of heating is very crucial in the process. Temperatures over 197°C (melting point), or allowing the molten mass to stand for a longer time, may result in decomposition of the product.
General Description
Chromium(VI) oxide (CrO3) is a hexavalent chromium that is majorly used as an oxidizing agent. It oxidizes the C-H bonds in the aromatic rings to form benzoic acid from alkyl benzene. It can be prepared by adding concentrated sulphuric acid in potassium dichromate.
Air & Water Reactions
Deliquescent. Water soluble, giving acidic solutions.
Reactivity Profile
CHROMIUM TRIOXIDE is a powerful oxidizing agent. Can react violently upon contact with reducing reagents, including organic matter, leading to ignition or explosion. Dangerously reactive with acetone, alcohols, alkali metals (sodium, potassium), ammonia, arsenic, dimethylformamide, hydrogen sulfide, phosphorus, peroxyformic acid, pyridine, selenium, sulfur, and many other chemicals [Sax, 9th ed., 1996, p. 852]. Noncombustible but can accelerate the burning of combustible materials. Sufficient heat may be generated from the reaction with combustible materials to ignite the mass. Aqueous solutions corrode many metals rapidly. Often mixed with sulfuric acid to make "cleaning solution" for glass. Used cleaning solution in closed bottles may explode due to the build up of gaseous carbon dioxide arising from oxidation of organic impurities [Bryson, W. R., Chem. Brit., 1975, 11, p. 377].
Health Hazard
Chromium(VI) oxide and other chromium(VI) salts are moderately toxic substances by ingestion; 1 to 15 g may be a fatal dose in humans. Ingestion of nonlethal doses of these compounds can cause stomach, liver, and kidney damage; symptoms may include clammy, cyanotic skin, sore throat, gastric burning, vomiting, and diarrhea. Chromic acid is irritating to the skin, and prolonged contact can cause ulceration. Inhalation of chromate dust or chromic acid mist can result in severe irritation of the nose, throat, bronchial tubes, and lungs and may cause coughing, labored breathing, and swelling of the larynx. Eye contact with chromium trioxide and its solutions can cause severe burns and possible loss of vision.
Occupational exposure to chromium(VI) compounds has been related to an increased risk of lung cancer. Several hexavalent compounds of chromium, including chromium trioxide, are listed in IARC Group 1 ("carcinogenic to humans") and are classified as "select carcinogens" under the criteria of the OSHA Laboratory Standard. Long-term exposure to chromium trioxide or chromium(VI) salts may cause ulceration of the respiratory system and skin. Exposure to chromium trioxide by inhalation or skin contact may lead to sensitization. Chromium trioxide has exhibited teratogenic activity in animal tests.
Fire Hazard
These substances will accelerate burning when involved in a fire. May explode from heat or contamination. Some may burn rapidly. Some will react explosively with hydrocarbons (fuels). May ignite combustibles (wood, paper, oil, clothing, etc.). Containers may explode when heated. Runoff may create fire or explosion hazard.
Flammability and Explosibility
Chromium(VI) oxide is not combustible but is a strong oxidizing agent and can accelerate the burning rate of combustible materials. Contact with easily oxidized organic or other combustible materials (including paper and oil) may result in ignition, violent combustion, or explosion. The use of dry chemical, carbon dioxide, Halon, or water spray extinguishers is recommended for fires involving chromium (VI) compounds.
Potential Exposure
Chromium trioxide is used in plating and metal treatment, as a corrosion inhibitor; and as an oxidant; in aluminum anodizing, dye; ink, and paint manufacturing, tanning, engraving; and photography.
storage
Chromium trioxide should be handled in a fume hood to avoid the inhalation of dust, and impermeable gloves should be worn at all times to prevent skin contact. The practice of using chromate solutions to clean glassware should be avoided. Chromium trioxide should be stored in areas separated from readily oxidized materials.
Shipping
UN1463 (anhydrous), Chromium trioxide, anhydrous, Chromium trioxide, anhydrous, Hazard Class: 5.1; Labels: 5.1-Oxidizer, 6.1-Poisonous materials, 8-Corrosive material. UN1755 (solution), Hazard class: 8; Labels: 8-Corrosive material.
Purification Methods
It separates when potassium or sodium dichromate are dissolved in conc H2SO4. Dry it in a vacuum desiccator over NaOH pellets. It is a hygroscopic, powerful oxidant and can ignite with organic compounds. It is a skin and pulmonary IRRITANT. [Keyes et al. Industrial Chemicals (Lowenheim & Moran eds.) 4th edn J. Wiley pp 270-274 1975.] CANCER SUSPECT.
Incompatibilities
Chromium trioxide is a strong oxidizer. The solution in water is a strong acid. Reacts violently with bases and is corrosive. Contact with reducing agents; fuels, organic chemicals, flammable and combustible materials, causing fire and explosion hazard. This chemical decomposes above 250C to chromic oxide and oxygen with increased fire hazard. Attacks metals in the presence of moisture.
Waste Disposal
Reduce to Cr(III). If material cannot be recovered and recycled, dispose of sludge in a chemical waste landfill.
Chromium(VI) oxide Preparation Products And Raw materials
Raw materials
1of2
chevron_rightPreparation Products
1of8
chevron_right| Supplier | Tel | Country | ProdList | Advantage | Inquiry |
|---|---|---|---|---|---|
| Evans Fine Chem | +91-9821340302 +91-9821340302 | Maharashtra, India | 286 | 58 | Inquiry |
| JSK Chemicals | +919879767970 | Gujarat, India | 3756 | 58 | Inquiry |
| Otto Chemie Pvt Ltd | +91-2222070099 +91-2266382599 | Maharashtra, India | 429 | 58 | Inquiry |
| Dhara Industries | +91-9322395199 +91-9322395199 | Mumbai, India | 190 | 58 | Inquiry |
| Merck Ltd | +91-2262109800 +91-2262109000 | Maharashtra, India | 272 | 58 | Inquiry |
| Osrics Pharmachem Pvt Ltd (Suryansh Group) | +91-1722268435 +91-9876082829 | Punjab, India | 21 | 58 | Inquiry |
| Svaks Biotech India Pvt Ltd | +91-7709222999 +91-7767806001 | Maharashtra, India | 81 | 58 | Inquiry |
| FINAR LTD | +91 - 2717 - 616717 | New Delhi, India | 660 | 58 | Inquiry |
| Maas Pharma Chemicals | +91-9958666224 | Delhi, India | 1607 | 58 | Inquiry |
| CLEARSYNTH LABS LTD. | +91-22-45045900 | Hyderabad, India | 6351 | 58 | Inquiry |
| Supplier | Advantage |
|---|---|
| Evans Fine Chem | 58 |
| JSK Chemicals | 58 |
| Otto Chemie Pvt Ltd | 58 |
| Dhara Industries | 58 |
| Merck Ltd | 58 |
| Osrics Pharmachem Pvt Ltd (Suryansh Group) | 58 |
| Svaks Biotech India Pvt Ltd | 58 |
| FINAR LTD | 58 |
| Maas Pharma Chemicals | 58 |
| CLEARSYNTH LABS LTD. | 58 |
Related articles
- Application and toxicity of Chromium(VI) oxide
- Chromium(VI) oxide has a wide range of applications, but it is highly toxic.
- Sep 7,2023
- Chromium trioxide - Hazard and Toxicity
- Chromium trioxide is not combustible but is a strong oxidizing agent and can accelerate the burning rate of combustible materi....
- Sep 11,2019
1333-82-0(Chromium(VI) oxide)Related Search:
1of4
chevron_right




