Potassium citrate monohydrate
- CAS No.
- 6100-05-6
- Chemical Name:
- Potassium citrate monohydrate
- Synonyms
- TRIPOTASSIUM CITRATE MONOHYDRATE;Urocit-K;Hydroxycitric acid (tripotassium hydrate);CITRIC ACID TRIPOTASSIUM SALT MONOHYDRATE;KALII CITRAS;tri-Potassium citrate;TrikaliumcitratH2O,BP;Potassium Citrate FCC;Tri-PotassiumCitrateGr;PotassiuM Citrate (AS)
- CBNumber:
- CB6280529
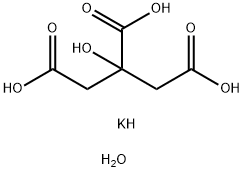
- Molecular Formula:
- C6H11KO8
- Molecular Weight:
- 250.24
- MOL File:
- 6100-05-6.mol
- MSDS File:
- SDS
- Modify Date:
- 2024/10/11 22:00:27
| Melting point | 275 °C (dec.)(lit.) |
|---|---|
| Density | 1.98 |
| storage temp. | Inert atmosphere,Room Temperature |
| solubility | H2O: 1 M at 20 °C, clear, colorless |
| form | Granular |
| Specific Gravity | 1.98 |
| color | White crystalline |
| Odor | Odorless |
| PH Range | 8 - 9.5 |
| PH | 7.5-9.0 (25℃, 50mg/mL in H2O) |
| Water Solubility | Soluble in water and glycerol. Practically insoluble in alcoholSoluble in water and glycerol. Insoluble in alcohol |
| Merck | 14,7623 |
| BRN | 3924344 |
| Stability | Stable. Incompatible with strong oxidizing agents. |
| InChIKey | PJAHUDTUZRZBKM-UHFFFAOYSA-K |
| CAS DataBase Reference | 6100-05-6(CAS DataBase Reference) |
| EPA Substance Registry System | Tripotassium citrate monohydrate (6100-05-6) |
SAFETY
Risk and Safety Statements
| Hazard Codes | F,T,C,Xi | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Risk Statements | 22-35 | |||||||||
| Safety Statements | 26-36/37/39-45-24/25 | |||||||||
| WGK Germany | 1 | |||||||||
| F | 3 | |||||||||
| TSCA | Yes | |||||||||
| HS Code | 29181500 | |||||||||
| NFPA 704 |
|
Potassium citrate monohydrate price More Price(18)
| Manufacturer | Product number | Product description | CAS number | Packaging | Price | Updated | Buy |
|---|---|---|---|---|---|---|---|
| Sigma-Aldrich(India) | P1722 | Potassium citrate tribasic monohydrate meets USP testing specifications | 6100-05-6 | 100G | ₹3258.33 | 2022-06-14 | Buy |
| Sigma-Aldrich(India) | P1722 | Potassium citrate tribasic monohydrate meets USP testing specifications | 6100-05-6 | 500G | ₹9374.45 | 2022-06-14 | Buy |
| Sigma-Aldrich(India) | C8385 | Potassium citrate tribasic monohydrate ≥98% (GC/titration) | 6100-05-6 | 500G | ₹2381.5 | 2022-06-14 | Buy |
| Sigma-Aldrich(India) | C8385 | Potassium citrate tribasic monohydrate ≥98% (GC/titration) | 6100-05-6 | 1KG | ₹3626.38 | 2022-06-14 | Buy |
| Sigma-Aldrich(India) | C3029 | Potassium citrate tribasic monohydrate suitable for cell culture | 6100-05-6 | 500G | ₹4330 | 2022-06-14 | Buy |
Potassium citrate monohydrate Chemical Properties,Uses,Production
Chemical Properties
White/clear crystalline powder
Uses
Tripotassium Citrate Monohydrate is used in manufacturing method of printed circuit board for autonomous vehicle to perform recognition, judgment, determination and execution.Citric acid is a weak organic acid that is known as a commodity chemical, as more than a million tonnes are produced every year by mycological fermentation on an industrial scale using crude sugar solutions, such as molasses and strains of Aspergillus niger. Citric acid is widely distributed in plants and in animal tissues and fluids and exist in greater than grace amounts in variety of fruits and vegetables, most notably in citrus fruits such as lemon and limes. Citric acid is mainly used as an acidifier, flavoring agent and chelating agent. Citric acid is also a metabolite of Dimethyl Fumarate (D464965), a compound
Production Methods
Potassium citrate is prepared by adding either potassium bicarbonate or potassium carbonate to a solution of citric acid until effervescence ceases. The resulting solution is then filtered and evaporated to dryness to obtain potassium citrate.
