Sodium tetraborate decahydrate
- CAS No.
- 1303-96-4
- Chemical Name:
- Sodium tetraborate decahydrate
- Synonyms
- BORAX;SODIUM BORATE;BORAX DECAHYDRATE;SODIUM BORATE DECAHYDRATE;neobor;pengsha;antipyonin;BORAX 10 MOL;bura;jaikin
- CBNumber:
- CB1187397
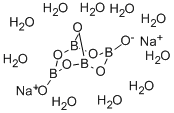
- Molecular Formula:
- B4H20Na2O17
- Molecular Weight:
- 381.37
- MOL File:
- 1303-96-4.mol
- MSDS File:
- SDS
- Modify Date:
- 2024/5/1 11:53:09
| Melting point | 75 °C |
|---|---|
| Boiling point | 320°C |
| Density | 1.73 g/mL at 25 °C(lit.) |
| vapor pressure | 0.213 hPa (20 °C) |
| storage temp. | 2-8°C |
| solubility | H2O: 0.1 M at 20 °C, clear, colorless |
| form | Powder/Solid |
| Specific Gravity | 1.73 |
| color | White, gray, bluish or greenish white |
| PH | 9.21(1 mM solution);9.17(10 mM solution);9.05(100 mM solution); |
| Odor | Odorless |
| PH Range | 9.2 |
| Water Solubility | 60 g/L (20 ºC) |
| λmax |
λ: 260 nm Amax: 0.012 λ: 280 nm Amax: 0.010 |
| Merck | 14,8590 |
| Exposure limits |
ACGIH: TWA 2 mg/m3; STEL 6 mg/m3 NIOSH: TWA 5 mg/m3 |
| InChIKey | CDMADVZSLOHIFP-UHFFFAOYSA-N |
| CAS DataBase Reference | 1303-96-4 |
| EPA Substance Registry System | Borax (1303-96-4) |
SAFETY
Risk and Safety Statements
| Symbol(GHS) |   GHS07,GHS08 |
|||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Signal word | Danger | |||||||||
| Hazard statements | H319-H360FD | |||||||||
| Precautionary statements | P202-P264-P280-P305+P351+P338-P308+P313-P337+P313 | |||||||||
| Hazard Codes | Xn,T | |||||||||
| Risk Statements | 62-61-60 | |||||||||
| Safety Statements | 22-24/25-36/37-45-53 | |||||||||
| OEB | B | |||||||||
| OEL | TWA: 5 mg/m3 | |||||||||
| RIDADR | UN 1458 | |||||||||
| WGK Germany | 1 | |||||||||
| RTECS | VZ2275000 | |||||||||
| TSCA | Yes | |||||||||
| HS Code | 28401990 | |||||||||
| Toxicity | LD50 orally in rats: 5.66 g/kg (Smyth) | |||||||||
| NFPA 704 |
|
Sodium tetraborate decahydrate price More Price(31)
| Manufacturer | Product number | Product description | CAS number | Packaging | Price | Updated | Buy |
|---|---|---|---|---|---|---|---|
| Sigma-Aldrich(India) | S9640 | Sodium tetraborate decahydrate ACS reagent, ≥99.5% | 1303-96-4 | 500G | ₹4849.6 | 2022-06-14 | Buy |
| Sigma-Aldrich(India) | S9640 | Sodium tetraborate decahydrate ACS reagent, ≥99.5% | 1303-96-4 | 25G | ₹6430.05 | 2022-06-14 | Buy |
| Sigma-Aldrich(India) | S9640 | Sodium tetraborate decahydrate ACS reagent, ≥99.5% | 1303-96-4 | 2.5KG | ₹9428.58 | 2022-06-14 | Buy |
| Sigma-Aldrich(India) | S9640 | Sodium tetraborate decahydrate ACS reagent, ≥99.5% | 1303-96-4 | 4X2.5KG | ₹21141.23 | 2022-06-14 | Buy |
| Sigma-Aldrich(India) | B9876 | Sodium tetraborate decahydrate ReagentPlus?, ≥99.5% | 1303-96-4 | 1KG | ₹3453.18 | 2022-06-14 | Buy |
Sodium tetraborate decahydrate Chemical Properties,Uses,Production
Chemical Properties
White cryst. powder
Physical properties
White monoclinic crystal; density 1.73 g/cm3; decomposes at 75°C; soluble in water; the vapor pressure of the pure compound 1.6 torr at 20°C and that of a saturated solution 130 torr at 58°C; the pH of a 1% aqueous solution 9.24 (the pH is nearly independent of concentration); readily dissolves in alcohols.
