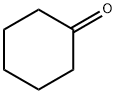
Cyclohexanone
- CAS No.
- 108-94-1
- Chemical Name:
- Cyclohexanone
- Synonyms
- HEXANONE;Anon;ANONE;Sextone;Hexanon;Cykloheksanon;CYCLOHEXANONE REAGENT (ACS);CYCLOHEXANONE, REAGENT (ACS)CYCLOHEXANONE, REAGENT (ACS)CYCLOHEXANONE, REAGENT (ACS);Nadone;hytrolo
- CBNumber:
- CB8347660
- Molecular Formula:
- C6H10O
- Molecular Weight:
- 98.14
- MOL File:
- 108-94-1.mol
- MSDS File:
- SDS
- Modify Date:
- 2024/7/2 14:09:54
| Melting point | -47 °C (lit.) |
|---|---|
| Boiling point | 155 °C (lit.) |
| Density | 0.947 g/mL at 25 °C (lit.) |
| vapor density | 3.4 (vs air) |
| vapor pressure | 3.4 mm Hg ( 20 °C) |
| refractive index |
n |
| FEMA | 3909 | CYCLOHEXANONE |
| Flash point | 116 °F |
| storage temp. | Store at +5°C to +30°C. |
| solubility | 90g/l |
| pka | 17(at 25℃) |
| form | Liquid |
| color | APHA: ≤10 |
| Relative polarity | 0.281 |
| PH | 7 (70g/l, H2O, 20℃) |
| Odor | Like peppermint and acetone. |
| explosive limit | 1.1%, 100°F |
| Odor Type | minty |
| Water Solubility | 150 g/L (10 ºC) |
| Merck | 14,2726 |
| JECFA Number | 1100 |
| BRN | 385735 |
| Henry's Law Constant | 1.2 x 10-5 atm?m3/mol at 25 °C (Hawthorne et al., 1985) 6.92 x 10-5 atm?m3/mol at 60.00 °C, 10.7 at 70.00 °C, 16.4 at 80.00 °C (headspace-GC, Hovorka et al., 2002) |
| Exposure limits | TLV-TWA 100 mg/m3 (25 ppm) (ACGIH); IDLH 5000 ppm (NIOSH). |
| Dielectric constant | 18.2(20℃) |
| Stability | Stable. Combustible. Incompatible with strong oxidizing agents. |
| LogP | 0.86 at 25℃ |
| CAS DataBase Reference | 108-94-1(CAS DataBase Reference) |
| IARC | 3 (Vol. 47, 71) 1999 |
| NIST Chemistry Reference | Cyclohexanone(108-94-1) |
| EPA Substance Registry System | Cyclohexanone (108-94-1) |
SAFETY
Risk and Safety Statements
| Symbol(GHS) |    GHS02,GHS05,GHS07 |
|||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Signal word | Danger | |||||||||
| Hazard statements | H226-H302+H312+H332-H315-H318 | |||||||||
| Precautionary statements | P210-P280-P301+P312-P303+P361+P353-P304+P340+P312-P305+P351+P338 | |||||||||
| Hazard Codes | Xn | |||||||||
| Risk Statements | 10-20-41-38-20/21/22 | |||||||||
| Safety Statements | 25-36/37/39-26 | |||||||||
| RIDADR | UN 1915 3/PG 3 | |||||||||
| OEB | A | |||||||||
| OEL | TWA: 25 ppm (100 mg/m3) [skin] | |||||||||
| WGK Germany | 1 | |||||||||
| RTECS | GW1050000 | |||||||||
| Autoignition Temperature | 788 °F | |||||||||
| TSCA | Yes | |||||||||
| HS Code | 2914 22 00 | |||||||||
| HazardClass | 3 | |||||||||
| PackingGroup | III | |||||||||
| Toxicity | LD50 orally in rats: 1.62 ml/kg (Smyth) | |||||||||
| IDLA | 700 ppm | |||||||||
| NFPA 704 |
|
Cyclohexanone price More Price(45)
| Manufacturer | Product number | Product description | CAS number | Packaging | Price | Updated | Buy |
|---|---|---|---|---|---|---|---|
| Sigma-Aldrich(India) | W390909 | Cyclohexanone 99.8% | 108-94-1 | 1SAMPLE | ₹5141.88 | 2022-06-14 | Buy |
| Sigma-Aldrich(India) | W390909 | Cyclohexanone 99.8% | 108-94-1 | 1KG | ₹6852.23 | 2022-06-14 | Buy |
| Sigma-Aldrich(India) | C102180 | Cyclohexanone ReagentPlus?, 99.8% | 108-94-1 | 25ML | ₹2619.65 | 2022-06-14 | Buy |
| Sigma-Aldrich(India) | W390909 | Cyclohexanone 99.8% | 108-94-1 | 25KG | ₹44155.18 | 2022-06-14 | Buy |
| Sigma-Aldrich(India) | C102180 | Cyclohexanone ReagentPlus?, 99.8% | 108-94-1 | 1L | ₹3247.5 | 2022-06-14 | Buy |
Cyclohexanone Chemical Properties,Uses,Production
Description
Cyclohexanone, a colorless liquid is a cyclic ketone. It is an important building block for the synthesis of a variety of organic compounds. Majority of the cyclohexanone synthesized is utilized as an intermediate in the synthesis of nylon.
