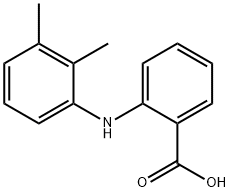
Mefenamic acid
- CAS No.
- 61-68-7
- Chemical Name:
- Mefenamic acid
- Synonyms
- Mefenamic;Mefanamic acid;MEFENAMATE;Ponstel;Ponstan;ac.mefenamico;Mephenamic acid;Mefenamic Acid (200 mg);in-M;HL 1
- CBNumber:
- CB5472051
- Molecular Formula:
- C15H15NO2
- Molecular Weight:
- 241.29
- MOL File:
- 61-68-7.mol
- MSDS File:
- SDS
- Modify Date:
- 2024/7/2 8:55:11
| Melting point | 230 °C |
|---|---|
| Boiling point | 384.06°C (rough estimate) |
| Density | 1.0944 (rough estimate) |
| refractive index | 1.5200 (estimate) |
| storage temp. | 2-8°C |
| solubility | Practically insoluble in water, slightly soluble in ethanol (96 per cent) and in methylene chloride. It dissolves in dilute solutions of alkali hydroxides |
| form | Solid |
| pka | 4.2(at 25℃) |
| color | White to Pale Yellow |
| Water Solubility | It is soluble in acetone, chloroform, dichloromethane, methanol. Insoluble in water. |
| Merck | 14,5798 |
| InChIKey | HYYBABOKPJLUIN-UHFFFAOYSA-N |
| LogP | 5.120 |
| CAS DataBase Reference | 61-68-7(CAS DataBase Reference) |
| NIST Chemistry Reference | Mefenamic acid(61-68-7) |
SAFETY
Risk and Safety Statements
| Symbol(GHS) |  GHS07 |
|||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Signal word | Warning | |||||||||
| Hazard statements | H302 | |||||||||
| Precautionary statements | P301+P312+P330 | |||||||||
| Hazard Codes | Xn | |||||||||
| Risk Statements | 22-40-20/21/22 | |||||||||
| Safety Statements | 22-36 | |||||||||
| WGK Germany | 3 | |||||||||
| RTECS | CB4550000 | |||||||||
| HS Code | 28142000 | |||||||||
| Toxicity | LD50 orally in mice, rats: 630, 790 mg/kg (Jahn, Adrian) | |||||||||
| NFPA 704 |
|
Mefenamic acid price More Price(8)
| Manufacturer | Product number | Product description | CAS number | Packaging | Price | Updated | Buy |
|---|---|---|---|---|---|---|---|
| Sigma-Aldrich(India) | M4267 | Mefenamic acid | 61-68-7 | 50G | ₹6040.35 | 2022-06-14 | Buy |
| Sigma-Aldrich(India) | M4267 | Mefenamic acid | 61-68-7 | 500G | ₹33968.85 | 2022-06-14 | Buy |
| Sigma-Aldrich(India) | 92574 | Mefenamic acid analytical standard | 61-68-7 | 250MG | ₹6365.1 | 2022-06-14 | Buy |
| TCI Chemicals (India) | M1782 | Mefenamic Acid | 61-68-7 | 25G | ₹2500 | 2022-05-26 | Buy |
| TCI Chemicals (India) | M1782 | Mefenamic Acid | 61-68-7 | 100G | ₹9100 | 2022-05-26 | Buy |
Mefenamic acid Chemical Properties,Uses,Production
Chemical Properties
white or light yellow crystalline powder, odorless, insoluble in water, slightly soluble in ethanol, chloroform, slightly soluble in ether. Melting point 230-231°C, mefenamic acid is an anti-inflammatory analgesic with antipyretic, analgesic and anti-inflammatory effects.
Uses
Mefenamic acid is used for the same indications as flufenamic acid. Synonyms for this drug are parkemed, ponstan, ponstel, and others.
Indications
Mefenamic acid (Ponstel) is indicated only for analgesia and primary dysmenorrhea when therapy will not exceed 1 week.
Definition
ChEBI: An aminobenzoic acid that is anthranilic acid in which one of the hydrogens attached to the nitrogen is replaced by a 2,3-dimethylphenyl group. Although classed as a non-steroidal anti-inflammatory drug, its anti-inflammatory properties are considered to b minor. It is used to relieve mild to moderate pain, including headaches, dental pain, osteoarthritis and rheumatoid arthritis.
brand name
Ponstel (Sciele, Parke Davis, USA), Lysalgo (SIT, Italy), Opustan (Opus Pharm, UK), Parkemed (Parke Davis, Germany), Ponstan (Werner-Lambert, Switzerland), Pontal (Sankyo, Japan).
General Description
Mefenamic acid (Ponstel, Ponstan) is one of the oldestNSAIDs, introduced into the market in 1967 for mild tomoderate pain and for primary dysmenorrhea. It is rapidly absorbed with peak plasma levels occurring 2 to 4 hoursafter oral administration. It undergoes hepatic benzylic hydroxylationof its 3'methyl group regioselectively into twoinactive metabolites, 3'-hydroxymethylmefenamic acid andthe 3'carboxylate metabolite (via further oxidation of thebenzylic alcohol group). The parent drugs and these metabolitesare conjugated with glucuronic acid and excreted primarilyin the urine. Thus, although patients with knownliver deficiency may be given lower doses, it is contraindicatedin patients with preexisting renal dysfunction.
