Boron oxide
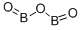
- CAS No.
- 1303-86-2
- Chemical Name:
- Boron oxide
- Synonyms
- B2O3;BORIC ANHYDRIDE;BORON TRIOXIDE;BORIC OXIDE;B-O;Boria;Boron anhydride;Boron sesquioxide;boricacid(hbo2),anhydride;BORON OXIDE
- CBNumber:
- CB1264526
- Molecular Formula:
- B2O3
- Molecular Weight:
- 69.62
- MOL File:
- 1303-86-2.mol
- MSDS File:
- SDS
- Modify Date:
- 2024/6/18 13:26:42
| Melting point | 450 °C(lit.) |
|---|---|
| Boiling point | 1860 °C |
| Density | 2.46 g/mL at 25 °C(lit.) |
| vapor density | >1 (vs air) |
| vapor pressure | 1Pa |
| Flash point | 1860°C |
| storage temp. | Inert atmosphere,Room Temperature |
| solubility | 36g/l |
| form | pellets |
| Specific Gravity | 2.46 +/- 0.01 |
| color | White |
| Odor | Odorless |
| PH | 4 (10g/l, H2O, 25℃) |
| Water Solubility | 36 g/L (25 ºC) |
| Sensitive | Hygroscopic |
| Merck | 14,1337 |
| Exposure limits |
ACGIH: TWA 10 mg/m3 OSHA: TWA 15 mg/m3 NIOSH: IDLH 2000 mg/m3; TWA 10 mg/m3 |
| Stability | Stable. Moisture sensitive. Incompatible with water. |
| InChIKey | JKWMSGQKBLHBQQ-UHFFFAOYSA-N |
| CAS DataBase Reference | 1303-86-2(CAS DataBase Reference) |
| NIST Chemistry Reference | Diboron trioxide(1303-86-2) |
| EPA Substance Registry System | Boric oxide (1303-86-2) |
SAFETY
Risk and Safety Statements
| Symbol(GHS) |  GHS08 |
|||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Signal word | Danger | |||||||||
| Hazard statements | H360D | |||||||||
| Precautionary statements | P201-P202-P280-P308+P313-P405-P501 | |||||||||
| Hazard Codes | Xi,T | |||||||||
| Risk Statements | 36/37/38-61-60 | |||||||||
| Safety Statements | 26-37/39-45-53 | |||||||||
| OEB | B | |||||||||
| OEL | TWA: 10 mg/m3 | |||||||||
| WGK Germany | 1 | |||||||||
| RTECS | ED7900000 | |||||||||
| F | 3-10 | |||||||||
| TSCA | Yes | |||||||||
| HS Code | 28100010 | |||||||||
| Toxicity | LD50 orally in Rabbit: 3150 mg/kg | |||||||||
| IDLA | 2,000 mg/m3 | |||||||||
| NFPA 704 |
|
Boron oxide price More Price(21)
| Manufacturer | Product number | Product description | CAS number | Packaging | Price | Updated | Buy |
|---|---|---|---|---|---|---|---|
| Sigma-Aldrich(India) | 339075 | Boric anhydride 99.98% trace metals basis | 1303-86-2 | 100G | ₹5802.2 | 2022-06-14 | Buy |
| Sigma-Aldrich(India) | 339075 | Boric anhydride 99.98% trace metals basis | 1303-86-2 | 500G | ₹20848.95 | 2022-06-14 | Buy |
| Sigma-Aldrich(India) | 15678 | Boric anhydride puriss. p.a., ≥98% (T) | 1303-86-2 | 50G | ₹3344.93 | 2022-06-14 | Buy |
| Sigma-Aldrich(India) | 202851 | Boric anhydride 99.999% trace metals basis | 1303-86-2 | 5G | ₹4957.85 | 2022-06-14 | Buy |
| Sigma-Aldrich(India) | 15678 | Boric anhydride puriss. p.a., ≥98% (T) | 1303-86-2 | 250G | ₹7155.33 | 2022-06-14 | Buy |
Boron oxide Chemical Properties,Uses,Production
Chemical Properties
Boron oxide is a noncombustible, colorless, semitransparent lumps or hard, white, odorless crystals, with slightly bitter taste.

Physical properties
Colorless glassy solid or vitreous crystal; hexagonal crystal system; slightly bitter taste; hygroscopic; density 2.55 g/cm3; melts at 450°C; vaporizes at 1,500°C; slightly soluble in cold water (3.3%), soluble in alcohol and boiling water (20%).
Uses
Boron oxide was used as the intermediate glass layer at a bonding temperature of 450°C. In preparation of fluxes; component of enamels and glass; catalyst in organic reaction.
In metallurgy; in analysis of silicates to determine SiO2 and alkalies; in blowpipe analysis.
Production Methods
Boric oxide is produced by thermal fusion of boric acid, forming a clear transparent glass-like solid that is subsequently ground into white vitreous granules. It is used principally in the manufacture of glass and vitreous products.
Preparation
Boric oxide is produced by treating borax with sulfuric acid in a fusion furnace. At temperatures above 750°C, the molten boric acid layer separates out from sodium sulfate. It then is decanted, cooled, and obtained in 96-97% purity. Boric acid above 99% purity may be obtained by fusing granular material.
