Cyanogen bromide
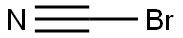
- CAS No.
- 506-68-3
- Chemical Name:
- Cyanogen bromide
- Synonyms
- BrCN;Cyanic bromide;BROMOCYANE;carbononitridic bromide;BROMOCYANIDE;Bromocyanogen;Cyanogens bromide;tl822;(CN)Br;TL 822
- CBNumber:
- CB9853692
- Molecular Formula:
- CBrN
- Molecular Weight:
- 105.92
- MOL File:
- 506-68-3.mol
- MSDS File:
- SDS
- Modify Date:
- 2024/3/14 15:18:33
| Melting point | 50-53 °C (lit.) |
|---|---|
| Boiling point | 61-62 °C (lit.) |
| Density | 1.443 g/mL at 25 °C |
| vapor density | 3.65 (vs air) |
| vapor pressure | 100 mm Hg ( 22.6 °C) |
| refractive index | 1.4670 (estimate) |
| Flash point | 61.4°C |
| storage temp. | 2-8°C |
| solubility | Soluble in chloroform, dichloromethane, ethanol, diethyl ether, benzene and acetonitrile. |
| form | Solution |
| color | White |
| Odor | Penetrating odor |
| Water Solubility | decomposed slowly by cold H2O [HAW93] |
| Sensitive | Moisture & Light Sensitive |
| Merck | 14,2693 |
| BRN | 1697296 |
| Exposure limits | No exposure limit is set. However, on the basis of the exposure limits of related compounds a ceiling limit of 0.5 ppm (2 mg/m3) is recommended. |
| Stability | Stable. Reacts violently with water and with mineral and organic acids. |
| CAS DataBase Reference | 506-68-3(CAS DataBase Reference) |
| NIST Chemistry Reference | Cyanogen bromide(506-68-3) |
| EPA Substance Registry System | Cyanogen bromide (506-68-3) |
SAFETY
Risk and Safety Statements
| Symbol(GHS) |    GHS05,GHS06,GHS09 |
|||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Signal word | Danger | |||||||||
| Hazard statements | H300+H310+H330-H314-H400 | |||||||||
| Precautionary statements | P260-P273-P280-P303+P361+P353-P304+P340+P310-P305+P351+P338 | |||||||||
| Hazard Codes | T+,N,F,C | |||||||||
| Risk Statements | 26/27/28-34-50/53-40-11-36/37-32-51/53 | |||||||||
| Safety Statements | 53-28-36/37/39-45-60-61-26-16-7/9-29-7 | |||||||||
| RIDADR | UN 3390 6.1/PG 1 | |||||||||
| WGK Germany | 3 | |||||||||
| RTECS | GT2100000 | |||||||||
| F | 8-17-19-21 | |||||||||
| TSCA | Yes | |||||||||
| HazardClass | 6.1 | |||||||||
| PackingGroup | I | |||||||||
| HS Code | 28530090 | |||||||||
| Toxicity | LCLO inhal (human) 92 ppm (398 mg/m3; 10 min) LCLO inhal (mouse) 115 ppm (500 mg/m3; 10 min) |
|||||||||
| NFPA 704 |
|
Cyanogen bromide price More Price(10)
| Manufacturer | Product number | Product description | CAS number | Packaging | Price | Updated | Buy |
|---|---|---|---|---|---|---|---|
| Sigma-Aldrich(India) | 8.20194 | Cyanogen bromide for synthesis | 506-68-3 | 250G | ₹27660 | 2022-06-14 | Buy |
| Sigma-Aldrich(India) | 8.20193 | Cyanogen bromide for synthesis | 506-68-3 | 50G | ₹11410 | 2022-06-14 | Buy |
| Sigma-Aldrich | 261610 | Cyanogen bromide solution 5.0?M in acetonitrile | 506-68-3 | 100ML | ₹8160 | 2022-06-14 | Buy |
| Sigma-Aldrich(India) | 261610 | Cyanogen bromide solution 5.0?M in acetonitrile | 506-68-3 | 100ML | ₹8160 | 2022-06-14 | Buy |
| SRL | 65291 | Cyanogen Bromide pure, 98% | 506-68-3 | 25Gms | ₹850 | 2022-05-26 | Buy |
Cyanogen bromide Chemical Properties,Uses,Production
Chemical Properties
Cyanogen bromide is a colorless or white, volatile, crystalline solid with a penetrating odor.
Uses
Cyanogen bromide is used in organic synthesis and as a reagent in bioanalysis.
Production Methods
Cyanogen bromide may be prepared by either the action of bromine on potassium cyanide or the interaction of sodium bromide, sodium cyanide, sodium chlorate, and sulfuric acid.
General Description
Cyanogen bromide is a colorless to white crystalline solid with a penetrating odor. Cyanogen bromide is slightly soluble in water. Cyanogen bromide is gradually decomposed by water and very rapidly by acids to give off hydrogen bromide, a flammable and poisonous gas. Contamination with many materials can cause rapid decomposition of the material. Cyanogen bromide is toxic by inhalation of its vapors or by the hydrogen cyanide from decomposition or by ingestion. Toxic oxides of nitrogen are produced in fire involving Cyanogen bromide. Cyanogen bromide is used in gold extraction, to make other chemicals, and as a fumigant.
Air & Water Reactions
Cyanogen bromide is slightly soluble in water. Cyanogen bromide is gradually decomposed by water and very rapidly by acids to give off hydrogen bromide, a poison gas.
