Thionyl chloride
- CAS No.
- 7719-09-7
- Chemical Name:
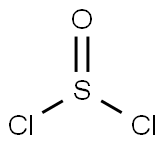
- Thionyl chloride
- Synonyms
- SOCl2;Sulfurous dichloride;ThionyL;THIONYL DICHLORIDE;Thionylchlorid;Sulfinyl chloride;SULFUROUS OXYCHLORIDE;Sulfinyl dichloride;THIONYL CHLORIDEGC STANDARD;Thionyl chloride, 1M soln. in dichloroMethane
- CBNumber:
- CB9190771
- Molecular Formula:
- Cl2OS
- Molecular Weight:
- 118.97
- MOL File:
- 7719-09-7.mol
- MSDS File:
- SDS
- Modify Date:
- 2024/5/28 19:59:05
| Melting point | -105 °C |
|---|---|
| Boiling point | 79 °C(lit.) |
| Density | 1.64 g/mL at 20 °C |
| vapor density | 4.1 (vs air) |
| vapor pressure | 97 mm Hg ( 20 °C) |
| refractive index |
n |
| Flash point | 105°C |
| storage temp. | Store at RT. |
| solubility | Miscible with toluene, chloroform, benzene, carbon tetrachloride and diethyl ether. |
| form | Liquid |
| color | ≤50(APHA) |
| Odor | Characteristic, pungent odor |
| Water Solubility | REACTS |
| Sensitive | Moisture Sensitive |
| Merck | 14,9348 |
| Exposure limits |
ACGIH: TWA 50 ppm OSHA: TWA 25 ppm; STEL 125 ppm NIOSH: IDLH 2300 ppm |
| Dielectric constant | 9.1(22℃) |
| Stability | Reacts violently with water. Incompatible with most common metals, strong reducing agents, strong bases, alcohols, amines. |
| CAS DataBase Reference | 7719-09-7(CAS DataBase Reference) |
| NIST Chemistry Reference | Thionyl chloride(7719-09-7) |
| EPA Substance Registry System | Thionyl chloride (7719-09-7) |
SAFETY
Risk and Safety Statements
| Symbol(GHS) |   GHS05,GHS06 |
|||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Signal word | Danger | |||||||||
| Hazard statements | H302-H314-H331-H335 | |||||||||
| Precautionary statements | P261-P280-P301+P312-P303+P361+P353-P304+P340+P310-P305+P351+P338 | |||||||||
| Hazard Codes | C | |||||||||
| Risk Statements | 14-20/22-29-35-40-34-20/21/22 | |||||||||
| Safety Statements | 26-36/37/39-45-28-27 | |||||||||
| RIDADR | UN 1836 8/PG 1 | |||||||||
| OEL | Ceiling: 1 ppm (5 mg/m3) | |||||||||
| WGK Germany | 1 | |||||||||
| RTECS | XM5150000 | |||||||||
| F | 19 | |||||||||
| TSCA | Yes | |||||||||
| HazardClass | 8 | |||||||||
| PackingGroup | I | |||||||||
| HS Code | 28121095 | |||||||||
| NFPA 704 |
|
Thionyl chloride Chemical Properties,Uses,Production
Chemical Properties
Thionyl chloride is a pale yellow to reddish liquid. Suffocating odor like sulfur dioxide. Fumes form when exposed to moist air.
Physical properties
Pale yellow to red fuming liquid; suffocating odor; refractive index 1.517 at 20°C; density 1.631 g/mL at 20°C; freezes at -101°C; boils at 75.6°C; decomposes at 140°C; decomposes in water; soluble in benzene, chloroform, and carbon tetrachloride.
Uses
Thionyl chloride (SOCl2) is used as a chlorinating agent in manufacturing organic compounds. It is also used as a solvent in high-energy lithium batteries.
