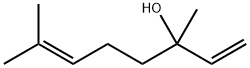
Linalool
- CAS No.
- 78-70-6
- Chemical Name:
- Linalool
- Synonyms
- 1,6-Octadien-3-ol, 3,7-dimethyl-;Linalol;3,7-Dimethyl-1,6-octadien-3-ol;LINALOOL COEUR;3,7-Dimethylocta-1,6-dien-3-ol;Linanool;Phantol;beta-Linalool;Linalool ex orange oil;2,6-dimethyl-2,7-octadien-6-ol (linalool)
- CBNumber:
- CB5347184
- Molecular Formula:
- C10H18O
- Molecular Weight:
- 154.25
- MOL File:
- 78-70-6.mol
- MSDS File:
- SDS
- Modify Date:
- 2024/6/19 22:55:17
| Melting point | 25°C |
|---|---|
| Boiling point | 194-197 °C/720 mmHg (lit.) |
| Density | 0.87 g/mL at 25 °C (lit.) |
| vapor pressure | 0.17 mm Hg ( 25 °C) |
| refractive index |
n |
| FEMA | 2635 | LINALOOL |
| Flash point | 174 °F |
| storage temp. | 2-8°C |
| solubility | ethanol: soluble1ml/4ml, clear, colorless (60% ethanol) |
| pka | 14.51±0.29(Predicted) |
| form | Liquid |
| Specific Gravity | 0.860 (20/4℃) |
| color | Clear colorless to pale yellow |
| PH | 4.5 (1.45g/l, H2O, 25℃) |
| Odor | at 100.00 %. citrus floral sweet bois de rose woody green blueberry |
| Odor Type | floral |
| explosive limit | 0.9-5.2%(V) |
| Water Solubility | 1.45 g/L (25 ºC) |
| JECFA Number | 356 |
| Merck | 14,5495 |
| BRN | 1721488 |
| Stability | Stable. Incompatible with strong oxidizing agents. Combustible. |
| InChIKey | CDOSHBSSFJOMGT-UHFFFAOYSA-N |
| LogP | 2.9 at 20℃ |
| CAS DataBase Reference | 78-70-6(CAS DataBase Reference) |
| NIST Chemistry Reference | 2,6-Dimethylocta-2,7-dien-6-ol(78-70-6) |
| EPA Substance Registry System | 3,7-Dimethyl-1,6-octadien-3-ol (78-70-6) |
SAFETY
Risk and Safety Statements
| Symbol(GHS) |  GHS07 |
|||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Signal word | Warning | |||||||||
| Hazard statements | H315-H317-H319 | |||||||||
| Precautionary statements | P261-P264-P272-P280-P302+P352-P305+P351+P338 | |||||||||
| Hazard Codes | Xi,Xn | |||||||||
| Risk Statements | 36/37/38-20/21/22 | |||||||||
| Safety Statements | 26-36 | |||||||||
| RIDADR | NA 1993 / PGIII | |||||||||
| WGK Germany | 1 | |||||||||
| RTECS | RG5775000 | |||||||||
| Autoignition Temperature | 235 °C | |||||||||
| TSCA | Yes | |||||||||
| HS Code | 29052210 | |||||||||
| Toxicity | LD50 orally in Rabbit: 2790 mg/kg LD50 dermal Rabbit 5610 mg/kg | |||||||||
| NFPA 704 |
|
Linalool price More Price(16)
| Manufacturer | Product number | Product description | CAS number | Packaging | Price | Updated | Buy |
|---|---|---|---|---|---|---|---|
| Sigma-Aldrich(India) | W263508 | Linalool ≥97%, FCC, FG | 78-70-6 | 1SAMPLE-K | ₹5141.88 | 2022-06-14 | Buy |
| Sigma-Aldrich(India) | W263508 | Linalool ≥97%, FCC, FG | 78-70-6 | 1KG | ₹6646.55 | 2022-06-14 | Buy |
| Sigma-Aldrich(India) | W263508 | Linalool ≥97%, FCC, FG | 78-70-6 | 9KG | ₹35614.25 | 2022-06-14 | Buy |
| Sigma-Aldrich(India) | W263508 | Linalool ≥97%, FCC, FG | 78-70-6 | 20KG | ₹61063.83 | 2022-06-14 | Buy |
| Sigma-Aldrich(India) | L2602 | Linalool 97% | 78-70-6 | 5G | ₹1948.5 | 2022-06-14 | Buy |
Linalool Chemical Properties,Uses,Production
Description
Linalool has a typical floral odor free from camphoraceous and
terpenic notes.1 Synthetic linalool exhibits a cleaner and fresher
note than the natural product. It can be prepared synthetically
starting from myrcene or from dehydrolinalool.
