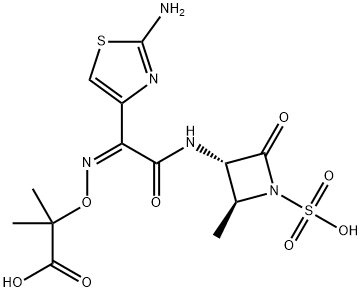
Aztreonam
- CAS No.
- 78110-38-0
- Chemical Name:
- Aztreonam
- Synonyms
- AZACTAM;clobetasol propionate powder;monobactam;Aztreonam/arginine;[2S-[2A,3B(Z)]]-2-[[[1-(2-AMINO-4-THIAZOLYL)-2-[(2-METHYL-4-OXO-1-SULFO-3-AZETIDINYL)AMINO]-2-OXOETHYLIDENE]AMINO]OXY]-2-METHYLPROPANOIC ACID;Azonam;Aztreon;NSC1245;sq26,776;Nebactam
- CBNumber:
- CB0386184
- Molecular Formula:
- C13H17N5O8S2
- Molecular Weight:
- 435.43
- MOL File:
- 78110-38-0.mol
- MSDS File:
- SDS
- Modify Date:
- 2024/6/13 17:03:25
| Melting point | 227°C |
|---|---|
| Density | 1.83 |
| refractive index | 1.6460 (estimate) |
| storage temp. | room temp |
| solubility | DMSO (Slightly), Water (Slightly, Heated, Sonicated) |
| form | solid |
| pka | pKa -0.7(H2O t=RT Iunde?ned) (Uncertain);2.75(H3O t=RT Iunde?ned) (Uncertain);3.91(H4O t=RT Iunde?ned) (Uncertain) |
| color | white to beige |
| Water Solubility | Soluble in DMF/water (1:1) at 50 mg/ml |
| Merck | 14,925 |
| InChIKey | WZPBZJONDBGPKJ-VEHQQRBSSA-N |
| SMILES | C(O)(=O)C(O/N=C(/C1=CSC(N)=N1)\C(N[C@@H]1C(=O)N(S(O)(=O)=O)[C@H]1C)=O)(C)C |
| CAS DataBase Reference | 78110-38-0(CAS DataBase Reference) |
SAFETY
Risk and Safety Statements
| Symbol(GHS) |   GHS08,GHS09 |
|---|---|
| Signal word | Danger |
| Hazard statements | H400-H334 |
| Precautionary statements | P273-P391-P501-P261-P285-P304+P341-P342+P311-P501 |
| Hazard Codes | Xn |
| Risk Statements | 20/21/22-36/37/38 |
| Safety Statements | 22-24/25-36-26 |
| WGK Germany | 2 |
| RTECS | UA2451400 |
| HS Code | 29419000 |
| Toxicity | TDLo ivn-rat: 1100 mg/kg (7-17D preg):TER NKRZAZ33(Suppl 1),203,85 |
Aztreonam price More Price(5)
| Manufacturer | Product number | Product description | CAS number | Packaging | Price | Updated | Buy |
|---|---|---|---|---|---|---|---|
| Sigma-Aldrich(India) | PZ0038 | Aztreonam ≥98% (HPLC) | 78110-38-0 | 5MG | ₹10619.33 | 2022-06-14 | Buy |
| Sigma-Aldrich(India) | PZ0038 | Aztreonam ≥98% (HPLC) | 78110-38-0 | 25MG | ₹42877.83 | 2022-06-14 | Buy |
| Sigma-Aldrich(India) | PHR1785 | Aztreonam Pharmaceutical Secondary Standard; Certified Reference Material | 78110-38-0 | 500MG | ₹24312.95 | 2022-06-14 | Buy |
| Sigma-Aldrich(India) | A6848 | Aztreonam analytical standard | 78110-38-0 | 50MG | ₹21888.15 | 2022-06-14 | Buy |
| SRL | 13741 | Aztreonam (AZN), 96% | 78110-38-0 | 100mg | ₹7120 | 2022-05-26 | Buy |
Aztreonam Chemical Properties,Uses,Production
Description
Aztreonam is the first member of the monobactam class of antibiotics to be introduced into the world market. It possesses high β-lactamase stability and moderately good activity against gram negative aerobes such as E. coli, S. marcescens, -9 Proteus Providencia, Salmonella, g. influenzae, E. gonorrhea, and &. pneumonia. While somewhat less potent against Pseudomonas aeruginosa, it is nonetheless one of the better β-lactams against this species. It has poor activity against gram positive organisms.
Chemical Properties
White Crystalline Powder
History
Aztreonam was synthesized by the Squibb Institute for Medical Research in 1981 starting with l-threonine. The synthesis was based on findings about bacterial β-lactam compounds of a monocyclic nature . The β-lactam compounds, called monobactams, were isolated from Chromobacterium violaceum, Agrobacterium radiobacter, etc. Such monocyclic βlactams of bacterial origin had previously been found independently in 1981 by Takeda Chemicals Industries in the culture broths of Pseudomonas acidophila and P. mesoacidophila and named sulfazecin and isosulfazecin, respectively. Aztreonam was selected from among hundreds of derivatives as a candidate for clinical trials because of its unique antibacterial spectrum and strong activity. This antibiotic shows excellent activity against a variety of gram-negative aerobic bacteria but no activity against gram-positive bacteria or anaerobes. Its efficacy and safety are now being clinically evaluated.