Preparation
Sulfurous oxychloride can be prepared by oxidation of sulfur dichloride with sulfur trioxide: SCl2 + SO3 → SOCl2 + SO2
Also, the compound can be prepared by reacting sulfur dioxide with phosphorus pentachloride: SO2 + PCl5 → SOCl2 + POCl3.
Definition
ChEBI: Thionyl chloride is a sulfinyl halide in which both of the halide atoms are chorines. It is a sulfinyl halide and a chlorine molecular entity.
General Description
Thionyl chloride appears as a colorless to yellow fuming liquid with a suffocating pungent odor. Boiling point 79 °C. A lachrymator. Highly corrosive and toxic. Long-term inhalation of low concentrations or short-term inhalation of high concentrations has adverse health effects.
Reactivity Profile
Thionyl chloride reacts, potentially explosively, with dimethyl sulfoxide or dimethylformamide containing traces of iron or zinc [Spitulnik, M. J., Chem. Eng. News, 1977, 55(31), p. 31]. Undergoes violent reactions with bases (ammonia, sodium hydroxide, potassium hydroxide, amines), alkali metals (sodium, potassium), esters (ethyl acetate), toluene mixed with ethanol / water [Bretherick, 5th ed., 1995, p. 1325]. Has an expansion ratio from gas to liquid of nearly 1000:1. Hence may cause an explosion if heated while contained [MCA Case History No. 1808]. May react vigorously or explosively if mixed with diisopropyl ether or other ethers in the presence of trace amounts of metal salts [J. Haz. Mat., 1981, 4, 291]. Perchloric acid ignites on contact with sulfinyl chloride. (Bailar, 1973, Vol. 2, 1442). SOCl2 reacts with esters, such as ethyl acetate, forming toxic SO2 gas and water soluble/toxic acyl chlorides, catalyzed by Fe or Zn (Spagnuolo, C.J. et al. 1992. Chemical and Engineering News 70(22):2.).
Hazard
Strong irritant to skin, tissue, and upper respiratory tract.
Health Hazard
CORROSIVE and/or TOXIC; inhalation, ingestion or contact (skin, eyes) with vapors, dusts or substance may cause severe injury, burns or death. Fire will produce irritating, corrosive and/or toxic gases. Reaction with water may generate much heat that will increase the concentration of fumes in the air. Contact with molten substance may cause severe burns to skin and eyes. Runoff from fire control or dilution water may cause pollution.
Fire Hazard
EXCEPT FOR ACETIC ANHYDRIDE (UN1715), THAT IS FLAMMABLE, some of these materials may burn, but none ignite readily. May ignite combustibles (wood, paper, oil, clothing, etc.). Substance will react with water (some violently), releasing corrosive and/or toxic gases and runoff. Flammable/toxic gases may accumulate in confined areas (basement, tanks, hopper/tank cars, etc.). Contact with metals may evolve flammable hydrogen gas. Containers may explode when heated or if contaminated with water. Substance may be transported in a molten form.
Safety Profile
Moderately toxic by inhalation. The material itself is more toxic than sulfur dioxide. Has a pungent odor similar to that of sulfur dioxide; it fumes upon exposure to air. Violent reaction with water releases hydrogen chloride and sulfur dioxide. Both these decomposition products constitute serious toxicity hazards. A corrosive irritant that causes burns to the skin and eyes. A powerful chlorinating agent. Potentially explosive reaction with ammonia, bis(dimethy1amino)sulfoxide - (above 80℃), chloryl perchlorate, 1,2,3- cyclohexanetrione trioxime + sulfur dioxide, dimethyl sulfoxide, hexafluoroisopropylideneaminolithium. Violent reaction or ignition with 2,4-hexadiyn-1-6-di01, onitrobenzoyl acetic acid, o-nitrophenylacetic acid, sodum (ignites at 300℃). Incompatible with ammonia, dimethyl formamide + trace iron or zinc, linseed oil + quinoline, toluene + ethanol + water. When heated to decomposition it emits toxic fumes of SOx and Cl-. See also HYDROGEN CHLORIDE and SULFUR DIOXIDE.
