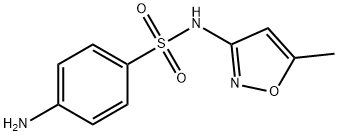
Sulfamethoxazole
- CAS No.
- 723-46-6
- Chemical Name:
- Sulfamethoxazole
- Synonyms
- SMZ;SMX;SULPHAMETHOXAZOLE;Sulfamethoxazol;Co-trimoxazole;Bactrim;N1-(5-METHYLISOXAZOL-3-YL)SULFANILAMIDE;Trib;Septra;Septrin
- CBNumber:
- CB6473303
- Molecular Formula:
- C10H11N3O3S
- Molecular Weight:
- 253.28
- MOL File:
- 723-46-6.mol
- MSDS File:
- SDS
- Modify Date:
- 2024/7/2 8:55:14
| Melting point | 166 °C |
|---|---|
| Boiling point | 482℃ |
| Density | 1.3915 (rough estimate) |
| refractive index | 1.6630 (estimate) |
| Flash point | >110°(230°F) |
| storage temp. | Keep in dark place,Inert atmosphere,Room temperature |
| solubility | Practically insoluble in water, freely soluble in acetone, sparingly soluble in ethanol (96 per cent). It dissolves in dilute solutions of sodium hydroxide and in dilute acids. |
| pka | pKa 5.60±0.05 (Uncertain) |
| form | Solid |
| color | White to Pale Yellow |
| Water Solubility | Soluble in ethanol or acetone. Very slightly soluble in water |
| Sensitive | Light Sensitive |
| Merck | 14,8918 |
| BRN | 6732984 |
| BCS Class | 2,4 |
| Stability | Stable, but light sensitive. Incompatible with strong oxidizing agents. |
| InChIKey | JLKIGFTWXXRPMT-UHFFFAOYSA-N |
| CAS DataBase Reference | 723-46-6(CAS DataBase Reference) |
| IARC | 3 (Vol. Sup 7, 79) 2001 |
| NIST Chemistry Reference | Sulfanilamide, n1-(5-methyl-3-isoxazolyl)-(723-46-6) |
| EPA Substance Registry System | Sulfamethoxazole (723-46-6) |
SAFETY
Risk and Safety Statements
| Symbol(GHS) |   GHS07,GHS08 |
|||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Signal word | Warning | |||||||||
| Hazard statements | H317-H361d | |||||||||
| Precautionary statements | P201-P280-P302+P352-P308+P313 | |||||||||
| Hazard Codes | Xi,Xn | |||||||||
| Risk Statements | 36/37/38-43-22 | |||||||||
| Safety Statements | 26-36-36/37/39-22 | |||||||||
| WGK Germany | 2 | |||||||||
| RTECS | WP0700000 | |||||||||
| TSCA | Yes | |||||||||
| HazardClass | IRRITANT | |||||||||
| HS Code | 29350090 | |||||||||
| Toxicity | LD50 orally in mice: 3662 mg/kg (Yamamoto) | |||||||||
| NFPA 704 |
|
Sulfamethoxazole price More Price(8)
| Manufacturer | Product number | Product description | CAS number | Packaging | Price | Updated | Buy |
|---|---|---|---|---|---|---|---|
| Sigma-Aldrich(India) | S7507 | Sulfamethoxazole analytical standard | 723-46-6 | 10G | ₹5434.15 | 2022-06-14 | Buy |
| Sigma-Aldrich(India) | S7507 | Sulfamethoxazole analytical standard | 723-46-6 | 100G | ₹27971.8 | 2022-06-14 | Buy |
| Sigma-Aldrich(India) | PHR1126 | Sulfamethoxazole Pharmaceutical Secondary Standard; Certified Reference Material | 723-46-6 | 1G | ₹8995.58 | 2022-06-14 | Buy |
| Sigma-Aldrich(India) | 76177 | Sulfamethoxazole certified reference material, TraceCERT? | 723-46-6 | 100MG | ₹9969.83 | 2022-06-14 | Buy |
| Sigma-Aldrich(India) | 31737 | Sulfamethoxazole VETRANAL?, analytical standard | 723-46-6 | 250MG | ₹4535.68 | 2022-06-14 | Buy |
Sulfamethoxazole Chemical Properties,Uses,Production
Description
Like sulfisoxazole, this drug is effective in treating infections caused by streptococci, gonococci, pneumococci, staphylococci as well as colon bacillus. Unlike sulfisoxazole, only about 70% of it binds with proteins in the plasma after oral administration, and it diffuses mostly to tissues and tissue fluids. However, since it is removed much slower than sulfisoxazole, it does not require frequent administration and is also the drug of choice for many systemic infections. Moreover, it is an ingredient of a combined drug named bactrim, biseptol, and so on (which will be examined later on), which has a fixed correlation with trimethoprim. Synonyms of this drug are gantanol, sinomin, sulfisomezole, and others.