Used as a polyvinyl chloride (PVC) solvent, cyclohexanone caused contact dermatitis in a woman manufacturing PVC fluidotherapy bags. Cyclohexanone probably does not cross react with cyclohexanone resin. A cyclohexanone-derived resin used in paints and varnishes caused contact dermatitis in painters.
Chemical Properties
Cyclohexanone is a water-white to slightly yellow liquid with a peppermint-like or acetone-like odor. The Odor Threshold is 0.12 0.24 ppm in air.
Physical properties
Clear, colorless to pale yellow, oily liquid with a peppermint-like odor. Experimentally determined detection and recognition odor threshold concentrations were identical: 480 μg/m3 (120 ppmv) (Hellman and Small, 1974).
Occurrence
Reported present in Cistus labdaniferus.
Uses
Cyclohexanone is used in the productionof adipic acid for making nylon; in thepreparation of cyclohexanone resins; and asa solvent for nitrocellulose, cellulose acetate,resins, fats, waxes, shellac, rubber, and DDT..
Definition
ChEBI: A cyclic ketone that consists of cyclohexane bearing a single oxo substituent.
General Description
A colorless to pale yellow liquid with a pleasant odor. Less dense than water . Flash point 111°F. Vapors heavier than air. Used to make nylon, as a chemical reaction medium, and as a solvent.
Air & Water Reactions
Flammable. Soluble in water.
Reactivity Profile
Cyclohexanone forms an explosive peroxide with H2O2, and reacts vigorously with oxidizing materials (nitric acid).
Health Hazard
The toxicity of cyclohexanone in test specieswas found to be low to moderate. Exposureto its vapors can produce irritation in the eyesand throat. Splashing into the eyes can damagethe cornea. Throat irritation in humansmay occur from 3–5 minute exposure to a50-ppm concentration in air. The symptomsof chronic toxicity in animals from its inhalationwere liver and kidney damage, as wellas weight loss. However, its acute toxicitywas low below 3000 ppm. The symptomsin guinea pigs were lacrimation, salivation,lowering of heart rate, and narcosis. Exposureto 4000 ppm for 4–6 hours was lethalto rats and guinea pigs.
The oral toxicity of this compound waslow. Ingestion may cause narcosis and depressionof the central nervous system. It canbe absorbed through the skin.
LD50 value, dermal (rabbits): 1000 mg/kg
LD50 value, intraperitoneal (rats): 1130 mg/kg.
Fire Hazard
HIGHLY FLAMMABLE: Will be easily ignited by heat, sparks or flames. Vapors may form explosive mixtures with air. Vapors may travel to source of ignition and flash back. Most vapors are heavier than air. They will spread along ground and collect in low or confined areas (sewers, basements, tanks). Vapor explosion hazard indoors, outdoors or in sewers. Runoff to sewer may create fire or explosion hazard. Containers may explode when heated. Many liquids are lighter than water.
Chemical Reactivity
Reactivity with Water No reaction; Reactivity with Common Materials: No reaction; Stability During Transport: Stable; Neutralizing Agents for Acids and Caustics: Not pertinent; Polymerization: Not pertinent; Inhibitor of Polymerization: Not pertinent.
Contact allergens
Used as a polyvinyl chloride solvent, cyclohexanone caused contact dermatitis in a woman manufacturing PVC fluidotherapy bags. Cyclohexanone probably does not cross-react with cyclohexanone resin. A cyclohexanone-derived resin used in paints and varnishes caused contact dermatitis in painters
Safety Profile
Suspected carcinogen. Moderately toxic by ingestion, inhalation, subcutaneous, intravenous, and intraperitoneal routes. A skin and severe eye irritant. Human systemic effects by inhalation: changes in the sense of smell, conjunctiva irritation, and unspecified respiratory system changes. Human irritant by inhalation. Mdd narcotic properties have also been ascribed to it. Human mutation data reported. Experimental reproductive effects. Flammable liquid when exposed to heat or flame; can react vigorously with oxidizing materials. Slight explosion hazard in its vapor form, when exposed to flame.Reaction with hydrogen peroxide + nitric acid forms an explosive peroxide. To fight fire, use alcohol foam, dry chemical, or COa. When heated to decomposition it emits acrid smoke and irritating fumes. See also KETONES and CYCLOHEXANE.
