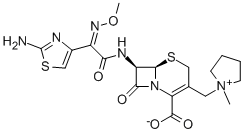
Cefepime
- CAS No.
- 88040-23-7
- Chemical Name:
- Cefepime
- Synonyms
- Cefepime impurity;CEFEPIME ARGININE;CFPM;Cefpim;CEFEPIME;FR 81335;Tsefepim;BMY 28142;Cefepime Jp;Cefepime, >98%
- CBNumber:
- CB1737417
- Molecular Formula:
- C19H24N6O5S2
- Molecular Weight:
- 480.56
- MOL File:
- 88040-23-7.mol
- MSDS File:
- SDS
- Modify Date:
- 2024/7/2 8:55:01
| Melting point | 150 C |
|---|---|
| form | Solid |
| color | White to light yellow |
| InChIKey | HVFLCNVBZFFHBT-IZVURRFBNA-N |
| SMILES | N12C([C@@H](NC(=O)/C(=N\OC)/C3N=C(N)SC=3)[C@H]1SCC(C[N+]1(C)CCCC1)=C2C([O-])=O)=O |&1:2,16,r| |
| CAS DataBase Reference | 88040-23-7(CAS DataBase Reference) |
SAFETY
Risk and Safety Statements
| Symbol(GHS) |  GHS07 |
|---|---|
| Signal word | Warning |
| Hazard statements | H302 |
| Precautionary statements | P280-P305+P351+P338 |
| Hazard Codes | Xi |
| Risk Statements | 36/37/38-42/43 |
| Safety Statements | 22-26-36/37/39 |
| HS Code | 29419000 |
Cefepime Chemical Properties,Uses,Production
Description
Cefepime is a new fourth-generation parenteral cephalosporine antibiotic launched in 1993 in Sweden and France. Cefepime has broad spectrum antimicrobial activity against Staphylococcus, Strepfococcus, Pseudomonas, and the Enterobacteriaceae, including many bacterial isolates that are resistant to commonly used ceftazidime and cefotaxime. Its efficacy has been demonstrated in the treatment of lower respiratory tract infections especially pneumonia, intra-abdominal and urinary tract infections, skin and soft tissue infections, chronic osteomyelitis and in prophylaxis of biliary tract and prostate infections. It is well tolerated by patients and is reported to exhibit no significant drug interactions.
Chemical Properties
colorless Powder
Uses
Cefepime is used for bacterial infections caused by microorganisms that are sensitive to drugs in septicemia, bacteriemia, complicated infections of the upper and lower sections of the urinary system, pneumonia, pulmonary abscesses, emphysema of the pleura, fever in patients with neutropenia, and infected skin and soft tissue wounds. Synonyms of this drug are maxipime, cepim, cepimex, and others.
Antimicrobial activity
Its activity against common pathogens is comparable to that of group 4 cephalosporins, but it is somewhat more active against Ps. aeruginosa. Like cefpirome it has low affinity for the molecular class C cephalosporinases of many Gram-negative rods and is consequently active against most strains of Citrobacter spp., Enterobacter spp., Serratia spp. and Ps. aeruginosa that are resistant to cefotaxime and ceftazidime. It has poor activity against L. monocytogenes and against anaerobic organisms.
Pharmacokinetics
Cmax 2 g intravenous (30-min infusion): c. 160 mg/L end infusion
Plasma half-life: c. 2 h
Volume of distribution: 14–20 L
Plasma protein binding: 10–19%
It is well distributed. Penetration into tissues, including lung,
appears to be similar to that of other aminothiazoyl cephalosporins.
Very low concentrations are achieved in CSF in the
absence of meningeal inflammation. It is secreted in breast
milk.
It is partially metabolized, but 85% of the dose is excreted
unchanged in the urine, achieving a concentration approaching
1 g/L within 4 h of a 1 g intravenous dose. Dosage adjustment
is required in patients with impaired renal function,
but hepatic impairment does not affect the pharmacokinetic
properties.
Clinical Use
Cefepime (Maxipime, Axepin) is a parenteral, β-lactamase–resistant cephalosporin that is chemically and microbiologicallysimilar to cefpirome. It also has a broadantibacterial spectrum, with significant activity against bothGram-positive and Gram-negative bacteria, including streptococci,staphylococci, Pseudomonas spp., and theEnterobacteriaceae. It is active against some bacterial isolatesthat are resistant to ceftazidime. The efficacy of cefepimehas been demonstrated in the treatment of urinary tract infections,lower respiratory tract infections, skin and soft tissueinfections, chronic osteomyelitis, and intra-abdominal andbiliary infections. It is excreted in the urine with a half-life of2.1 hours. It is bound minimally to plasma proteins. Cefepimeis also a fourth-generation cephalosporin.
Side effects
It is used in the treatment of serious infections, particularly those in which resistant Gram-negative pathogens are known or suspected to be involved.
Cefepime Preparation Products And Raw materials
Raw materials
1of2
chevron_rightPreparation Products
| Supplier | Tel | Country | ProdList | Advantage | Inquiry |
|---|---|---|---|---|---|
| CHEMXTREE STANDARDS | +919872732255 | Punjab, India | 96 | 58 | Inquiry |
| PROTECH TELELINKS | +91-8571891912 +91-9855060837 | Himachal Pradesh, India | 62 | 58 | Inquiry |
| Concept Pharmaceuticals Ltd | +91-2242418888 +91-2242418888 | Maharashtra, India | 19 | 58 | Inquiry |
| CLEARSYNTH LABS LTD. | +91-22-45045900 | Hyderabad, India | 6351 | 58 | Inquiry |
| Pharmaffiliates Analytics and Synthetics P. Ltd | +91-172-5066494 | Haryana, India | 6773 | 58 | Inquiry |
| SynZeal Research Pvt Ltd | +1 226-802-2078 | Gujarat, India | 6522 | 58 | Inquiry |
| Pharma Affiliates | 172-5066494 | Haryana, India | 6761 | 58 | Inquiry |
| Medilink Pharmachem | +91 (79) 3007-0133 | New Delhi, India | 424 | 50 | Inquiry |
| ORCHID PHARMA LTD | +91 - 44 - 2821 1000 | New Delhi, India | 69 | 58 | Inquiry |
| Wuhan Jingkang en Biomedical Technology Co., Ltd | +8613720134139 | China | 4692 | 58 | Inquiry |
Related articles
- Cefepime: Indications, Mechanism of Action and Side Effects
- Cefepime is a broad-spectrum fourth-generation cephalosporin with activity against both gram-positive and gram-negative bacter....
- Jun 25,2024
- Cefepime: Antimicrobial Activity, Susceptibility, Administration and Dosage, Clinical Uses etc.
- Cefepime is an oxymino b-lactam with an amino thiazolyn side chain classified as a fourth-generation cephalosporin. In general....
- Mar 25,2022
88040-23-7(Cefepime)Related Search:
1of4
chevron_right