Chemical Properties
solid
Definition
ChEBI: An isoxazole (1,2-oxazole) compound having a methyl substituent at the 5-position and a 4-aminobenzenesulfonamido group at the 3-position.
Antimicrobial activity
The intrinsic activity is similar to that of sulfadiazine.
General Description
Sulfamethoxazole’s plasma half-life is 11 hours. Sulfamethoxazole is a sulfonamide drug closely relatedto sulfisoxazole in chemical structure and antimicrobial activity.It occurs as a tasteless, odorless, almost white crystallinepowder. The solubility of sulfamethoxazole in the pHrange of 5.5 to 7.4 is slightly lower than that of sulfisoxazole but higher than that of sulfadiazine, sulfamerazine, or sulfamethazine.Following oral administration, sulfamethoxazole is notabsorbed as completely or as rapidly as sulfisoxazole, andits peak blood level is only about 50% as high.
Air & Water Reactions
Insoluble in water.
Fire Hazard
Flash point data for Sulfamethoxazole are not available but Sulfamethoxazole is probably non-flammable.
Pharmaceutical Applications
This is the sulfonamide component of co-trimoxazole. It is slightly soluble in water.
Pharmacokinetics
Oral absorption: 85%
Cmax 800 mg oral: c.50 mg/L after 3–6 h
Plasma half-life: 6–20 h
Volume of distribution: 12–18 L
Plasma protein binding: 65%
Penetration of extravascular sites, including the CSF, is good.
It crosses the placenta and achieves levels in breast milk of
about 10% of the simultaneous plasma concentration. It is
extensively metabolized, but about 30% of the dose is excreted
unchanged in urine so that high concentrations are achieved.
Clinical Use
Sulfamethoxazole is used only in combination with the diaminopyrimidine trimethoprim.
Side effects
Unwanted effects are those common to sulfonamides. In addition, benign intracranial hypertension has been reported in children. Most side effects of co-trimoxazole are thought to be attributable to the sulfonamide component.
Safety Profile
Moderately toxic by ingestion and intraperitoneal routes. Questionable carcinogen with experimental tumorigenic data. When heated to decomposition it emits very toxic fumes of NOx and SOx.
Sulfamethoxazole Preparation Products And Raw materials
Raw materials
1of3
chevron_rightPreparation Products
| Supplier | Tel | Country | ProdList | Advantage | Inquiry |
|---|---|---|---|---|---|
| ANWITA APIS | +919000311012 | Hyderabad, India | 198 | 58 | Inquiry |
| Reenold Enterprises | +91-8208173540 +91-8208173540 | Maharashtra, India | 28 | 58 | Inquiry |
| Dr. Silviu Pharmachem Pvt., Ltd. | +91-8390608382 +91-8390608382 | Mumbai, India | 248 | 58 | Inquiry |
| Virchow Groups | +919394860022 | Telangana, India | 24 | 58 | Inquiry |
| Refsyn Biosciences Pvt., Ltd. | 91-413-2200273 | Tamil Nadu, India | 226 | 58 | Inquiry |
| CLEANCHEM LABORATORIES LLP | +91 98921 69560 | New Delhi, India | 326 | 58 | Inquiry |
| Anant Pharmaceuticals Pvt. ltd | +91 8550986868 | Maharashtra, India | 736 | 58 | Inquiry |
| B Joshi Agrochem Pharma | 08048984231 | Mumbai, India | 181 | 58 | Inquiry |
| Espee Group | 91-79-40052086 | Gujarat, India | 14 | 58 | Inquiry |
| J. C. Enterprises | 91-22-26128080 | Maharashtra, India | 94 | 58 | Inquiry |
| Supplier | Advantage |
|---|---|
| ANWITA APIS | 58 |
| Reenold Enterprises | 58 |
| Dr. Silviu Pharmachem Pvt., Ltd. | 58 |
| Virchow Groups | 58 |
| Refsyn Biosciences Pvt., Ltd. | 58 |
| CLEANCHEM LABORATORIES LLP | 58 |
| Anant Pharmaceuticals Pvt. ltd | 58 |
| B Joshi Agrochem Pharma | 58 |
| Espee Group | 58 |
| J. C. Enterprises | 58 |
Related articles
- Sulfamethoxazole: A Comprehensive Overview for Chemistry Professionals
- Sulfamethoxazole is a synthetic antibiotic, commonly used in combination with trimethoprim to treat a variety of bacterial inf....
- Jun 6,2024