Potential Exposure
Thionyl chloride is used as specialty chlorinating agent, particularly in preparation of organic acid chlorides; in organic synthesis; as a catalyst.
Shipping
UN1836 Thionyl chloride, Hazard class: 8; Labels: 8-Corrosive material.
Purification Methods
Crude SOCl2 can be freed from sulfuryl chloride, sulfur monochloride and sulfur dichloride by refluxing it with sulfur and then fractionally distilling twice. [The SOCl2 is converted to SO2 and sulfur chlorides. The S2Cl2 (b 135.6o) is left in the residue, whereas SCl2 (b 59o) passes over in the forerun.] The usual purification is to distil it from quinoline (50g SOCl2 to 10g quinoline) to remove acid impurities, followed by distillation from boiled linseed oil (50g SOCl2 to 20g of oil). Precautions must be taken to exclude moisture. Thionyl chloride is used extensively in organic syntheses and can be prepared by distillation of technical SOCl2 in the presence of diterpene (12g/250mL SOCl2), and avoiding overheating. Further purification is achieved by redistillation from linseed oil (1-2%) [Rigby Chem Ind (London) 1508 1969]. Gas chromatographically pure material is obtained by distillation from 10% (w/w) triphenyl phosphite [Friedman & Wetter J Chem Soc (A) 36 1967, Larsen et al. J Am Chem Soc 108 6950 1986]. HARMFUL VAPOURS.
Incompatibilities
Reacts violently with water releasing sulfur dioxide and hydrogen chloride. Keep away from water, acids, alcohols, alkalis, ammonia, chloryl perchlorate.
Waste Disposal
Spray on a thick layer of a (1:1) mixture of dry soda ash and slaked lime behind a shield. After mixing, spray water from an atomizer with great precaution. Transfer slowly into a large amount of water. Neutralize and drain into the sewer with sufficient water.
Thionyl chloride Preparation Products And Raw materials
Raw materials
1of2
chevron_rightPreparation Products
1of9
chevron_right| Supplier | Tel | Country | ProdList | Advantage | Inquiry |
|---|---|---|---|---|---|
| GLR Innovations | +91 9891111994 | New Delhi, India | 4542 | 58 | Inquiry |
| ANJI BIOSCIENCES | +91-9000100077 +91-9000100077 | Hyderabad, India | 430 | 58 | Inquiry |
| P C SHROFF AND CO | +91-9825066948 +91-9825066948 | Gujarat, India | 11 | 58 | Inquiry |
| JSK Chemicals | +919879767970 | Gujarat, India | 3756 | 58 | Inquiry |
| Panoli Intermediates (India) Pvt., Limited. (Kutch Chemical Industries Ltd.) | +91-2652397013 +91-7226032610 | Gujarat, India | 45 | 58 | Inquiry |
| LANXESS India Pvt. Ltd. | +91-8356878763 +91-8356878763 | Maharashtra, India | 190 | 58 | Inquiry |
| Shree Sulphurics Pvt Ltd (SSPL) | +91-2224915709 +91-9833300322 | Mumbai, India | 30 | 58 | Inquiry |
| Sima Chemicals (Sima Products) | +91-9821136312 +91-9321375313 | Maharashtra, India | 20 | 58 | Inquiry |
| AKASH PHARMA EXPORTS | +91-9388123451 +91-9846039283 | Kerela, India | 470 | 58 | Inquiry |
| RYZE CHEMIE | +91-9702966440 +91-9702966440 | Maharashtra, India | 335 | 58 | Inquiry |
Related articles
- Thionyl chloride- a Sulfinyl Halide
- Thionyl chloride is an inorganic compound with the chemical formula SOCl2. Thionyl chloride is a sulfinyl halide in which both....
- Nov 18,2019
7719-09-7(Thionyl chloride)Related Search:
1of4
chevron_right




