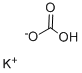
Potassium bicarbonate
- CAS No.
- 298-14-6
- Chemical Name:
- Potassium bicarbonate
- Synonyms
- bicarbonate;POTASSIUM HYDROGEN CARBONATE;K-Lyte;POTASSIUM ACID CARBONATE;Kafylox;POTASSIUM BICARBONAT;POTASSIUM BICARBONATE;Kaliumhydrogencarbonat;kalii hydrogenocarbonas;Potassium bicarbonate,AR
- CBNumber:
- CB3260704
- Molecular Formula:
- CHKO3
- Molecular Weight:
- 100.12
- MOL File:
- 298-14-6.mol
- MSDS File:
- SDS
- Modify Date:
- 2024/7/25 20:04:51
| Melting point | 292 °C |
|---|---|
| Density | 2,17 g/cm3 |
| storage temp. | Store at +15°C to +25°C. |
| solubility | H2O: 1 M at 20 °C, clear, colorless |
| form | fine crystals |
| pka | 10.33(at 25℃) |
| color | White |
| Specific Gravity | 2.17 |
| Odor | Odorless |
| PH Range | 8 - 9 |
| PH | 8.27(1 mM solution);8.25(10 mM solution);8.13(100 mM solution); |
| Water Solubility | Soluble in water. Insoluble in alcohol. |
| λmax |
λ: 260 nm Amax: 0.010 λ: 280 nm Amax: 0.01 |
| Merck | 14,7609 |
| BRN | 4535309 |
| Stability | Stable. |
| InChIKey | TYJJADVDDVDEDZ-UHFFFAOYSA-M |
| CAS DataBase Reference | 298-14-6(CAS DataBase Reference) |
| EPA Substance Registry System | Potassium bicarbonate (298-14-6) |
SAFETY
Risk and Safety Statements
| Symbol(GHS) |  GHS07 |
|||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Signal word | Warning | |||||||||
| Hazard statements | H302-H315-H319-H335 | |||||||||
| Precautionary statements | P261-P280-P305+P351+P338 | |||||||||
| Hazard Codes | F,T,C,Xi | |||||||||
| Risk Statements | 22-35 | |||||||||
| Safety Statements | 24/25-45-36/37/39-26 | |||||||||
| WGK Germany | 1 | |||||||||
| RTECS | FG1840000 | |||||||||
| F | 3 | |||||||||
| TSCA | Yes | |||||||||
| HS Code | 28364000 | |||||||||
| Toxicity | LD50 orally in Rabbit: > 2000 mg/kg | |||||||||
| NFPA 704 |
|
Potassium bicarbonate price More Price(20)
| Manufacturer | Product number | Product description | CAS number | Packaging | Price | Updated | Buy |
|---|---|---|---|---|---|---|---|
| Sigma-Aldrich(India) | 60339 | Potassium bicarbonate BioUltra, ≥99.5% (T) | 298-14-6 | 500G | ₹4806.3 | 2022-06-14 | Buy |
| Sigma-Aldrich(India) | 60339 | Potassium bicarbonate BioUltra, ≥99.5% (T) | 298-14-6 | 1KG | ₹5369.2 | 2022-06-14 | Buy |
| Sigma-Aldrich(India) | 431583 | Potassium bicarbonate ≥99.95% trace metals basis, 99.7-100.5% dry basis | 298-14-6 | 50G | ₹8227 | 2022-06-14 | Buy |
| Sigma-Aldrich(India) | 431583 | Potassium bicarbonate ≥99.95% trace metals basis, 99.7-100.5% dry basis | 298-14-6 | 250G | ₹27712 | 2022-06-14 | Buy |
| Sigma-Aldrich(India) | 237205 | Potassium bicarbonate ACS reagent, 99.7%, powder, crystals or granules | 298-14-6 | 100G | ₹3150.08 | 2022-06-14 | Buy |
Potassium bicarbonate Chemical Properties,Uses,Production
Chemical Properties
Potassium bicarbonate occurs as colorless, transparent crystals or as a white granular or crystalline powder. It is odorless, with a saline or weakly alkaline taste.
Uses
Potassium Bicarbonate is an alkali and leavening agent obtained as colorless prisms or white powder. it is very soluble, with 1 g dis- solving in 2.8 ml of water. upon heating, it liberates carbon dioxide which provides leavening in baked goods. it is also used in confec- tionary products.
Production Methods
Potassium bicarbonate can be made by passing carbon dioxide into
a concentrated solution of potassium carbonate, or by exposing
moist potassium carbonate to carbon dioxide, preferably under
moderate pressure.
Potassium bicarbonate also occurs naturally in the mineral
calcinite.
Definition
ChEBI: A potassium salt that is the monopotassium salt of carbonic acid. It has fungicidal properties and is used in organic farming for the control of powdery mildew and apple scab.
General Description
Potassium bicarbonate is water soluble alkaline potassium salt with monoclinic crystalline structure. It is a raw material for the synthesis of many potassium compounds. It is a better coolant than sodium bicarbonate in the aerosol fire extinguishing apparatus. It shows potential as an antifungal agent.