Potential Exposure
May form explosive mixture with air. Contact with oxidizing agents or nitric acid may cause a violent reaction. Do not use brass, copper, bronze, or lead fittings. Attacks many coatings and plastic materials.
Carcinogenicity
IARC considers the animal data for cyclohexanone as inadequate evidence of carcinogenicity and listed cyclohexanone as not classifiable for carcinogenicity (IARC Category 3).
Environmental Fate
Biological. In activated sludge inoculum, 96.0% COD removal was achieved. The average rate
of biodegradation was 30.0 mg COD/g?h (Pitter, 1976).
Photolytic. Atkinson (1985) reported an estimated photooxidation rate constant of 1.56 x 10-11
cm3/molecule?sec for the reaction of cyclohexanone and OH radicals in the atmosphere at 298 K.
Chemical/Physical. Cyclohexanone will not hydrolyze because it has no hydrolyzable
functional group.
At an influent concentration of 1,000 mg/L, treatment with GAC resulted in effluent
concentration of 332 mg/L. The adsorbability of the carbon used was 134 mg/g carbon (Guisti et
al., 1974). Similarly, at influent concentrations of 10, 1.0, 0.1, and 0.01 mg/L, the GAC adsorption
capacities were 36, 6.2, 1.1, and 0.19 mg/g, respectively (Dobbs and Cohen, 1980).
Shipping
UN1915 Cyclohexanone, Hazard Class: 3; Labels: 3-Flammable liquid.
Purification Methods
Dry cyclohexanone with MgSO4,CaSO4, Na2SO4 or Linde type 13X molecular sieves, then distil it. Cyclohexanol and other oxidisable impurities can be removed by treatment with chromic acid or dilute KMnO4. More thorough purification is possible by conversion to the bisulfite addition compound, or the semicarbazone, followed by decomposition with Na2CO3 and steam distillation. [For example, equal weights of the bisulfite adduct (crystallised from water) and Na2CO3 are dissolved in hot water and, after steam distillation, the distillate is saturated with NaCl and extracted with Et2O which is then dried (anhydrous MgSO4 or Na2SO4), filtered and the solvent evaporated prior to further distillation.] FLAMMABLE [Beilstein 7 III 14, 7 IV 15.]
Incompatibilities
May form explosive mixture with air. Contact with oxidizing agents or nitric acid may cause a violent reaction. Do not use brass, copper, bronze, or lead fittings. Attacks many coatings and plastic materials.
Waste Disposal
Dissolve or mix the material with a combustible solvent and burn in a chemical incinera- tor equipped with an afterburner and scrubber. All federal, state, and local environmental regulations must be observed.
Cyclohexanone Preparation Products And Raw materials
Raw materials
1of3
chevron_rightPreparation Products
1of8
chevron_right| Supplier | Tel | Country | ProdList | Advantage | Inquiry |
|---|---|---|---|---|---|
| Soham Chemical Industries | +91-7016081644 +91-7016081644 | Mumbai, India | 83 | 58 | Inquiry |
| SHIVAM INDUSTRIES | +91-9820823043 +91-9820823043 | Mumbai, India | 30 | 58 | Inquiry |
| Choice Chemicals | +91-9895031268 +91-9895031268 | Kerela, India | 118 | 58 | Inquiry |
| JSK Chemicals | +919879767970 | Gujarat, India | 3756 | 58 | Inquiry |
| Gujarat State Fertilizers And Chemicals Ltd | +91-2652242451 +91-9879275045 | Gujarat, India | 21 | 58 | Inquiry |
| LANXESS India Pvt. Ltd. | +91-8356878763 +91-8356878763 | Maharashtra, India | 190 | 58 | Inquiry |
| Punjab Chemicals & Crop Protection Limited | +91 (22) 2674-7900 | New Delhi, India | 125 | 0 | Inquiry |
| TCI Chemicals (India) Pvt. Ltd. | 1800 425 7889 | New Delhi, India | 6778 | 58 | Inquiry |
| Otto Chemie Pvt. Ltd. | +91 9820041841 | Mumbai, India | 5873 | 58 | Inquiry |
| Alfa Aesar | 1 800 209 7001 | Maharashtra, India | 6913 | 58 | Inquiry |
Related articles
- Cyclohexanone: Chemical and physical properties, Production and Uses
- Cyclohexanone is a six-carbon ring molecule with a ketone functional group. It is a colourless to pale yellow oily liquid with....
- Apr 26,2024
- Cyclohexanone: Toxicity and Safety
- The passage introduces the toxicity and safety of Cyclohexanone.
- Nov 21,2022
- A pharmaceutical intermediate:Cyclohexanone
- Cyclohexanone appears as a colorless to pale yellow liquid with a pleasant odor. Less dense than water . Flash point 111°F. Va....
- Mar 16,2022
108-94-1(Cyclohexanone)Related Search:
1of4
chevron_right