Reactivity Profile
Cyanogen bromide is not combustible itself, but impure Cyanogen bromide decomposes rapidly and tends to explode. A violent reaction may take place on contact with large quantities of acid. Avoid physical damage, contact with acids or water, and store away from a location where water may be needed for fire control. [EPA, 1998]. Benzene and cyanogen halides yield HCl as a byproduct (Hagedorn, F. H. Gelbke, and Federal Republic of Germany. 2002. Nitriles. In Ullman Encyclopedia of Industrial Chemistry. Wiley-VCH Verlag GmbH & Co. KGaA.).
Health Hazard
Exposure to cyanogen bromide is dangerous. The chemical substance is poisonous and causes fatal injury if swallowed, inhaled, or absorbed through the skin. It is corrosive and the vapors cause severe irritation to the eyes and respiratory tract, and cause burns to any area of contact. On contact with acids, cyanogen bromide liberates poisonous gas, affecting the blood, cardiovascular system, CNS, and thyroid.
Fire Hazard
Cyanogen bromide is not combustible itself, but impure Cyanogen bromide decomposes rapidly and tends to explode. A violent reaction may take place on contact with large quantities of acid. Vapors are highly irritating. When material is heated to decomposition, Cyanogen bromide emits very toxic fumes of cyanide and bromide. Avoid water, acids. Avoid physical damage, contact with acids or water, and store away from a location where water may be needed for fire control.
Flammability and Explosibility
Cyanogen bromide is noncombustible. Impure material decomposes rapidly and can be explosive.
Safety Profile
A human and experimental poison by inhalation. Corrosive. When heated to decomposition it emits very toxic fumes of CNand Br-. Possibly unstable. See also other cyanogen entries; CYANIDE; and BROMIDES.
Potential Exposure
Used as an activating reagent for insoluble supports for affinity absorption. In danger are those manufacturing this compound or using it in organic synthesis or as a fumigant; in textile treatment; in gold cyaniding. It may have been used as a military poison gas.
storage
work with BrCN should be conducted in a fume hood to prevent exposure by inhalation, and splash goggles and impermeable gloves should be worn at all times to prevent eye and skin contact. Containers of cyanogen bromide should be kept tightly sealed and stored under nitrogen in a secondary container in a refrigerator.
Shipping
UN1889 Cyanogen bromide, Hazard class: 6.1; Labels: 6.1-Poisonous materials, 8-Corrosive material. A DOT regulated marine pollutant.
Purification Methods
All operations with this substance should be performed in a very efficient fume cupboard-it is very POISONOUS and should be handled in small amounts. Fresh commercial material is satisfactory for nearly all purposes and does not need to be purified. It is a white crystalline solid with a strong cyanide odour. If it is reddish in colour and partly liquid or paste-like, then it is too far gone to be purified, and fresh material should be sought. It can be purified by distillation using small amounts at a time, and using a short wide-bore condenser because it readily solidifies to a crystalline white solid which may clog the condenser. An appropriate gas mask should be used when transferring the molten solid from one container to another, and the operation should be done in an efficient fume cupboard. The melting point (m 49-51o) should be measured in a sealed tube. [Hartman & Dreger Org Synth Coll Vol II 150 1948.]
Incompatibilities
May be unstable unless dry and pure; impure cyanogen bromide decomposes rapidly and tends to explode. Cyanogen bromide decomposes violently on heat- ing or on contact with water, acids, or acid vapors; produc- ing highly toxic and flammable hydrogen cyanide and corrosive hydrogen bromide. Avoid physical damage, con- tact with acids or water, and store away from a location where water may be needed for fire control . Violent reaction with ammonia, amines.
Waste Disposal
May be added to strong alka- line solution of calcium hypochlorite, let stand for 24 hours and flush to sewer. May also be dissolved in flammable solvent and sprayed into an incinerator equipped with after- burner and scrubber.
Cyanogen bromide Preparation Products And Raw materials
Raw materials
Preparation Products
1of4
chevron_right| Supplier | Tel | Country | ProdList | Advantage | Inquiry |
|---|---|---|---|---|---|
| GLR Innovations | +91 9891111994 | New Delhi, India | 4542 | 58 | Inquiry |
| ALS INDIA LIFE SCIENCES | +91-8977036379 +91-8977036379 | Hyderabad, India | 1879 | 58 | Inquiry |
| Purecha Group (Sonal Plasrub Industries Pvt Ltd) | +91-2240949499 +91-2240949499 | Maharashtra, India | 34 | 58 | Inquiry |
| REDDY N REDDY PHARMACEUTICALS | +91-9848096759 +91-9848096759 | Hyderabad, India | 1989 | 58 | Inquiry |
| JSK Chemicals | +919879767970 | Gujarat, India | 3756 | 58 | Inquiry |
| SKS FINE CHEM PVT LTD | +91-9923051674 +91-9923051674 | Maharashtra, India | 17 | 58 | Inquiry |
| Otto Chemie Pvt. Ltd. | +91 9820041841 | Mumbai, India | 5873 | 58 | Inquiry |
| Sisco Research Laboratories Pvt. Ltd. | +91-22-4268 5800 | Mumbai, India | 4317 | 58 | Inquiry |
| Alfa Aesar | 1 800 209 7001 | Maharashtra, India | 6913 | 58 | Inquiry |
| Central Drug House(P) Ltd. | 91-11-49404040 | New Delhi, India | 6160 | 58 | Inquiry |
Related articles
- Cyanogen bromide-Hazard and Toxicity
- Cyanogen bromide is a colorless to white crystalline solid with a penetrating odor. Cyanogen bromide is slightly soluble in wa....
- Sep 12,2019





