Trichloroethylene
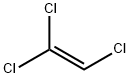
- CAS No.
- 79-01-6
- Chemical Name:
- Trichloroethylene
- Synonyms
- TCE;TRI;TRICHLORETHYLENE;TRICHLOROETHENE;1,1,2-TRICHLOROETHENE;1979/1/6;Triol;C2HCl3;Trichlorethylen;Triad
- CBNumber:
- CB5406573
- Molecular Formula:
- C2HCl3
- Molecular Weight:
- 131.39
- MOL File:
- 79-01-6.mol
- MSDS File:
- SDS
- Modify Date:
- 2024/3/19 17:10:56
| Melting point | -86 °C |
|---|---|
| Boiling point | 87 °C |
| Density | 1.463 g/mL at 25 °C(lit.) |
| vapor density | 4.5 (vs air) |
| vapor pressure | 61 mm Hg ( 20 °C) |
| refractive index |
n |
| Flash point | 90°C |
| storage temp. | 2-8°C |
| solubility | Soluble in acetone, ethanol, chloroform, ether (U.S. EPA, 1985), and other organic solvents including bromoform, carbon tetrachloride, methylene chloride, trichloroethylene, and tetrachloroethylene. |
| form | Liquid |
| color | Clear colorless |
| Odor | Chloroform-like; ethereal. |
| Odor Threshold | 3.9ppm |
| Water Solubility | Slightly soluble. 0.11 g/100 mL |
| Merck | 14,9639 |
| BRN | 1736782 |
| Henry's Law Constant | 3.14 at 1.8 °C, 8.47 at 21.6 °C, 19.0 at 40.0 °C, 26.5 at 50 °C, 35.8 at 60 °C, 56.6 at 70 °C (EPICS-GC, Shimotori and Arnold, 2003) |
| Exposure limits | TLV-TWA 50 ppm (~270 mg/m3) (ACGIH), 100 ppm (MSHA and OSHA); TLV-STEL 200 ppm (ACGIH); ceiling 200 ppm (OSHA); carcinogenicity: Animal Lim ited Evidence, Human Inadequate Evidence (IARC). |
| Dielectric constant | 3.4(16℃) |
| Stability | Stable. Incompatible with oxidizing agents, aluminium, magnesium, strong bases, reducing agents. Light-sensitive. Reacts violently with many metals, ozone, potassium nitrate, potassium hydroxide, sodium hydroxide. |
| LogP | 2.53 at 20℃ |
| CAS DataBase Reference | 79-01-6(CAS DataBase Reference) |
| IARC | 1 (Vol. Sup 7, 63, 106) 2014 |
| NIST Chemistry Reference | Trichloroethylene(79-01-6) |
| EPA Substance Registry System | Trichloroethylene (79-01-6) |
SAFETY
Risk and Safety Statements
| Symbol(GHS) |   GHS07,GHS08 |
|||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Signal word | Danger | |||||||||
| Hazard statements | H315-H317-H319-H336-H341-H350-H412 | |||||||||
| Precautionary statements | P202-P273-P280-P302+P352-P305+P351+P338-P308+P313 | |||||||||
| Hazard Codes | T,F | |||||||||
| Risk Statements | 45-36/38-52/53-67-68-39/23/24/25-23/24/25-11 | |||||||||
| Safety Statements | 53-45-61-36/37-16-7 | |||||||||
| RIDADR | UN 1710 6.1/PG 3 | |||||||||
| OEB | B | |||||||||
| OEL | TWA: 25.0 ppm, STEL: 2.0 ppm | |||||||||
| WGK Germany | 3 | |||||||||
| RTECS | KX4550000 | |||||||||
| TSCA | Yes | |||||||||
| HazardClass | 6.1 | |||||||||
| PackingGroup | III | |||||||||
| HS Code | 29032200 | |||||||||
| Toxicity | LD50 orally in rats: 4.92 ml/kg; LC (4 hrs) in rats: 8000 ppm (Smyth) | |||||||||
| IDLA | 1,000 ppm | |||||||||
| NFPA 704 |
|
Trichloroethylene price More Price(20)
| Manufacturer | Product number | Product description | CAS number | Packaging | Price | Updated | Buy |
|---|---|---|---|---|---|---|---|
| Sigma-Aldrich(India) | 372145 | Trichloroethylene anhydrous, contains 40?ppm diisopropylamine as stabilizer, ≥99% | 79-01-6 | 100ML | ₹3301.63 | 2022-06-14 | Buy |
| Sigma-Aldrich(India) | 372145 | Trichloroethylene anhydrous, contains 40?ppm diisopropylamine as stabilizer, ≥99% | 79-01-6 | 1L | ₹19333.45 | 2022-06-14 | Buy |