The optically active forms (d- and ι-) and the optically inactive
form occur naturally in more than 2 0 0 oils from herbs, leaves,
flowers, and wood; the ι-form is present in the largest amounts
(80 - 85%) in the distillates from leaves of Cinnamomum cam phora var. orientalis and Cinnamomum camphora var. occidentalis
and in the distillate from Cajenne rosewood; it also has been
reported in: champaca, ylang-ylang, neroli, Mexican linaloe, ber gamot, lavandin, and others; a mixture of d- and ι-linalool has
been reported in Brazil rosewood (85%); the d-form has been
found in palmarosa, mace, sweet orange-flower distillate, petit grain, coriander (60 - 70%), marjoram, Orthodon linalooliferum
(80%), and others; the inactive form has been reported in clary
sage, jasmine, and Nectandra elaiophora.
Chemical Properties
Linalool occurs as one of its
enantiomers in many essential oils, where it is often the main component. (3R)-
(?)-Linalool, for example, occurs at a concentration of 80–85% in Ho
oils from Cinnamomum camphora; rosewood oil contains about 80%. (3S)-(+)-
Linalool makes up 60–70% of coriander oil (“coriandrol”).
Linalool is used frequently in perfumery for fruity notes and for many
floral fragrance compositions (lily of the valley, lavender, and neroli). Because of its
relatively high volatility, it imparts naturalness to top notes. Since linalool is stable
in alkali, it can be used in soaps and detergents. Linalyl esters can be prepared from
linalool.Most of the manufactured linalool is used in the production of vitamin E.
Physical properties
Properties. Racemic linalool is, similarly to the individual enantiomers, a colorless liquid with a floral, fresh odor, reminiscent of lily of the valley. However, the enantiomers differ slightly in odor. Together with its esters, linalool is one of the most frequently used fragrance substances and is produced in large quantities. In the presence of acids, linalool isomerizes readily to geraniol, nerol, and α-terpineol. It is oxidized to citral, for example, by chromic acid. Oxidation with peracetic acid yields linalool oxides, which occur in small amounts in essential oils and are also used in perfumery. Hydrogenation of linalool gives tetrahydrolinalool, a stable fragrance material. Its odor is not as strong as, but is fresher than, that of linalool. Linalool can be converted into linalyl acetate by reaction with ketene or with an excess of boiling acetic anhydride.
Occurrence
The optically active forms (d- and l-) and the optically inactive form occur naturally in more than 200 oils from herbs, leaves, flowers and wood; the l-form is present in the largest amounts (80 to 85%) in the distillates from leaves of Cinnamomum camphora var. orientalis and Cinnamomum camphora var. occidentalis and in the distillate from Cajenne rosewood; it also has been reported in champaca, ylang-ylang, neroli, Mexican linaloe, bergamot and lavandin; a mixture of d- and l-linalool has been reported in Brazil rosewood (85%); the d-form has been found in palmarosa, mace, sweet orange-flower distillate, petitgrain, coriander (60 to 70%), marjoram and Orthodon linalooliferum (80%); the inactive form has been reported in clary sage, jasmine and Nectandra elaiophora. Also reported found in over 280 products including apple, citrus peel oils and juices, berries, grapes, guava, celery, peas, potato, tomato, cinnamon, cloves, cassia, cumin, ginger, mentha oils, mustard, nutmeg, pepper, thymus, cheeses, grape wines, butter, milk, rum, cider, tea, passion fruit, olive, mango, beans, coriander, cardamom and rice.
