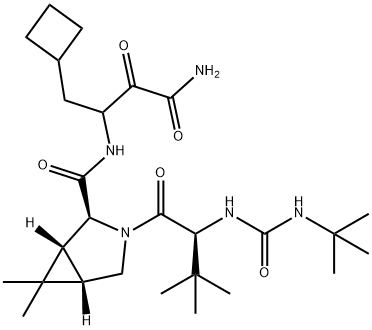
Boceprevir
- CAS No.
- 394730-60-0
- Chemical Name:
- Boceprevir
- Synonyms
- CS-354;EBP 520;Sch 503034;Bocepravir;Boceprevir;Boceprevir, >=98%;Boceprevir(EBP 520;Boceprevir USP/EP/BP;Boceprevir(Sch 503034);TIANFUCHEM-- Boceprevir
- CBNumber:
- CB32491465
- Molecular Formula:
- C27H45N5O5
- Molecular Weight:
- 519.68
- MOL File:
- 394730-60-0.mol
- MSDS File:
- SDS
- Modify Date:
- 2023/6/8 9:02:27
| Melting point | >107°C (dec.) |
|---|---|
| Density | 1.162 |
| storage temp. | -20°C |
| solubility | Soluble in DMSO (up to 15 mg/ml with warming) |
| form | solid |
| pka | 12.82±0.40(Predicted) |
| color | White or off-white |
| Stability | Stable for 2 years from date of purchase as supplied. Solutions in DMSO may be stored at -20°C for up to 3 months. |
SAFETY
Risk and Safety Statements
| Symbol(GHS) |  GHS08 |
|||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Signal word | Warning | |||||||||
| Hazard statements | H361f | |||||||||
| Precautionary statements | P201-P202-P280-P308+P313-P405-P501 | |||||||||
| HS Code | 2933599590 | |||||||||
| NFPA 704 |
|
Boceprevir price More Price(2)
Boceprevir Chemical Properties,Uses,Production
Description
In May 2011, the U.S. FDA approved boceprevir (SCH-503034), to be
given in combination with peginterferon alfa plus ribavirin, for the treatment
of patients with chronic hepatitis C genotype 1 viral infection.
Boceprevir and telaprevir are the first hepatitis C virus (HCV)
protease inhibitors to be approved for the treatment of HCV infection.
Boceprevir is an inhibitor of HCV NS3-4A protease, an essential enzyme required by HCV for
posttranslational processing of viral proteins into their mature forms.
Boceprevir binds covalently, but reversibly, to the active site serine by addition
of the hydroxyl group to the keto-amide functionality. Boceprevir
inhibits HCV NS3-4A protease with a Ki of 14 nM. In cell culture, the EC50
of boceprevir was 200 nM for an HCV replicon constructed from genotype
1b. Boceprevir was two-to threefold less potent against HCV replicon from
genotypes 1a, 2, and 3. The potency of boceprevir decreased threefold in the
presence of human serum. Boceprevir was discovered through a series of
systematic truncations and modifications of a keto-amide undecapeptide
lead molecule.
Boceprevir is synthesized by coupling of 3-amino-4-
cyclobutyl-2-hydroxybutyramide or the related oxobutyramide with a
cyclopropyl-pyrrolidine carboxylic acid intermediate. The pyrrolidine derivative
can be prepared via cyclopropanation of a bicyclic lactam derivative
or by conversion of 3,3-dimethylcyclopropane-1,2-dicarboxylic acid to the
pyrrolidine in a multistep route. Boceprevir is a 1:1 mixture of diastereomers
at the readily epimerizable position a to the keto group.
Chemical Properties
Off-White to Pale Yellow Solid
Uses
An NS3 serine protease inhibitor of hepatitis C virus, for the treatment of HCV infection. It is a COVID19-related research product.
Definition
ChEBI: A synthetic tripeptide consisting of N-(tert-butylcarbamoyl)-3-methyl-L-valyl, a cyclopropyl-fused prolyl and 3-amino-4-cyclobutyl-2-oxobutanamide residues joined in sequence. Used for treatment of chronic h patitis C virus genotype 1 infection.
Clinical Use
Boceprevir is an oral inhibitor of HCV NS3/4A protease for the treatment of the chronic hepatitis C genotype infection. It is approved as combination therapy with Peg-IFN-alpha and ribavarin to treat adult patients with compensated liver diseasewhoare either treatment naive or who have experienced prior failed therapy with interferon and ribavarin. Boceprevir was initially discovered by Schering-Plough and developed and marketed by Merck & Co. since its acquisition of Schering-Plough in 2009. Several publications have highlighted the discovery of this drug, which evolved from a potent initial undecapeptide lead structure to boceprevir (VII) as a drug candidate with potent activity and desirable PK properties.
Boceprevir Preparation Products And Raw materials
Raw materials
1of3
chevron_rightPreparation Products
| Supplier | Tel | Country | ProdList | Advantage | Inquiry |
|---|---|---|---|---|---|
| BDR Pharmaceuticals International Pvt Ltd | +91-2240560560 +91-7718884418 | Maharashtra, India | 206 | 58 | Inquiry |
| Pharma Affiliates | 172-5066494 | Haryana, India | 6761 | 58 | Inquiry |
| CLEARSYNTH LABS LTD. | +91-22-45045900 | Hyderabad, India | 6351 | 58 | Inquiry |
| A.J Chemicals | 91-9810153283 | New Delhi, India | 6124 | 58 | Inquiry |
| Triveni chemicals | 08048762458 | New Delhi, India | 6093 | 58 | Inquiry |
| Pharmaffiliates Analytics and Synthetics P. Ltd | +91-172-5066494 | Haryana, India | 6773 | 58 | Inquiry |
| Taj Mahal Vision Chemicals Private Limited | 07942557933 | Mumbai, India | 206 | 58 | Inquiry |
| Alpha Biopharmaceuticals Co., Ltd | +86-15542445688 | China | 888 | 58 | Inquiry |
| Henan Tianfu Chemical Co.,Ltd. | +86-0371-55170693 +86-19937530512 | China | 21675 | 55 | Inquiry |
| career henan chemical co | +86-0371-86658258 +8613203830695 | China | 29900 | 58 | Inquiry |