| Sigma-Aldrich(India) | 1.00958 | Trichloroethylene EMPLURA? | 79-01-6 | 1009580521 | ₹990 | 2022-06-14 | Buy |
| Sigma-Aldrich(India) | 1.00958 | Trichloroethylene EMPLURA? | 79-01-6 | 1009582521 | ₹3510 | 2022-06-14 | Buy |
| Sigma-Aldrich(India) | 1.00958 | Trichloroethylene EMPLURA? | 79-01-6 | 1009589027 | ₹15910 | 2022-06-14 | Buy |
Trichloroethylene Chemical Properties,Uses,Production
Description
Trichloroethylene (IUPAC), CHClCCl2, is a stable, low-boiling, colorless liquid with a chloroform-like odor. It is not corrosive to the common metals even in the presence of moisture. It is slightly soluble in water and is nonflammable. It is toxic by inhalation, with a TLV of 50 ppm and an IDLH of 1000 ppm in air. The FDA has prohibited its use in foods, drugs, and cosmetics. The four-digit UN identification number is 1710. The NFPA 704 designation is health 2, flammability 1, and reactivity 0. Its primary uses are in metal degreasing, dry cleaning, as a refrigerant and fumigant, and for drying electronic parts.
Chemical Properties
Trichloroethylene, a colorless (often dyed blue), nonflammable, noncorrosive liquid that has the “sweet” odor characteristic of some chlorinated hydrocarbons. The Odor Threshold is 25-50 ppm.
Physical properties
Clear, colorless, watery-liquid with a chloroform-like odor. Odor threshold concentrations determined in air were 21.4 ppmv (Leonardos et al., 1969) and 3.9 ppmv (Nagata and Takeuchi, 1990). The average least detectable odor threshold concentrations in water at 60 °C and in air at 40 °C were 10 and 2.6 mg/L, respectively (Alexander et al., 1982).
Uses
Trichloroethylene is used as a solvent, in drycleaning, in degreasing, and in limited use asa surgical anesthetic.
Production Methods
TCE has been in commercial use for almost 60 years. TCE has been used as a solvent because of its powerful ability to dissolve fats, greases, and waxes. It has been widely used in the dry cleaning industry and as a metal degreaser and in the electronic components industry where workers have been observed using it as a cleaning solvent without any protective equipment, thus allowing uncontrolled skin contact and inhalation exposures.
Definition
ChEBI: A member of the class of chloroethenes that is ethene substituted by chloro groups at positions 1, 1 and 2.
General Description
A clear colorless volatile liquid having a chloroform-like odor. Denser than water and is slightly soluble in water. Noncombustible. Used as a solvent, fumigant, in the manufacture of other chemicals, and for many other uses.
Air & Water Reactions
Slightly soluble in water.
Reactivity Profile
Trichloroethylene has been determined experimentally that mixtures of finely divided barium metal and a number of halogenated hydrocarbons possess an explosive capability. Specifically, impact sensitivity tests have shown that granular barium in contact with monofluorotrichloromethane, trichlorotrifluoroethane, carbon tetrachloride, Trichloroethylene, or tetrachloroethylene can detonate (ASESB Pot. Incid. 39. 1968; Chem. Eng. News 46(9):38. 1968). Trichloroethylene has been determined experimentally that a mixture of beryllium powder with carbon tetrachloride or with Trichloroethylene will flash or spark on heavy impact (ASESB Pot. Incid. 39. 1968). A mixture of powdered magnesium with Trichloroethylene or with carbon tetrachloride will flash or spark under heavy impact (ASESB Pot. Incid, 39. 1968).