Common side effects associated with its use include diarrhea,drowsiness, and headache. The possibility of blood disordershas also prompted limitation of its administration to 7days. It is not recommended for children or during pregnancy.
Clinical Use
Mefenamic acid is synthesized from o-chlorobenzoic acid and 2,3-dimethylaniline under catalytic conditions. Mefenamic acid is the only fenamic acid derivative that produces analgesia centrally and peripherally. Mefenamic acid is indicated for the short-term relief of moderate pain and for primary dysmenorrhea.
Safety
Mefenamic acid has mild anti-inflammatory properties and is used primarily as a short-term analgesic. Gastrointestinal disturbances, including possibly allergic diarrhea and potential renal toxicity, limit its use.
Synthesis
Mefenamic acid, N-(2,3-xylyl)anthranylic acid (3.2.19), is synthesized
in basically the same manner, by the reaction of the potassium salt of 2-bromobenzoic acid
with 2,3-dimethylaniline in the presence of copper (II) acetate [80,81].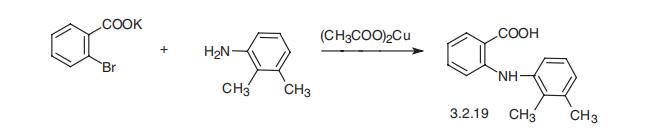
Synthesis 2: mefenamic acid is prepared via
the Jourdan – Ullmann – Goldberg synthesis utilizing either anthranilic acid and 3-bromo-1,2-
dimethylbenzene or 2,3-dimethylaniline and an
o-halobenzoic acid in the presence of a copper
catalyst and a proton acceptor.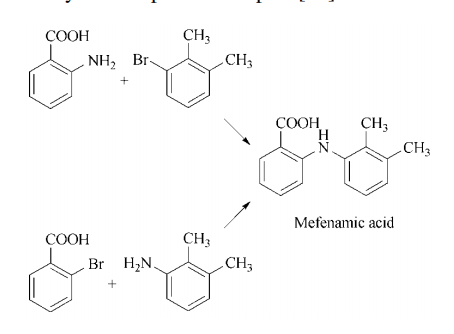
Metabolism
Mefenamic acid is absorbed rapidly following oral administration, with peak plasma levels being attained within 2 to 4 hours. It is highly bound to plasma proteins (78.5%) and has a plasma half-life of 2 to 4 hours. Metabolism occurs through regioselective oxidation of the 3′-methyl group and glucuronidation of mefenamic acid and its metabolites. Urinary excretion accounts for approximately 50 to 55% of an administered dose, with unchanged drug accounting for 6%, the 3′-hydroxymethyl metabolite (primarily as the glucuronide) accounting for 25%, and the remaining 20% as the dicarboxylic acid (of which 30% is the glucuronide conjugate). These metabolites are essentially inactive.
Mefenamic acid Preparation Products And Raw materials
| Supplier | Tel | Country | ProdList | Advantage | Inquiry |
|---|---|---|---|---|---|
| THE MOLECULEZ | +91-7506703683 +91-7506703683 | Maharashtra, India | 79 | 58 | Inquiry |
| SNECOFRi Pvt Ltd | +91-9032850129 +91-9032850129 | Telangana, India | 404 | 58 | Inquiry |
| AARTIA KEM SCIENCE | +91-8291072530 +91-8291072530 | Maharashtra, India | 70 | 58 | Inquiry |
| GLP Pharma Standards | +91 9866074638 | Hyderabad, India | 1644 | 58 | Inquiry |
| Gonane Pharma | +91-9819380043 +91-9819380043 | NaviMumbai, India | 192 | 58 | Inquiry |
| Medi Pharma Drug House | +919930911911 | Mumbai, India | 143 | 58 | Inquiry |
| Shree Chemopharma Ankleshwar Pvt., Ltd. | +91-9377992523 +91-9909242468 | Gujarat, India | 12 | 58 | Inquiry |
| Varahi International | +91-7922844991 +91-7922844991 | Gujarat, India | 24 | 58 | Inquiry |
| Acute Research | +91-9825068697 +91-9825068697 | Gujarat, India | 43 | 58 | Inquiry |
| Wanbury Limited | +91-2271963222 +91-2267942222 | Maharashtra, India | 17 | 58 | Inquiry |
| Supplier | Advantage |
|---|---|
| THE MOLECULEZ | 58 |
| SNECOFRi Pvt Ltd | 58 |
| AARTIA KEM SCIENCE | 58 |
| GLP Pharma Standards | 58 |
| Gonane Pharma | 58 |
| Medi Pharma Drug House | 58 |
| Shree Chemopharma Ankleshwar Pvt., Ltd. | 58 |
| Varahi International | 58 |
| Acute Research | 58 |
| Wanbury Limited | 58 |
61-68-7(Mefenamic acid )Related Search:
1of4
chevron_right