Boric oxide may be prepared by heating boric acid.
2B(OH)3 → B2O3 + 3H2O
General Description
Colorless, semi-transparent glassy lumps or hard white odorless crystals. Mp 450°C; bp: 1860°C. Density: 2.46 g cm-3. Moderately soluble in water. Used as an insecticide; as the starting material for the synthesis of other boron compounds; as a fluxing agent in enamels and glasses; and in mixture with 2-6% boron nitride, as a bonding agent in the hot isostatic pressing of boron nitride ceramics.
Reactivity Profile
Boron oxide is non-combustible. Of generally low chemical reactivity. Reacts exothermically but slowly with water to form boric acid, a weak acid. Reacts exothermically with strong bases. May react with strong reducing agents such as metal hydrides, metal alkyls to generate flammable or explosive gases. May react violently on contact with bromine pentafluoride. Corrosive to metals in the presence of air.
Hazard
Eye and upper respiratory tract irritant.
Health Hazard
Boron oxide is an eye and respiratory irritant. In 113 workers exposed to boron oxide and boric acid dusts, there were statistically significant increases in symptoms of eye irrita- tion; dryness of the mouth, nose, and throat; sore throat; and productive cough compared with controls. The mean exposure level was 4.1mg/m3 , with a range of 1.2–8.5mg/m3 . Exposures may occasionally have exceeded 10 mg/m3 . Because of mixed exposures, the study does not indicate whether boron oxide or boric acid dust is more important in causing symp- toms, nor does it indicate the minimum duration of exposure necessary to produce symptoms.
Excessive absorption of boron oxide may lead to cardiovascular collapse, alterations in temperature regulation, and coma.
Potential Exposure
Boron oxide is used in glass manufacture and the production of other boron compounds. It is used in fluxes, enamels, drying agents, and as a catalyst.
Shipping
UN3077 Environmentally hazardous substances, solid, n.o.s., Hazard class: 9; Labels: 9—Miscellaneous hazardous material, Technical Name Required.
Incompatibilities
Incompatible with bromine pentafluoride, calcium oxide. Reacts slowly with water, forming boric acid. Reacts exothermically with alkaline material and strong bases. May react with strong reducing agents such as metal hydrides, metal alkyls to generate flammable or explosive gases. May react violently on contact with bromine pentafluoride. Corrosive to metals in the presence of air.
Boron oxide Preparation Products And Raw materials
Raw materials
Preparation Products
1of2
chevron_right| Supplier | Tel | Country | ProdList | Advantage | Inquiry |
|---|---|---|---|---|---|
| Yogi Dye Chem Industries | +91-9821098577 +91-9821098577 | Mumbai, India | 124 | 58 | Inquiry |
| PERFECT CHEMICAL | +91-9820465461 +91-9820465461 | Mumbai, India | 391 | 58 | Inquiry |
| Otto Chemie Pvt Ltd | +91-2222070099 +91-2266382599 | Maharashtra, India | 429 | 58 | Inquiry |
| Kronox Lab Sciences Pvt Ltd | +91-9313231074 +91-9313231074 | Gujarat, India | 240 | 58 | Inquiry |
| CLEARSYNTH LABS LTD. | +91-22-45045900 | Hyderabad, India | 6351 | 58 | Inquiry |
| Scientific OEM | +91-22- 2343 7546 / 2341 3094 | New Delhi, India | 1996 | 38 | Inquiry |
| ALPHA CHEMIKA | +91-22-22061123 +91-22-66382501 | Mumbai, India | 1681 | 43 | Inquiry |
| Otto Chemie Pvt. Ltd. | +91 9820041841 | Mumbai, India | 5873 | 58 | Inquiry |
| LOBA CHEMIE PVT.LTD. | 91-22-6663 6699 | Mumbai, India | 3077 | 58 | Inquiry |
| Central Drug House(P) Ltd. | 91-11-49404040 | New Delhi, India | 6160 | 58 | Inquiry |
| Supplier | Advantage |
|---|---|
| Yogi Dye Chem Industries | 58 |
| PERFECT CHEMICAL | 58 |
| Otto Chemie Pvt Ltd | 58 |
| Kronox Lab Sciences Pvt Ltd | 58 |
| CLEARSYNTH LABS LTD. | 58 |
| Scientific OEM | 38 |
| ALPHA CHEMIKA | 43 |
| Otto Chemie Pvt. Ltd. | 58 |
| LOBA CHEMIE PVT.LTD. | 58 |
| Central Drug House(P) Ltd. | 58 |
Related articles
- Boron Oxide: Unique Characteristics, Applications in Lithium-Ion Batteries and Environmental Risk
- Boron oxide, valuable for industry and battery tech, demands caution due to low toxicity but potential health risks.
- May 7,2024
- Boron oxide:Uses,Production,Reactions
- Boron oxide is a colorless transparent solid, almost always glassy (amorphous), which can be crystallized only with great diff....
- Feb 28,2023
1303-86-2(Boron oxide)Related Search:
1of4
chevron_right