Health Hazard
The toxic effects manifested in humansfrom inhaling trichloroethylene vapors areheadache, dizziness, drowsiness, fatigue, andvisual disturbances. A 2-hour exposure to a1000-ppm concentration affected the visualperception. Higher concentrations can pro duce narcotic effects. Heavy exposures maycause death due to respiratory failure or car diac arrest. A 4-hour exposure to 8000 ppmwas lethal to rats. Chronic exposure causedincrease in kidney and liver weights in testanimals.
The symptoms of poisoning from oralintake of trichloroethylene are nausea, vom iting, diarrhea, and gastric disturbances. Theacute oral toxicity, however, is low. Theoral LD50 value in mice is in the range2500 mg/kg. Trichloroethylene metabolizesto trichloroacetic acid, which is excreted inthe urine.
Although trichloroethylene exhibits lowtoxicity, its metabolite trichloroethanol, andoxidative degradation products phosgene,COCl2, and chlorine, can cause severe unex pected health hazards. Kawakami andassociates (1988) reported a case of Steven–Johnson syndrome in a worker in a printingfactory. In another case, fire on a stove in ametal-degreasing workplace produced phos gene and chlorine inhalation, which causeddyspnea, fever, and fatigue.
Trichloroethylene exhibited evidence ofcarcinogenicity in laboratory animals. Oraladministration produced liver tumors, whileinhalation caused lung and blood tumors inmice and rats.
Fire Hazard
Special Hazards of Combustion Products: Toxic and irritating gases are produced in fire situations.
Chemical Reactivity
Reactivity with Water No reaction; Reactivity with Common Materials: No reaction; Stability During Transport: Stable; Neutralizing Agents for Acids and Caustics: Not pertinent; Polymerization: Not pertinent; Inhibitor of Polymerization: Not pertinent.
Contact allergens
Trichloroethylene is a chlorinated hydrocarbon used as a detergent or solvent for metals, oils, resins, sulfur, and as general degreasing agent. It can cause irritant contact dermatitis, generalized exanthema, Stevens-Johnson- like syndrome, pustular or bullous eruption, scleroderma, as well as neurological and hepatic disorders.
Potential Exposure
Trichloroethylene is used as a vapor degreaser of metal parts, as a solvent; and as a drug; It is also used for extracting caffeine from coffee, as a dry-cleaning agent; and as a chemical intermediate in the production of pesticides; in making waxes, gums, resins, tars, paints, varnishes, and specific chemicals; such as chloroacetic acid.
Carcinogenicity
Trichloroethylene is reasonably anticipated to be a human carcinogen based on limited evidence of carcinogenicity from studies in humans, sufficient evidence of carcinogenicity from studies in experimental animals, and information from studies on mechanisms of carcinogenesis.
Metabolic pathway
From the photooxidation reaction medium (1) of trichloroethylene, the formation of dichloroacetyl chloride, CO, phosgene, and pentachloroethane and their conversion to the final product, CO2, are identified. By the second TiO2 photocatalyst reaction (2), trichloroacetaldehyde, dichloroacetyl chloride, CO, and phosgene with the new identified intermediates oxalyl chloride, trichloroacetyl chloride, and trichloroacetic acid are observed.
Shipping
UN1710 Trichloroethylene, Hazard Class: 6.1; Labels: 6.1-Poisonous materials.
