Cupric bromide
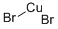
- CAS No.
- 7789-45-9
- Chemical Name:
- Cupric bromide
- Synonyms
- COPPER BROMIDE;COPPER(II) BROMIDE;Dibromocopper;brominating agent;copperbromide(cubr2);CUPRIC BROMIDE;copperdibromide;Copper(Ⅱ) bromide;COPPER(+2)BROMIDE;Brominated ketone
- CBNumber:
- CB9307690
- Molecular Formula:
- Br2Cu
- Molecular Weight:
- 223.35
- MOL File:
- 7789-45-9.mol
- MSDS File:
- SDS
- Modify Date:
- 2023/9/11 17:45:53
| Melting point | 498 °C(lit.) |
|---|---|
| Boiling point | 900 °C |
| Density | 4.77 g/mL at 25 °C(lit.) |
| Flash point | 900°C |
| storage temp. | Store below +30°C. |
| solubility | 1200g/l |
| form | Crystalline Powder |
| Specific Gravity | 4.77 |
| color | Gray-blue |
| Water Solubility | soluble |
| Sensitive | Hygroscopic |
| Merck | 14,2629 |
| Exposure limits |
ACGIH: TWA 1 mg/m3 NIOSH: IDLH 100 mg/m3; TWA 1 mg/m3 |
| Stability | Stable. Incompatible with alkali metals, moisture. Reacts violently with potassium. |
| CAS DataBase Reference | 7789-45-9(CAS DataBase Reference) |
| NIST Chemistry Reference | copper(II) dibromide(7789-45-9) |
| EPA Substance Registry System | Copper bromide (CuBr2) (7789-45-9) |
SAFETY
Risk and Safety Statements
| Symbol(GHS) |    GHS05,GHS07,GHS09 |
|||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Signal word | Danger | |||||||||
| Hazard statements | H302-H314-H410 | |||||||||
| Precautionary statements | P260-P273-P280-P301+P312-P303+P361+P353-P305+P351+P338 | |||||||||
| Hazard Codes | C,Xi | |||||||||
| Risk Statements | 22-34-36/37/38 | |||||||||
| Safety Statements | 26-36/37/39-45-24/25 | |||||||||
| RIDADR | UN 3260 8/PG 3 | |||||||||
| WGK Germany | 3 | |||||||||
| TSCA | Yes | |||||||||
| HS Code | 2827 59 00 | |||||||||
| HazardClass | 8 | |||||||||
| PackingGroup | III | |||||||||
| Toxicity | LD50 orally in Rabbit: 536 mg/kg | |||||||||
| NFPA 704 |
|
Cupric bromide price More Price(21)
| Manufacturer | Product number | Product description | CAS number | Packaging | Price | Updated | Buy |
|---|---|---|---|---|---|---|---|
| Sigma-Aldrich(India) | 8.14658 | Copper(II) bromide for synthesis | 7789-45-9 | 50G | ₹2670 | 2022-06-14 | Buy |
| Sigma-Aldrich(India) | 8.14658 | Copper(II) bromide for synthesis | 7789-45-9 | 250G | ₹6450 | 2022-06-14 | Buy |
| Sigma-Aldrich(India) | 437867 | Copper(II) bromide 99.999% trace metals basis | 7789-45-9 | 5G | ₹5250.13 | 2022-06-14 | Buy |
| Sigma-Aldrich | 221775 | Copper(II) bromide 99% | 7789-45-9 | 5G | ₹1309.83 | 2022-06-14 | Buy |
| Sigma-Aldrich(India) | 221775 | Copper(II) bromide 99% | 7789-45-9 | 5G | ₹1309.83 | 2022-06-14 | Buy |
Cupric bromide Chemical Properties,Uses,Production
Chemical Properties
Black crystal
Uses
As intensifier in photography; as brominating agent in organic synthesis; as humidity indicator; as wood preservative; in solid-electrolyte battery; as stabilizer for acetylated polyformaldehyde.
General Description
Odorless black solid. Sinks and mixes with water.
Air & Water Reactions
Deliquescent. Water soluble.
Reactivity Profile
Acidic inorganic salts, such as Cupric bromide, are generally soluble in water. The resulting solutions contain moderate concentrations of hydrogen ions and have pH's of less than 7.0. They react as acids to neutralize bases. These neutralizations generate heat, but less or far less than is generated by neutralization of inorganic acids, inorganic oxoacids, and carboxylic acid. They usually do not react as either oxidizing agents or reducing agents but such behavior is not impossible. Many of these compounds catalyze organic reactions.
Health Hazard
Inhalation of dust causes irritation of throat and lungs. Ingestion of large amounts causes violent vomiting and purging, intense pain, collapse, coma, convulsions, and paralysis. Contact with solutions causes eye irritation; contact with solid causes severe eye surface injury and skin irritation.
Fire Hazard
Special Hazards of Combustion Products: Irritating hydrogen bromide gas may form in fire.
Purification Methods
Crystallise it twice by dissolving it in water (140mL/g), filtering to remove any Cu2Br2, and concentrating under vacuum at 30o until crystals appear. The cupric bromide is then allowed to crystallise by leaving the solution in a vacuum desiccator containing P2O5 [Hope et al. J Chem Soc 5226 1960, Glemser & Sauer in Handbook of Preparative Inorganic Chemistry (Ed. Brauer) Academic Press Vol II p 1009 1965].
Cupric bromide Preparation Products And Raw materials
Raw materials
Preparation Products
1of4
chevron_right| Supplier | Tel | Country | ProdList | Advantage | Inquiry |
|---|---|---|---|---|---|
| GLR Innovations | +91 9891111994 | New Delhi, India | 4542 | 58 | Inquiry |
| LEEDS KEM LTD | +919284646484 | Maharashtra, India | 9 | 58 | Inquiry |
| NOVOCHEM INDUSTRIES PVT LTD | +91-9823022540 +91-9823022540 | New Delhi, India | 12 | 58 | Inquiry |
| Yogi Dye Chem Industries | +91-9821098577 +91-9821098577 | Mumbai, India | 124 | 58 | Inquiry |
| JSK Chemicals | +919879767970 | Gujarat, India | 3756 | 58 | Inquiry |
| Otto Chemie Pvt Ltd | +91-2222070099 +91-2266382599 | Maharashtra, India | 429 | 58 | Inquiry |
| Parad Corporation Pvt Ltd | +91-7433900100 +91-9426510550 | Gujarat, India | 37 | 58 | Inquiry |
| Brom India | +91-9920046827 +91-9920046827 | Maharashtra, India | 23 | 58 | Inquiry |
| CLEARSYNTH LABS LTD. | +91-22-45045900 | Hyderabad, India | 6351 | 58 | Inquiry |
| A.J Chemicals | 91-9810153283 | New Delhi, India | 6124 | 58 | Inquiry |
| Supplier | Advantage |
|---|---|
| GLR Innovations | 58 |
| LEEDS KEM LTD | 58 |
| NOVOCHEM INDUSTRIES PVT LTD | 58 |
| Yogi Dye Chem Industries | 58 |
| JSK Chemicals | 58 |
| Otto Chemie Pvt Ltd | 58 |
| Parad Corporation Pvt Ltd | 58 |
| Brom India | 58 |
| CLEARSYNTH LABS LTD. | 58 |
| A.J Chemicals | 58 |
7789-45-9(Cupric bromide)Related Search:
1of4
chevron_right




