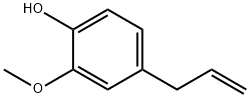
Eugenol
- CAS No.
- 97-53-0
- Chemical Name:
- Eugenol
- Synonyms
- 4-ALLYL-2-METHOXYPHENOL;Eug;Engenol;4-allyl-2-methoxyphenol (eugenol);p-Eugenol;FEMA 2467;Eugenol,99%;5-allylguaiacol;2-Methoxy-4-prop-2-enylphenol;2-METHOXY-4-(2-PROPENYL)PHENOL
- CBNumber:
- CB7208326
- Molecular Formula:
- C10H12O2
- Molecular Weight:
- 164.2
- MOL File:
- 97-53-0.mol
- MSDS File:
- SDS
- Modify Date:
- 2024/7/26 15:15:06
| Melting point | -12--10 °C (lit.) |
|---|---|
| Boiling point | 254 °C (lit.) |
| Density | 1.067 g/mL at 25 °C (lit.) |
| vapor pressure | <0.1 hPa (25 °C) |
| FEMA | 2467 | EUGENOL |
| refractive index |
n |
| Flash point | >230 °F |
| storage temp. | 2-8°C |
| solubility | 2.46g/l |
| form | Liquid |
| pka | pKa 9.8 (Uncertain) |
| color | Clear pale yellow to yellow |
| Odor | at 10.00 % in dipropylene glycol. sweet spicy clove woody |
| Odor Type | spicy |
| Water Solubility | slightly soluble |
| Sensitive | Air Sensitive |
| Merck | 14,3898 |
| JECFA Number | 1529 |
| BRN | 1366759 |
| Dielectric constant | 6.1(18℃) |
| Stability | Stable. Combustible. Incompatible with strong oxidizing agents. |
| InChIKey | RRAFCDWBNXTKKO-UHFFFAOYSA-N |
| LogP | 1.83 at 30℃ |
| CAS DataBase Reference | 97-53-0(CAS DataBase Reference) |
| IARC | 3 (Vol. 36, Sup 7) 1987 |
| NIST Chemistry Reference | Eugenol(97-53-0) |
| EPA Substance Registry System | Eugenol (97-53-0) |
SAFETY
Risk and Safety Statements
| Symbol(GHS) |  GHS07 |
|||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Signal word | Warning | |||||||||
| Hazard statements | H317-H319 | |||||||||
| Precautionary statements | P261-P264-P272-P280-P302+P352-P305+P351+P338 | |||||||||
| Hazard Codes | Xn,Xi | |||||||||
| Risk Statements | 22-36/37/38-42/43-38-40-43-36/38 | |||||||||
| Safety Statements | 26-36-24/25-23-36/37 | |||||||||
| RIDADR | UN1230 - class 3 - PG 2 - Methanol, solution | |||||||||
| WGK Germany | 1 | |||||||||
| RTECS | SJ4375000 | |||||||||
| F | 10-23 | |||||||||
| TSCA | Yes | |||||||||
| HS Code | 29095090 | |||||||||
| Toxicity | LD50 in rats, mice (mg/kg): 2680, 3000 orally (Hagan) | |||||||||
| NFPA 704 |
|
Eugenol price More Price(24)
| Manufacturer | Product number | Product description | CAS number | Packaging | Price | Updated | Buy |
|---|---|---|---|---|---|---|---|
| Sigma-Aldrich(India) | W246719 | Eugenol natural, ≥98%, FG | 97-53-0 | 1SAMPLE-K | ₹5347.55 | 2022-06-14 | Buy |
| Sigma-Aldrich(India) | W246719 | Eugenol natural, ≥98%, FG | 97-53-0 | 1KG | ₹9341.98 | 2022-06-14 | Buy |
| Sigma-Aldrich(India) | W246719 | Eugenol natural, ≥98%, FG | 97-53-0 | 5KG | ₹42628.85 | 2022-06-14 | Buy |
| Sigma-Aldrich(India) | W246719 | Eugenol natural, ≥98%, FG | 97-53-0 | 10KG | ₹71044.48 | 2022-06-14 | Buy |
| Sigma-Aldrich(India) | W246700 | Eugenol ≥98%, FCC, FG | 97-53-0 | 1SAMPLE-K | ₹5141.88 | 2022-06-14 | Buy |
Eugenol Chemical Properties,Uses,Production
Description
Sensitization to eugenol mainly occurs in those in dental professions. Eugenol is contained in the "fragrance mix".
Chemical Properties
Eugenol is the main component
of several essential oils; clove leaf oil and cinnamon leaf oilmay contain>90%.Eugenol occurs in small amounts in many other essential oils. It is a colorless to
slightly yellow liquid with a spicy, clove odor.
Catalytic hydrogenation (e.g., in the presence of noble metal catalysts) yields
dihydroeugenol. Isoeugenol is obtained fromeugenol by shifting the double bond.
Esterification and etherification of the hydroxy group of eugenol yield valuable
fragrance and flavor materials (e.g., eugenol acetate and eugenol methyl ether).
Occurrence
Reported found as a constituent in several volatile oils: clove oil, laurel and cinnamon leaf oil. Smaller amounts of eugenol are also present in the oil of camphor, Java citronella, California laurel and acacia flowers; remarkable amounts of eugenol are found in Ocimum sanctum (70%) and Ocimum gratissimum (60%). Eugenol is also found in the oil from violet flowers (21%); in some plants, eugenol probably occurs as glucoside. Reported found in apricot, citrus oils, raspberry, strawberry, tomato, anise, cinnamon (leaf, bark and roots), clove bud and stem, nutmeg, mace, pepper, smoked fish, beer, whiskey, grape wines, cocoa, mango, tarragon, laurel, myrtle leaf, and pimento berry and leaf.