Purification Methods
Tricloroethylene undergoes decomposition in a similar way as CHCl3, giving HCl, CO, COCl2 and organic products. It reacts with KOH, NaOH and 90% H2SO4, and forms azeotropes with water, MeOH, EtOH, and acetic acid. It is purified by washing successively with 2M HCl, water and 2M K2CO3, then dried with K2CO3 and CaCl2, then fractionally distilled before use. It has also been steam distilled from 10% Ca(OH)2 slurry, most of the water being removed from the distillate by cooling to -30o to -50o and filtering off the ice through chamois skin: the trichloroethylene is then fractionally distilled at 250mm pressure and collected in a blackened container. [Carlisle & Levine Ind Eng Chem (Anal Ed) 24 1164 1932, Beilstein 1 IV 712.]
Incompatibilities
Contact with strong caustics causes decomposition and the production of highly toxic and flammable dichloroacetylene. Violent reaction with chemically active metals; powders, or shavings, such as aluminum, barium, lithium, sodium, magnesium, and titanium. Violent reaction with aluminum in the presence of dilute hydrochloric acid. Thermal decomposition of trichloroethylene, due to contact with hot metal or UV radiation, forms hazardous products including chlorine gas, hydrogen chloride; and phosgene. Keep this chemical away from high temperatures, such as arc welding or cutting, unshielded resistance heating; open flames; and high intensity UV light. Slowly decomposed by light in presence of moisture, with formulation of hydrochloric acid.
Waste Disposal
Consult with environmental regulatory agencies for guidance on acceptable disposal practices. Generators of waste containing this contaminant (≥100 kg/mo) must conform with EPA regulations governing storage, transportation, treatment, and waste disposal. Incineration, preferably after mixing with another combustible fuel. Care must be exercised to assure complete combustion to prevent the formation of phosgene. An acid scrubber is necessary to remove the halo acids produced. An alternative to disposal for TCE is recovery and recycling.
Trichloroethylene Preparation Products And Raw materials
Raw materials
1of2
chevron_rightPreparation Products
1of6
chevron_right| Supplier | Tel | Country | ProdList | Advantage | Inquiry |
|---|---|---|---|---|---|
| Choice Chemicals | +91-9895031268 +91-9895031268 | Kerela, India | 118 | 58 | Inquiry |
| PAARICHEM RESOURCES LLP | +91-8104961021 +91-8104961021 | Maharashtra, India | 82 | 58 | Inquiry |
| JSK Chemicals | +919879767970 | Gujarat, India | 3756 | 58 | Inquiry |
| Ultra Chemical Works | +91-9820078105 +91-9820078105 | Mumbai, India | 124 | 58 | Inquiry |
| DCW Limited | +91-2222871916 +91-2222871914 | Maharashtra, India | 8 | 58 | Inquiry |
| Baron Organics | +91-9825029778 +91-9825029778 | Gujarat, India | 13 | 58 | Inquiry |
| Innovacorp India Pvt Ltd | +91-9312871070 +91-9911981992 | New Delhi, India | 23 | 58 | Inquiry |
| Acuro Organics Limited | +91-8882777000 +91-8882777000 | New Delhi, India | 44 | 58 | Inquiry |
| Lords Chloro Alkali Limited | +91-9871174015 +91-9818832747 | New Delhi, India | 4 | 58 | Inquiry |
| Benzer Multitech India Pvt Ltd | +91-9769550991 +91-9730539955 | Maharashtra, India | 45 | 58 | Inquiry |
| Supplier | Advantage |
|---|---|
| Choice Chemicals | 58 |
| PAARICHEM RESOURCES LLP | 58 |
| JSK Chemicals | 58 |
| Ultra Chemical Works | 58 |
| DCW Limited | 58 |
| Baron Organics | 58 |
| Innovacorp India Pvt Ltd | 58 |
| Acuro Organics Limited | 58 |
| Lords Chloro Alkali Limited | 58 |
| Benzer Multitech India Pvt Ltd | 58 |
Related articles
- Trichloroethylene: Properties, Production process and Uses
- Trichloroethylene is an excellent solvent that can be used as a substitute for benzene and gasoline. It can be used to dissolv....
- Mar 19,2024