Uses
linalool is a fragrant component of both lavender and coriander. It can be incorporated into cosmetics for perfuming, deodorant, or odor-masking activity.
Definition
ChEBI: A monoterpenoid that is octa-1,6-diene substituted by methyl groups at positions 3 and 7 and a hydroxy group at position 3. It has been isolated from plants like Ocimum canum.
General Description
Linalool is a monoterepene compound. It is the major component of many essential oils. Anti-inflammatory properties of (?) linalool, a naturally occurring enantiomer, has been studied. It is also the major constituent of the basil and thyme essential oil and its anti-microbial effect was determined using the agar well diffusion assay. Linalool is reported to have dose-dependent marked sedative effects at the central nervous system (CNS), including hypnotic, anticonvulsant and hypothermic properties. Linalool is reported to have a lemon like odor.
Contact allergens
Linalool is a terpene chief constituent of linaloe oil,also found in oils of Ceylon cinnamon, sassafras,orange flower, bergamot, Artemisia balchanorum, ylang-ylang. This frequently used scented substance is a sensitizer by the way of primary or secondary oxida-tion products. As a fragrance allergen, linalool has to be mentioned by name in cosmetics within the EU
Anticancer Research
Studies of antitumor activities and toxicity were done on solid S-180 tumor-bearingSwiss albino mice. It results in an induction of oxidative stress with an antitumoractivities result. In comparison with cyclophosphamide, antioxidant effects wereobserved in the liver and modulation of proliferation of spleen cells in tumor-bearingmice challenged with lipopolysaccharides, while both were seriously affected bycyclophosphamide (Costa et al. 2015).
Linalool Preparation Products And Raw materials
Raw materials
1of4
chevron_rightPreparation Products
1of3
chevron_right| Supplier | Tel | Country | ProdList | Advantage | Inquiry |
|---|---|---|---|---|---|
| Nagar Haveli Perfumes & Aromatics | +917506551149 | Mumbai, India | 230 | 58 | Inquiry |
| NEERU MENTHOL PRIVATE LIMITED | +91-8884114465 +91-8884114465 | Uttar Pradesh, India | 44 | 58 | Inquiry |
| Expo Essential Oils | +919210059326 | New Delhi, India | 72 | 58 | Inquiry |
| AOS Products Pvt Ltd | +91-8285111618 +91-8285111618 | Uttar Pradesh, India | 76 | 58 | Inquiry |
| Neeru Menthol Pvt Ltd | +91-8884114465 +91-9837322285 | New Delhi, India | 70 | 58 | Inquiry |
| D D Shah Essential Oil Co | +91-2222850577 +91-2222850577 | Maharashtra, India | 34 | 58 | Inquiry |
| HRV Global Life Sciences | +91-9820219686 +91-9820219686 | Telangana, India | 379 | 58 | Inquiry |
| Ghaziabad Aromatics | +91-9811327736 +91-9654012346 | Uttar Pradesh, India | 80 | 58 | Inquiry |
| Eco Organics | +91-9412251810 +91-9720999980 | Uttar Pradesh, India | 23 | 58 | Inquiry |
| JD Chem India | +91-9899117932 +91-9555260045 | New Delhi, India | 41 | 58 | Inquiry |
| Supplier | Advantage |
|---|---|
| Nagar Haveli Perfumes & Aromatics | 58 |
| NEERU MENTHOL PRIVATE LIMITED | 58 |
| Expo Essential Oils | 58 |
| AOS Products Pvt Ltd | 58 |
| Neeru Menthol Pvt Ltd | 58 |
| D D Shah Essential Oil Co | 58 |
| HRV Global Life Sciences | 58 |
| Ghaziabad Aromatics | 58 |
| Eco Organics | 58 |
| JD Chem India | 58 |
Related articles
- Linalool: Properties, Synthesis and Uses
- Linalool is a fragrance and flavor substance. It is a blending raw material for various artificial essential oils.
- Mar 5,2024
- Anticonvulsant properties of linalool
- Linalool is a component of many essential oils, including orange, lavender, rose, rosewood, and coriander. The main problem t....
- Nov 12,2019
78-70-6(Linalool)Related Search:
1of4
chevron_right