Uses
Eugenol is a dental compound which shows cytotoxicity to human oral squamous cell carcinoma and oral cells. When glucosylated, this compound exhibits anti-inflammatory activity.
Preparation
Since sufficient eugenol can be isolated from cheap essential oils, synthesis is not industrially important. Eugenol is still preferentially isolated from clove leaf and cinnamon leaf oil (e.g., by extraction with sodium hydroxide solution). Nonphenolic materials are then removed by steam distillation. After the alkaline solution is acidified at low temperature, pure eugenol is obtained by distillation.
Definition
ChEBI: A phenylpropanoid formally derived from guaiacol with an allyl chain substituted para to the hydroxy group.
General Description
Clear colorless pale yellow or amber-colored liquid. Odor of cloves. Spicy pungent taste.
Air & Water Reactions
Darkens and thickens on exposure to air. Also darkens with age. Eugenol may decompose on exposure to light. Insoluble in water.
Reactivity Profile
Eugenol is incompatible with strong oxidizers. This includes ferric chloride and potassium permanganate. Eugenol reacts with strong alkalis. Eugenol is incompatible with iron and zinc.
Hazard
Questionable carcinogen.
Fire Hazard
Eugenol is combustible.
Contact allergens
Eugenol is a fragrance allergen obtained from many natural sources. Occupational sensitization to eugenol may occur in dental profession workers. Eugenol is contained in “fragrance mix” and has to be listed by name in cosmetics within the EU.
Clinical Use
4-Allyl-2-methoxyphenol is obtained primarily from cloveoil. It is a pale-yellow liquid with a strong aroma of clovesand a pungent taste. Eugenol is only slightly soluble in waterbut is miscible with alcohol and other organic solvents.Eugenol possesses both local anesthetic and antiseptic activityand can be directly applied on a piece of cotton to relievetoothaches. Eugenol is also used in mouthwashes because ofits antiseptic property and pleasant taste. The phenol coefficientof eugenol is 14.4.
Anticancer Research
This compound was tested on a model of skin tumor induced by DMBA croton oilin Swiss mice. The eugenol affects the cellular proliferation by increasing apoptosiscellular death. There is evidence for a downregulation of c-myc, H-ras, and Bcl-2expression and an upregulation of p53, Bax, and active caspase-3 (Grondona et al.2014).
Safety Profile
Moderately toxic by ingestion, intraperitoneal, and subcutaneous routes. Human mutation data reported. A human skin irritant. Questionable carcinogen with experimental carcinogenic and tumorigenic data. Combustible liquid. When heated to decomposition it emits acrid smoke and irritating fumes. See also ALLYL COMPOUNDS.
Metabolism
No absorption of eugenol occurred within 2hr of application to the intact shaved skin of mice (Meyer & Meyer, 1959). Following ip injec tion of [14C]eugenol into rats, radioactivity was dis tributed in various organs and the presence of 14CO2 in the expired air indicated the demethylation of eugenol (Weinberg, Rabinowitz, Zanger & Gennaro, 1972). Over 70% of an oral dose of eugenol was excreted in the urine of rabbits (Schr?der & Vollmer, 1932).
Purification Methods
Fractional distillation of eugenol gives a pale yellow liquid which darkens and thickens on exposure to air. It should be stored under N2 at -20o. [Waterman & Priedster Recl Trav Chim Pays-Bas 48 1272 1929, Beilstein 6 H 961, 6 IV 6337.]
Eugenol Preparation Products And Raw materials
Raw materials
1of4
chevron_rightPreparation Products
| Supplier | Tel | Country | ProdList | Advantage | Inquiry |
|---|---|---|---|---|---|
| Nagar Haveli Perfumes & Aromatics | +917506551149 | Mumbai, India | 230 | 58 | Inquiry |
| JSK Chemicals | +919879767970 | Gujarat, India | 3756 | 58 | Inquiry |
| Yasho Industries Ltd | +91-2262510100 +91-8291955977 | Mumbai, India | 70 | 58 | Inquiry |
| Ghaziabad Aromatics | +91-9811327736 +91-9654012346 | Uttar Pradesh, India | 80 | 58 | Inquiry |
| PSC Aromatics | +91-9655115000 +91-9443115000 | Tamil Nadu, India | 23 | 58 | Inquiry |
| Jindal Drugs Limilted | +91-2243418888 +91-2243418888 | Maharashtra, India | 11 | 58 | Inquiry |
| Swati Menthol and Allied Chemicals Ltd | +91-9837848873 +91-9837848871 | Uttar Pradesh, India | 49 | 58 | Inquiry |
| Aamirav Ingredients And Specialties Pvt Ltd | +91-2224980102 +91-9820077171 | Maharashtra, India | 138 | 58 | Inquiry |
| Arpan Aromatics | +91-9824514546 +91-9824029204 | Gujarat, India | 59 | 58 | Inquiry |
| Neeru Menthol Pvt Ltd | +91-8884114465 +91-9837322285 | New Delhi, India | 70 | 58 | Inquiry |
Related articles
- Eugenol: Overview and its Biological Activities
- Eugenol from clove oil exhibits antioxidant and antimicrobial properties, scavenging free radicals and disrupting bacterial me....
- May 15,2024
- Application and Pharmacology of Eugenol
- Eugenol is a phenylpropane compound existing in Syringa plants. It is a liquid with lilac flower flavor. Its chemical name is ....
- Jun 30,2022
- Introduction of eugenol
- Eugenol (C10H12O2) (CAS No:?97-53-0) is a volatile phenolic constituent of clove essential oil obtained from Eugenia caryophyl....
- Jan 14,2022
97-53-0(Eugenol)Related Search:
1of4
chevron_right