Occurrence
Borax decahydrate occurs in nature as mineral, borax (tincal). It is one of the most common sodium borate ores. The compound has several industrial applications. The refined material is mostly used in household cleaning products. It is used to make pyrex and other borosilicate glasses. Borax is added to fertilizers in small quantities as a source of boron, as a trace nutrient for plants. High purity grade borax is used in cosmetics, toilet products and electrolytic capacitors. It also is used in fire retardants, adhesives and herbicides.
Uses
Sodium Tetraborate Decahydrate is used as a buffer in antigetn-retrieval procedure for bromodeoxyuridine immunolabeling with concurrent labeling of nuclear DNA and antigens damaged by hydrochloric acid pretreatment.
Production Methods
Sodium borate can be prepared from minerals such as borosodium calcite, pandermite, or tinkal; these are natural sodium or calcium borates. Treatment of the mineral with sodium carbonate and sodium hydrogencarbonate yields the sodium borate decahydrate. In the USA, brine from salt lakes is also an important source of sodium borate.
Hazard
Toxic by inhalation.
Health Hazard
Borates are irritants of the eyes, nose, and throat; at high concentrations ingestion of the compounds can result in gastrointestinal irritation, kidney injury, and even death from central nervous system depression or cardiovascular collapse.
Pharmaceutical Applications
Sodium borate is used in pharmaceutical applications similarly to
boric acid (see Boric Acid). It has been used externally as a mild
astringent and as an emulsifying agent in creams. It has also been
used in lozenges, mouthwashes, otic preparations (0.3% w/v), and
ophthalmic solutions (0.03–1.0% w/v). Sodium borate has additionally
been investigated in the prevention of crystal formation in
freeze-dried solutions.
Preparations of sodium borate in honey have historically been
used as paints for the throat, tongue, and mouth, but such use is
now inadvisable because of concerns about toxicity in such applications. Sodium borate is also used in cosmetics
such as moisturizers, deodorants, and shampoos.
Agricultural Uses
Solubor is a type of borate containing 20.3% boron. It is chemically a polyborate, similar to borax, and is represented as Na2B2O7?5H2O +Na2B10O16?10H2O. is a finely-ground, white product specially designed for foliar, liquid or dust applications, to correct boron deficiency.
Safety Profile
Experimental poison by subcutaneous route. Moderately toxic to humans by ingestion. Moderately toxic experimentally by ingestion, intravenous, and intraperitoneal routes. Experimental reproductive effects. Mutation data reported. Ingestion of 5-10 g of borax by children can cause severe vomiting, diarrhea, shock, death. Incompatible with acids, metallic salts. When heated to decomposition it emits toxic fumes of Na2O, boron. See also BORON COMPOUNDS. Used in ant poisons, for fly control around refuse and manure piles, as a larvicide, in manufacture of glazes, enamels, cleaning compounds, and in soldering metals.
Safety
Sodium borate has weak bacteriostatic and astringent properties.
Historically, sodium borate has been used as a disinfectant in skin
lotions and eye-, nose-, and mouthwashes. However, boric acid is
easily absorbed via mucous membranes and damaged skin, and
severe toxicity has been observed, especially in babies and
children. Consequently, the use of sodium borate as a disinfectant
is now considered somewhat obsolete and careful use is recommended.
The toxic effects of sodium borate include vomiting,
diarrhea, erythema, CNS depression, and kidney damage. The
lethal oral intake is approximately 20 g in adults and 5 g in
children.
LD50 (guinea pig, oral): 5.33 g/kg
LD50 (mouse, IP): 2.711 g/kg
LD50 (mouse, IV): 1.320 g/kg
LD50 (mouse, oral): 2.0 g/kg
LD50 (rat, oral): 2.66 g/kg
Potential Exposure
Borax is used as a soldering flux, preservative against wood fungus; and as an antiseptic. Used in ant poisons, for fly control around refuse and manure piles, as a larvicide. It is used in the manufacture of enamels and glazes, fiberglass insulation; sodium perborate bleach; in tanning, cleaning compounds; for fireproofing fabrics and wood; and in artificial aging of wood.
