Cupric hydroxide
- CAS No.
- 20427-59-2
- Chemical Name:
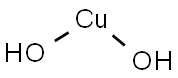
- Cupric hydroxide
- Synonyms
- COPPER HYDROXIDE;Cu(OH)2;COPPER(II) HYDROXIDE;Kocide;Parasol;COPPER HYDRATE;Cooper Hydroxide;copper dihydroxide;Comac;cuzin
- CBNumber:
- CB7455575
- Molecular Formula:
- CuH2O2
- Molecular Weight:
- 97.56
- MOL File:
- 20427-59-2.mol
- MSDS File:
- SDS
- Modify Date:
- 2024/5/23 18:06:45
| Density | 3.4 |
|---|---|
| vapor pressure | 0Pa at 25℃ |
| storage temp. | Inert atmosphere,2-8°C |
| solubility | H2O: insoluble(lit.) |
| form | Powder |
| color | Blue |
| PH | 7.69(1 mM solution);7.69(10 mM solution);7.69(100 mM solution) |
| Water Solubility | Slightly soluble in waterSoluble in acids, ammonium hydroxide, dilute hydrochloric acid, concentrated alkali and potassium cyanide. Insoluble in water, ethanol, and acetone. |
| Sensitive | Hygroscopic |
| Merck | 14,2642 |
| Solubility Product Constant (Ksp) | pKsp: 19.66 |
| Exposure limits |
ACGIH: TWA 1 mg/m3 NIOSH: IDLH 100 mg/m3; TWA 1 mg/m3 |
| Stability | Stable, but hygroscopic. Store in dry conditions. |
| CAS DataBase Reference | 20427-59-2(CAS DataBase Reference) |
| NIST Chemistry Reference | Copper hydroxide(20427-59-2) |
| EPA Substance Registry System | Cupric hydroxide (20427-59-2) |
SAFETY
Risk and Safety Statements
| Symbol(GHS) |    GHS05,GHS06,GHS09 |
|||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Signal word | Danger | |||||||||
| Hazard statements | H302-H318-H330-H410 | |||||||||
| Precautionary statements | P260-P273-P280-P301+P312-P304+P340+P310-P305+P351+P338 | |||||||||
| Hazard Codes | Xn,N,T | |||||||||
| Risk Statements | 22-37/38-41-50/53-23 | |||||||||
| Safety Statements | 26-36-39 | |||||||||
| RIDADR | 3262 | |||||||||
| WGK Germany | 3 | |||||||||
| RTECS | GL7600000 | |||||||||
| TSCA | Yes | |||||||||
| HS Code | 28255000 | |||||||||
| NFPA 704 |
|
Cupric hydroxide price More Price(2)
Cupric hydroxide Chemical Properties,Uses,Production
Chemical Properties
Blue powder.Soluble in acids and ammonium hydroxide; insoluble in water.
Uses
In manufacture of rayon, battery electrodes, other Cu salts; as mordant in dyeing; as pigment; in fungicides, insecticides; as feed additive; in treating and staining paper; in preparation of Schweitzer's reagent; in catalysts.
General Description
The orthorhombic nature of copper hydroxide crystals was determined by X ray diffraction. Copper hydroxide can act as a heterogeneous catalyst in the selective oxidative cross coupling of terminal alkynes to yield their corresponding ynamides.
Hazard
Toxic by ingestion and inhalation.
Potential Exposure
Inorganic copper fungicide, nematicide, and microbiocide
Shipping
UN3077 Environmentally hazardous substances, solid, n.o.s., Hazard class: 9; Labels: 9-Miscellaneous hazardous material, Technical Name Required.
Incompatibilities
Reacts with calcium (metal hydroxides), nitroethane, nitromethane, 1-nitropropane, zirconium
Waste Disposal
Copper-containing soluble wastes can be concentrated through the use of ion exchange, reverse osmosis, or evaporators to the point where copper can be electrolytically removed and sent to a reclaiming firm. If recovery is not feasible, the copper can be precipitated through the use of caustics and the sludge deposited in a chemical waste landfill Copper-containing wastes can be concentrated through the use of ion exchange, reverse osmosis, or evaporators to the point where copper can be electrolytically removed and sent to a reclaiming firm. Details of copper recovery from a variety of industrial wastes have been published. If recovery is not feasible, the copper can be precipitated by the use of caustics and the sludge deposited in a chemical waste landfill. Recommendable methods: Precipitation, solidification, landfill, discharge to sewer, & incineration. Peer-review: Precipitate copper with alkali, filter, solidify precipitate. (Do not use ammonia as alkali). Cation exchange will allow recovery of copper. Eluate from cation exchanger can be passed through anion exchanger to remove (or reduce) naphthenic acid content. Exhausted ion exchange resins can be landfilled. (Peer-review conclusions of an IRPTC expert consultation)
Cupric hydroxide Preparation Products And Raw materials
Raw materials
Preparation Products
| Supplier | Tel | Country | ProdList | Advantage | Inquiry |
|---|---|---|---|---|---|
| Shyam Chemicals Pvt Ltd | +91-9820028842 +91-9820028842 | Mumbai, India | 10 | 58 | Inquiry |
| Kundan Pestichem Pvt Ltd | +91-9825801520 +91-7929706677 | Gujarat, India | 5 | 58 | Inquiry |
| Parikh Enterprises Pvt Ltd | +91-7922871471 +91-9925000571 | Gujarat, India | 10 | 58 | Inquiry |
| Vimal Crop Care Pvt. Ltd | +91-079-22821390 +91-8511174812 | Gujarat, India | 2 | 58 | Inquiry |
| CLEARSYNTH LABS LTD. | +91-22-45045900 | Hyderabad, India | 6351 | 58 | Inquiry |
| S.K. CHEMICAL INDUSTRIES | +91-22-66370299 Ext 21 / 91-22-23437380 Ext 25 | New Delhi, India | 262 | 30 | Inquiry |
| Nanochemazone Inc. | 08048616195 | Haryana, India | 79 | 58 | Inquiry |
| Manglam Chemtech | 08048980268 | Vadodara, India | 3 | 58 | Inquiry |
| Todini Metals And Chemicals India Pvt Ltd | 08048952331 | Mumbai, India | 39 | 58 | Inquiry |
| PK Chlorochem Private Limited | 08048372472Ext 860 | Vadodara, India | 109 | 58 | Inquiry |
20427-59-2(Cupric hydroxide)Related Search:
1of4
chevron_right




