Beraprost sodium
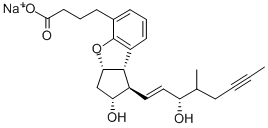
- CAS No.
- 88475-69-8
- Chemical Name:
- Beraprost sodium
- Synonyms
- BERAPROST SODIUM SALT;Dorner;ML 1129;TRK 100;Berasus;Careload;PROCYLIN;Procyclin;MDL 201129;Befraprost
- CBNumber:
- CB8761736
- Molecular Formula:
- C24H29NaO5
- Molecular Weight:
- 420.47
- MOL File:
- 88475-69-8.mol
- MSDS File:
- SDS
- Modify Date:
- 2024/5/14 10:14:29
| Melting point | 204-206°C |
|---|---|
| storage temp. | -20°C |
| solubility |
DMF:77.0(Max Conc. mg/mL);183.12(Max Conc. mM) Ethanol:20.0(Max Conc. mg/mL);47.56(Max Conc. mM) PBS (pH 7.2):19.0(Max Conc. mg/mL);45.19(Max Conc. mM) |
| form | powder |
| color | white to beige |
| InChIKey | YTCZZXIRLARSET-LIIFYGAANA-M |
| SMILES | [C@H]12[C@H](C[C@@H](O)[C@@H]1/C=C/[C@@H](O)C(C)CC#CC)OC1C(=CC=CC2=1)CCCC([O-])=O.[Na+] |&1:0,1,3,5,8,r| |
| CAS DataBase Reference | 88475-69-8(CAS DataBase Reference) |
SAFETY
Risk and Safety Statements
| Symbol(GHS) |  GHS07 |
|||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Signal word | Warning | |||||||||
| Hazard statements | H315-H319-H335 | |||||||||
| Precautionary statements | P261-P264-P271-P280-P302+P352-P305+P351+P338 | |||||||||
| NFPA 704 |
|
Beraprost sodium price More Price(2)
Beraprost sodium Chemical Properties,Uses,Production
Description
Beraprost sodium is a new, orally active antithrombotic epoprostenol analog introduced as a treatment for peripheral vascular disease, including Raynaud's syndrome and Buerger's disease. The antiplatelet activity may be due to its elevation of cyclic AMP in platelets which is achieved by activating adenylate cyclase in the platelet membrane, followed by the inhibition ofCa++- influx and thromboxane A2 formation.
Chemical Properties
White Solid
Uses
Platelet aggregation inhibitor; stable analog of Prostacyclin. Antithrombotic; vasodilator (peripheral).
General Description
Beraprost sodium is an orally active prostacyclin analog.
Side effects
Disease-related events had similar incidence in patients receiving beraprost and those receiving placebo. Drug-related adverse events like headache, flushing, jaw pain, and diarrhea were more common in patients treated with beraprost sodium and occurred mostly during the six-week titration period. The incidence was markedly reduced in the maintenance period. Six patients (9%) in the beraprost sodium group and two (3%) in the placebo group withdrew prematurely from the study because of adverse events. No clinically adverse changes in hematologic or biochemical variables were seen in the beraprost sodium group.
Beraprost sodium Preparation Products And Raw materials
Raw materials
Preparation Products
| Supplier | Tel | Country | ProdList | Advantage | Inquiry |
|---|---|---|---|---|---|
| CLEARSYNTH LABS LTD. | +91-22-45045900 | Hyderabad, India | 6351 | 58 | Inquiry |
| Pharmaffiliates Analytics and Synthetics P. Ltd | +91-172-5066494 | Haryana, India | 6773 | 58 | Inquiry |
| Pharma Affiliates | 172-5066494 | Haryana, India | 6761 | 58 | Inquiry |
| Beijing Hope Pharmaceutical Co., Ltd. | +86-010-67886402 +8613611125266 | China | 71 | 58 | Inquiry |
| Hebei Weimiao Import and Export Trade Co., Ltd. | +undefined19948166995 | China | 73 | 58 | Inquiry |
| Shanghai Getian Industrial Co., LTD | +86-15373193816 +86-15373193816 | China | 269 | 58 | Inquiry |
| Henan Tianfu Chemical Co.,Ltd. | +86-0371-55170693 +86-19937530512 | China | 21670 | 55 | Inquiry |
| Hangzhou FandaChem Co.,Ltd. | 008657128800458; +8615858145714 | China | 9337 | 55 | Inquiry |
| career henan chemical co | +86-0371-86658258 +8613203830695 | China | 29897 | 58 | Inquiry |
| BOC Sciences | +1-631-485-4226 | United States | 19553 | 58 | Inquiry |
88475-69-8(Beraprost sodium)Related Search:
1of4
chevron_right