Carcinogenicity
Sodium borate tested negatively in the Ames bioassay but was found to be cytotoxic to cultured human fibroblasts.
storage
Sodium borate should be stored in a well-closed container in a cool, dry, place.
Shipping
UN3077 Environmentally hazardous substances, solid, n.o.s., Hazard class: 9; Labels: 9—Miscellaneous hazardous material, Technical Name Required.
Purification Methods
Crystallise the borate from water (3.3mL/g), keeping below 55o to avoid formation of the pentahydrate. Filter it off at the pump, wash it with water and equilibrate it for several days in a desiccator containing an aqueous solution saturated with respect to sucrose and NaCl. Borax can be prepared more quickly (but its water content is somewhat variable) by washing the recrystallised material at the pump with water, followed by 95% EtOH, then Et2O, and dried in air at room temperature for 12-18hours on a clock glass. [Becher in Handbook of Preparative Inorganic Chemistry (Ed. Brauer) Academic Press Vol I pp 794-795 1963.]
Incompatibilities
Dissolves in water forming a basic solution. Boron dust may form explosive mixture with air. Contact with strong oxidizers may be violent. Boron is incompatible with ammonia, bromine tetrafluoride, cesium carbide, chlorine, fluorine, interhalogens, iodic acid, lead dioxide, nitric acid, nitric oxide, nitrosyl fluoride, nitrous oxide, potassium nitrite, rubidium carbide, silver fluoride.
Waste Disposal
Borax, dehydrated: The material is diluted to the recommended provisional limit (0.10 mg/L) in water. The pH is adjusted to between 6.5 and 9.1 and then the material can be discharged into sewers or natural streams.
Regulatory Status
Accepted for use as a food additive in Europe. Included in the FDA Inactive Ingredients Database (otic preparations; ophthalmic solutions and suspensions). Included in nonparenteral medicines licensed in the UK, Italy, France, Germany, and Japan. Included in the Canadian List of Acceptable Non-medicinal Ingredients.
Sodium tetraborate decahydrate Preparation Products And Raw materials
Raw materials
1of2
chevron_rightPreparation Products
| Supplier | Tel | Country | ProdList | Advantage | Inquiry |
|---|---|---|---|---|---|
| DMCC Specialty Chemical Co Ltd | +91-9892080813 +91-9892080813 | Mumbai, India | 26 | 58 | Inquiry |
| ADVENT CHEMBIO PRIVATE LIMITED | +91-9821524116 +91-9821524116 | Mumbai, India | 353 | 58 | Inquiry |
| sheetalchemicals | +91-9820130159 +91-9820130159 | Maharashtra, India | 77 | 58 | Inquiry |
| PERFECT CHEMICAL | +91-9820465461 +91-9820465461 | Mumbai, India | 391 | 58 | Inquiry |
| ALLIPO CHEMICALS | +91-8238998187 +91-8238998187 | Gujarat, India | 32 | 58 | Inquiry |
| JSK Chemicals | +919879767970 | Gujarat, India | 3756 | 58 | Inquiry |
| UNILOSA INTERNATINAL PRIVATE LIMITED | +91-9999069917 +91-9999069917 | New Delhi, India | 152 | 58 | Inquiry |
| Kronox Lab Sciences Pvt Ltd | +91-9313231074 +91-9313231074 | Gujarat, India | 240 | 58 | Inquiry |
| Fluobor India Pvt Ltd | +91-8691012149 +91-8691012149 | Maharashtra, India | 7 | 58 | Inquiry |
| Sujata Chemicals | +91-9898025496 +91-9898025496 | Gujarat, India | 73 | 58 | Inquiry |
| Supplier | Advantage |
|---|---|
| DMCC Specialty Chemical Co Ltd | 58 |
| ADVENT CHEMBIO PRIVATE LIMITED | 58 |
| sheetalchemicals | 58 |
| PERFECT CHEMICAL | 58 |
| ALLIPO CHEMICALS | 58 |
| JSK Chemicals | 58 |
| UNILOSA INTERNATINAL PRIVATE LIMITED | 58 |
| Kronox Lab Sciences Pvt Ltd | 58 |
| Fluobor India Pvt Ltd | 58 |
| Sujata Chemicals | 58 |
Related Qustion
- Q:Can you mix borax and vinegar?
- A:Borax and vinegar are two safe ingredients that can be combined to create a good general cleaning solution.
- Mar 13,2024





