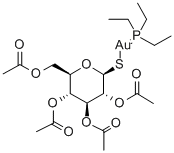
Auranofin
- CAS No.
- 34031-32-8
- Chemical Name:
- Auranofin
- Synonyms
- Adtil;ridaura;Auranon;Crisofin;Crisinor;Ridauran;AURANOFIN;sk&f39162;AURANOFINE;sk&f39162d
- CBNumber:
- CB5363410
- Molecular Formula:
- C20H34AuO9PS
- Molecular Weight:
- 678.48
- MOL File:
- 34031-32-8.mol
- Modify Date:
- 2023/8/30 13:35:17
SAFETY
Risk and Safety Statements
| Symbol(GHS) |   GHS06,GHS08 |
|||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Symbol(GHS) |   GHS06,GHS08 |
|||||||||
| Signal word | Danger | |||||||||
| Signal word | Danger | |||||||||
| Hazard statements | H301-H361 | |||||||||
| Hazard statements | H301-H361 | |||||||||
| Precautionary statements | P201-P202-P264-P270-P280-P301+P310 | |||||||||
| Precautionary statements | P201-P202-P264-P270-P280-P301+P310 | |||||||||
| Hazard Codes | Xn | |||||||||
| Risk Statements | 63-22 | |||||||||
| Safety Statements | 36/37 | |||||||||
| RIDADR | UN 2811 6.1 / PGIII | |||||||||
| WGK Germany | 3 | |||||||||
| RTECS | MD6500000 | |||||||||
| Toxicity | LD50 in rats, mice (mg/kg): 265, 310 orally (Payne, Walz) | |||||||||
| NFPA 704 |
|
Auranofin price More Price(4)
| Manufacturer | Product number | Product description | CAS number | Packaging | Price | Updated | Buy |
|---|---|---|---|---|---|---|---|
| Sigma-Aldrich(India) | A6733 | Auranofin ≥98% (HPLC) | 34031-32-8 | 10MG | ₹10966.8 | 2022-06-14 | Buy |
| Sigma-Aldrich(India) | A6733 | Auranofin ≥98% (HPLC) | 34031-32-8 | 50MG | ₹43567.5 | 2022-06-14 | Buy |
| ottokemi | A 3305 | Auranofin, for HPLC ≥98% | 34031-32-8 | 10mg | ₹10602 | 2022-05-26 | Buy |
| ottokemi | A 3305 | Auranofin, for HPLC ≥98% | 34031-32-8 | 50mg | ₹42309 | 2022-05-26 | Buy |
Auranofin Chemical Properties,Uses,Production
Description
Auranofin is the first orally effective gold compound to be marketed for the treatment of severe rheumatoid arthritis. It is better tolerated and more convenient than gold sodium thiomalate, which is administered intramuscularly.
Chemical Properties
White to Off-White Solid
Uses
Auranofin is a new oral gold-based antiarthritis drug. Auranofin inhibits various leukocyte activation pathways at multiple sites. Auranofin inhibits the release of inflammatory mediators from human m acrophages, basophils, and pulmonary mast cells. Auranofin is an efficient inducer of mitochondrial membrane permeability transition pore in the presence of calcium ions related to its inhibition of m itochondrial thioredoxin reductase.
Definition
ChEBI: An S-glycosyl compound consisting of 2,3,4,6-tetra-O-acetyl-1-thio-beta-D-glucopyranose with the sufur atom coordinated to (triethylphosphoranylidene)gold. It is administered orally fo the treatment of active progressive rheumatoid arthritis.
brand name
Ridaura (Promethus, Smith Kline Beecham, USA), Aktil (Lek, Yugoslavia), Ridauran (Robapharm, France).
Pharmaceutical Applications
Auranofin ([tetra-O-acetyl-β-D- (glucopyranosyl)thio]-triethylphosphine)gold(I) is a second-generation gold-based drug, licensed as an orally available gold drug for the treatment of RA. It features a linear S Au P geometry, as shown by X-ray analysis. It is more lipophilic than the first-generation drugs, which makes oral administration possible. Treatment with Auranofin requires less visits to the clinic, but it is believed to be less successful in the treatment of RA compared to gold drugs being administered intramuscularly.
Clinical Use
Auranofin is indicated in adults with active rheumatoid arthritis who have not responded sufficiently to one or more NSAIDs.
Safety Profile
Poison by ingestion,intraperitoneal, and intravenous routes. Human systemiceffects by ingestion: ulceration or bleeding from stomach.An experimental teratogen. Other experimentalreproductive effects. Human mutation data reported.When heated to decomp
Synthesis
Synthesis: ethanolic thiodiglycol is treated
first with aqueous gold(I) acid chloride trihydrate, then with ethanolic triethylphosphine to
give triethylphosphine gold(I) chloride, which is
added to an aqueous solution of S-(2,3,4,6-tetra-O-acetylglucopyranosyl)pseudothiourea hydrobromide and potassium carbonate to give the desired auranofin.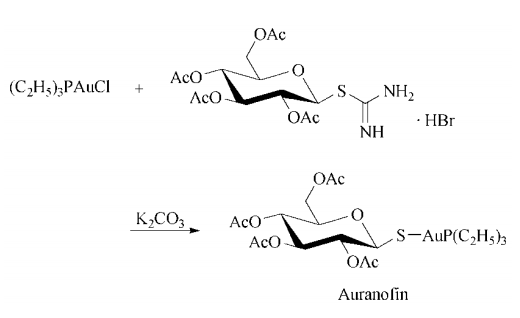
Metabolism
On a mg gold/kg basis, it is reported to be as effective in the rat adjuvant arthritis assay as the parenterally effective drugs. Daily oral doses produce a rapid increase in kidney and blood gold levels for the first 3 days of treatment, with a more gradual increase on subsequent administration. Plasma gold levels are lower than those attained with parenteral gold compounds. The major route of excretion is via the urine. Auranofin may produce fewer adverse reactions than parenteral gold compounds, but its therapeutic efficacy also may be less.
Auranofin Preparation Products And Raw materials
Raw materials
1of2
chevron_rightPreparation Products
| Supplier | Tel | Country | ProdList | Advantage | Inquiry |
|---|---|---|---|---|---|
| CLEARSYNTH LABS LTD. | +91-22-45045900 | Hyderabad, India | 6351 | 58 | Inquiry |
| Otto Chemie Pvt. Ltd. | +91 9820041841 | Mumbai, India | 5873 | 58 | Inquiry |
| Hebei Mojin Biotechnology Co., Ltd | +8613288715578 | China | 12458 | 58 | Inquiry |
| Shanghai Daken Advanced Materials Co.,Ltd | +86-371-66670886 | China | 16209 | 58 | Inquiry |
| Henan Tianfu Chemical Co.,Ltd. | +86-0371-55170693 +86-19937530512 | China | 21669 | 55 | Inquiry |
| Hebei Guanlang Biotechnology Co., Ltd. | +86-19930503282 | China | 8826 | 58 | Inquiry |
| BOC Sciences | +1-631-485-4226 | United States | 19553 | 58 | Inquiry |
| CONIER CHEM AND PHARMA LIMITED | +8618523575427 | China | 49391 | 58 | Inquiry |
| career henan chemical co | +86-0371-86658258 +8613203830695 | China | 29826 | 58 | Inquiry |
| TargetMol Chemicals Inc. | +1-781-999-5354 +1-00000000000 | United States | 19892 | 58 | Inquiry |
34031-32-8(Auranofin)Related Search:
1of4
chevron_right