Definition
ChEBI: A hydrate that is the monohydrate form of potassium citrate.
General Description
Potassium citrate tribasic monohydrate (KCTM) is a potassium salt that has been reported to be efficient in forming aqueous two-phase system (ATPS) with UCON 50-HB-5100, a random copolymer. This product is a high quality pharmacopoeia product that meets the testing specifications of USP (United States Pharmacopoeia). It can be employed in research and pilot studies.
Pharmaceutical Applications
Potassium citrate is used in beverages, foods, and oral pharmaceutical formulations as a buffering and alkalizing agent. It is also used as a sequestering agent and as a therapeutic agent to alkalinize the urine and to relieve the painful irritation caused by cystitis.
Side effects
The most common side effects of potassium citrate are gastrointestinal. Potassium citrate can be irritating to the stomach. Potassium citrate may cause an upset stomach, nausea or vomiting, and diarrhea or loose stools.
Safety
Potassium citrate is used in oral pharmaceutical formulations and is
generally regarded as a nontoxic and nonirritant material by this
route of administration.
Most potassium citrate safety data relate to its use as a
therapeutic agent, for which up to 10 g may be administered daily,
in divided doses, as a treatment for cystitis. Although there are
adverse effects associated with excessive ingestion of potassium
salts, the quantities of potassium citrate used as a pharmaceutical
excipient are insignificant in comparison to those used therapeutically.
(IV, dog): 0.17 g/kg
storage
Potassium citrate is a stable, though hygroscopic material, and should be stored in an airtight container in a cool, dry place. It is deliquescent in moist air.
Purification Methods
It solubility in H2O is 154% and it loses H2O at 180o. [Beilstein 3 III 1091.]
Incompatibilities
Aqueous potassium citrate solutions are slightly alkaline and will react with acidic substances. Potassium citrate may also precipitate alkaloidal salts from their aqueous or alcoholic solutions. Calcium and strontium salts will cause precipitation of the corresponding citrates. Potassium citrate is incompatible with strong oxidizing agents.
Regulatory Status
GRAS listed. Accepted as a food additive in Europe. Included in the FDA Inactive Ingredients Database (oral solutions and suspensions; topical emulsions and aerosol foams). Included in nonparenteral medicines (cutaneous foams and emulsions; oral liquids, granules, mixtures and soluble tablets; topical liquids, emulsions and mousses) licensed in the UK. Included in the Canadian List of Acceptable Non-medicinal Ingredients.
Potassium citrate monohydrate Preparation Products And Raw materials
| Supplier | Tel | Country | ProdList | Advantage | Inquiry |
|---|---|---|---|---|---|
| Rubexco Pvt Ltd | +91-9825231936 +91-9909917613 | Gujarat, India | 36 | 58 | Inquiry |
| Rivashaa Agrotech Biopharma Pvt. Ltd. | +91-26463395 +91-7926462688 | Gujarat, India | 1615 | 58 | Inquiry |
| Nutracare International | +91-2228065292 +91-9820315046 | Maharashtra, India | 36 | 58 | Inquiry |
| G Amphray Laboratories | +91-2222663150 +91-9820084212 | Maharashtra, India | 13 | 58 | Inquiry |
| Rishi Chemical Works Pvt Ltd | +91-3322290068 +91-3322290071 | Kolkata, India | 43 | 58 | Inquiry |
| SYNOVA CHEMICALS | +91-9920741772 +91-9920741772 | Mumbai, India | 428 | 58 | Inquiry |
| Kronox Lab Sciences Pvt Ltd | +91-9313231074 +91-9313231074 | Gujarat, India | 240 | 58 | Inquiry |
| India Phosphate | +91-9993319656 +91-9993319656 | Madhya Pradesh, India | 61 | 58 | Inquiry |
| New Alliance Dye Chem Pvt Ltd | +91-9820112338 +91-9820112338 | Maharashtra, India | 104 | 58 | Inquiry |
| Annexe Chem Pvt Ltd | +91-9724624061 +91-9898722162 | Gujarat, India | 57 | 58 | Inquiry |
| Supplier | Advantage |
|---|---|
| Rubexco Pvt Ltd | 58 |
| Rivashaa Agrotech Biopharma Pvt. Ltd. | 58 |
| Nutracare International | 58 |
| G Amphray Laboratories | 58 |
| Rishi Chemical Works Pvt Ltd | 58 |
| SYNOVA CHEMICALS | 58 |
| Kronox Lab Sciences Pvt Ltd | 58 |
| India Phosphate | 58 |
| New Alliance Dye Chem Pvt Ltd | 58 |
| Annexe Chem Pvt Ltd | 58 |
6100-05-6(Potassium citrate monohydrate)Related Search:
1of4
chevron_right