Uses
Aztreonam is a synthetic β-lactam antibiotic of the monobactam class. It is effective against Gram-negative bacteria but inactive against Gram-positive bacteria. Its mechanism of action involves the inhibition of mucopeptide synthesis in the bacterial cell wall, thereby blocking peptidoglycan crosslinking. Aztreonam is resistant to hydrolysis by some β-lactamases, but is inactivated by extended-spectrum β-lactamases.
Antimicrobial activity
Concentrations (mg/L) inhibiting 50% of other organisms are: Aeromonas spp., 0.1;Acinetobacter spp., 16; Mor. catarrhalis, 0.1; Burkholderia cepacia, 2; and Yersinia spp., 0.1. Synergy has been shown with gentamicin, tobramycin and amikacin against 52–89% of strains of Ps. aeruginosa and gentamicin-resistant Gram-negative bacteria.
Pharmacokinetics
Cmax 1 g intravenous: 90 mg/L end infusion
1 g intramuscular: 46 mg/L after 1 h
Plasma half-life: 1.7 h
Volume of distribution: 0.18 L/kg
Plasma protein binding: 56%
Absorption and distribution
Oral bioavailability is less than 1%. Peak concentrations
above the median MIC for most Gram-negative pathogens
are achieved in most tissues and body fluids after 1 g intramuscular
or intravenous doses.
Metabolism and excretion
It is not extensively metabolized, the most prominent product,
resulting from opening the β-lactam ring, being scarcely
detectable in the serum and accounting for about 6% of the
dose in the urine and 3% in the feces.
It is predominantly eliminated in the urine, where 58–72%
appears within 8 h. Less than 12% is eliminated unchanged in
the feces, suggesting low biliary excretion.
Clinical Use
Urinary tract infections, including pyelonephritis and cystitis
Lower respiratory tract infections, including pneumonia and bronchitis
caused by Gram-negative bacilli
Septicemia
Skin and skin structure infections, including postoperative wounds, ulcers
and burns
Intra-abdominal infections, including peritonitis
Gynecological infections, including endometritis and pelvic cellulitis
Side effects
Local reactions occasionally occur at the injection site.
Systemic reactions include diarrhea, nausea and/or vomiting
and rash (1–1.3%). Neutropenia was seen in 11.3% of the
pediatric patients younger than 2 years. Pseudomembranous
colitis has been reported.
There are no reactions in patients with immunoglobulin E
(IgE) antibodies to benzylpenicillin or penicillin moieties. It
is rarely cross-reactive with other β-lactam antibiotics and is
weakly immunogenic.
Safety Profile
Moderately toxic by severalroutes. An experimental teratogen. Other experimentalreproductive effects. When heated to decomposition itemits toxic fumes of NOx and SOx.
Aztreonam Preparation Products And Raw materials
Raw materials
1of3
chevron_rightPreparation Products
| Supplier | Tel | Country | ProdList | Advantage | Inquiry |
|---|---|---|---|---|---|
| DAVY CHEM LABS | +91-9603460845 +91-9493444489 | Hyderabad, India | 109 | 58 | Inquiry |
| PROTECH TELELINKS | +91-8571891912 +91-9855060837 | Himachal Pradesh, India | 62 | 58 | Inquiry |
| Unitrade Agencies | 08048372548Ext 632 | Mumbai, India | 197 | 58 | Inquiry |
| Globofarm International Private Limited | 91-22-49716506 | Maharashtra, India | 188 | 58 | Inquiry |
| Hemarsh Technologies | 08068441092Ext 237 | Vadodara, India | 23 | 58 | Inquiry |
| Anand Agencies | 91-20-24454597 | Maharashtra, India | 2337 | 58 | Inquiry |
| Dharmanandan Export Pvt., Ltd. | 91-79-48402200 | Gujarat, India | 161 | 58 | Inquiry |
| Maas Pharma Chemicals | +91-9958666224 | Delhi, India | 1607 | 58 | Inquiry |
| Nakshatra Chemicals | 09819631316 | Mumbai, India | 162 | 58 | Inquiry |
| Shivam Pharma Chemicals | 91-22-26403900 | Maharashtra, India | 656 | 58 | Inquiry |
| Supplier | Advantage |
|---|---|
| DAVY CHEM LABS | 58 |
| PROTECH TELELINKS | 58 |
| Unitrade Agencies | 58 |
| Globofarm International Private Limited | 58 |
| Hemarsh Technologies | 58 |
| Anand Agencies | 58 |
| Dharmanandan Export Pvt., Ltd. | 58 |
| Maas Pharma Chemicals | 58 |
| Nakshatra Chemicals | 58 |
| Shivam Pharma Chemicals | 58 |
Related articles
- Aztreonam: Antimicrobial Activity, Susceptibility, Administration and Dosage, Clinical Uses etc.
- Aztreonam belongs to the class of beta-lactam antibiotics known as monobactams. Unlike penicillins and cephalosporins the mono....
- Mar 25,2022