Pharmaceutical Applications
As an excipient, potassium bicarbonate is generally used in
formulations as a source of carbon dioxide in effervescent
preparations, at concentrations of 25–50% w/w. It is of particular
use in formulations where sodium bicarbonate is unsuitable, for
example, when the presence of sodium ions in a formulation needs
to be limited or is undesirable. Potassium bicarbonate is often
formulated with citric acid or tartaric acid in effervescent tablets or
granules; on contact with water, carbon dioxide is released through
chemical reaction, and the product disintegrates. On occasion, the
presence of potassium bicarbonate alone may be sufficient in tablet formulations, as reaction with gastric acid can be sufficient to cause
effervescence and product disintegration.
Potassium bicarbonate has also been investigated as a gasforming
agent in alginate raft systems.The effects of potassium
bicarbonate on the stability and dissolution of paracetamol and
ibuprofen have been described.
Potassium bicarbonate is also used in food applications as an
alkali and a leavening agent, and is a component of baking powder.
Therapeutically, potassium bicarbonate is used as an alternative
to sodium bicarbonate in the treatment of certain types of metabolic
acidosis. It is also used as an antacid to neutralize acid secretions in
the gastrointestinal tract and as a potassium supplement.
Agricultural Uses
Potassium bicarbonate (KHCO3), also called potassium
hydrogen carbonate, is a white crystalline solid, soluble
in water (insoluble in ethanol). It decomposes at about
120°C. Potassium bicarbonate contains about 28%
potassium (K2O) and used as a potassium supplying
fertilizer.
Potassium bicarbonate, which occurs naturally as
calcinite, is made by passing carbon dioxide into
saturated potassium carbonate solution. It is used as
baking powder and as a fire extinguisher.
Safety
Potassium bicarbonate is used in cosmetics, foods, and oral pharmaceutical formulations, where it is generally regarded as a relatively nontoxic and nonirritant material when used as an excipient. However, excessive consumption of potassium bicarbonate or other potassium salts may produce toxic manifestations of hyperkalemia.
storage
Potassium bicarbonate should be stored in a well-closed container in a cool, dry location. Potassium bicarbonate is stable in air at normal temperatures, but when heated to 100–200°C in the dry state, or in solution, it is gradually converted to potassium carbonate.
Purification Methods
It is crystallised from water at 65-70o (1.25mL/g) by filtering and then cooling to 15o (~0.4ml/g). During all operations, CO2 is passed through the stirred mixture. The crystals are sucked dry at the pump, washed with distilled water, dried in air and then over H2SO4 in an atmosphere of CO2. It is much less soluble than the carbonate in H2O (see below).
Incompatibilities
Potassium bicarbonate reacts with acids and acidic salts with the evolution of carbon dioxide.
Regulatory Status
E501 refers to potassium carbonates). Included in nonparenteral medicines licensed in the UK and USA (chewable tablets; effervescent granules; effervescent tablets; lozenges; oral granules; oral suspensions; powder for oral solutions). Included in the Canadian List of Acceptable Non-medicinal Ingredients.
Potassium bicarbonate Preparation Products And Raw materials
Raw materials
1of2
chevron_rightPreparation Products
1of5
chevron_right| Supplier | Tel | Country | ProdList | Advantage | Inquiry |
|---|---|---|---|---|---|
| ANJI BIOSCIENCES | +91-9000100077 +91-9000100077 | Hyderabad, India | 430 | 58 | Inquiry |
| Rishi Chemical Works Pvt Ltd | +91-3322290068 +91-3322290071 | Kolkata, India | 43 | 58 | Inquiry |
| Yogi Dye Chem Industries | +91-9821098577 +91-9821098577 | Mumbai, India | 124 | 58 | Inquiry |
| UNILOSA INTERNATINAL PRIVATE LIMITED | +91-9999069917 +91-9999069917 | New Delhi, India | 152 | 58 | Inquiry |
| JSK Chemicals | +919879767970 | Gujarat, India | 3756 | 58 | Inquiry |
| Annexe Chem Pvt Ltd | +91-9724624061 +91-9898722162 | Gujarat, India | 57 | 58 | Inquiry |
| HRV Global Life Sciences | +91-9820219686 +91-9820219686 | Telangana, India | 379 | 58 | Inquiry |
| Sujata Chemicals | +91-9898025496 +91-9898025496 | Gujarat, India | 73 | 58 | Inquiry |
| Nandu Chemical Industries | +91-8362330469 +91-8362330469 | Karnataka, India | 84 | 58 | Inquiry |
| Expresolv Limited | +91-787495635 +91-7069552255 | Gujarat, India | 49 | 58 | Inquiry |
| Supplier | Advantage |
|---|---|
| ANJI BIOSCIENCES | 58 |
| Rishi Chemical Works Pvt Ltd | 58 |
| Yogi Dye Chem Industries | 58 |
| UNILOSA INTERNATINAL PRIVATE LIMITED | 58 |
| JSK Chemicals | 58 |
| Annexe Chem Pvt Ltd | 58 |
| HRV Global Life Sciences | 58 |
| Sujata Chemicals | 58 |
| Nandu Chemical Industries | 58 |
| Expresolv Limited | 58 |
Related articles
- Potassium bicarbonate: The all-rounder of the chemical world
- This article popularize the chemistry of potassium bicarbonate in an all-round way and bring you to appreciate its unique styl....
- Jul 2,2024
- Preparation of potassium bicarbonate
- Potassium bicarbonate is an inorganic substance with the chemical formula KHCO3.
- Apr 13,2022





