Calcium cyanamide
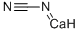
- CAS No.
- 156-62-7
- Chemical Name:
- Calcium cyanamide
- Synonyms
- Calcium carbimide;nitrolime;Metformin Impurity 8;dormex;alzodef;ccc[qr];cy-l500;nitrolim;usafcy-2;nci-c02937
- CBNumber:
- CB0297271
- Molecular Formula:
- CCaN2
- Molecular Weight:
- 80.1
- MOL File:
- 156-62-7.mol
- MSDS File:
- SDS
- Modify Date:
- 2024/3/14 15:18:25
| Melting point | >300 °C(lit.) |
|---|---|
| Density | 2.29 |
| vapor pressure | 0.51Pa at 20℃ |
| solubility | reacts with H2O |
| form | Powder |
| color | Gray to dark gray |
| Specific Gravity | 2.29 |
| Water Solubility | insoluble H2O, but undergoes hydrolysis releasing acetylene and ammonia [HAW93] [MER06] |
| Sensitive | Moisture Sensitive |
| Merck | 14,1662 |
| BRN | 4124391 |
| InChIKey | QFSRQFUIHVTIDL-UHFFFAOYSA-N |
| LogP | -0.72 at 20℃ |
| CAS DataBase Reference | 156-62-7(CAS DataBase Reference) |
| EPA Substance Registry System | Calcium cyanamide (156-62-7) |
SAFETY
Risk and Safety Statements
| Symbol(GHS) |    GHS02,GHS05,GHS07 |
|||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Signal word | Danger | |||||||||
| Hazard statements | H261-H302-H315-H317-H318-H335 | |||||||||
| Precautionary statements | P231+P232-P280-P301+P312+P330-P304+P340+P312-P305+P351+P338+P310-P333+P313 | |||||||||
| Hazard Codes | Xn,F | |||||||||
| Risk Statements | 22-37-41-15-43-37/38 | |||||||||
| Safety Statements | 26-39-43-36/37/39-22-45-28 | |||||||||
| RIDADR | UN 1403 4.3/PG 3 | |||||||||
| OEB | C | |||||||||
| OEL | TWA: 0.5 mg/m3 | |||||||||
| WGK Germany | 2 | |||||||||
| RTECS | GS6000000 | |||||||||
| F | 9-21 | |||||||||
| TSCA | Yes | |||||||||
| HazardClass | 4.3 | |||||||||
| PackingGroup | III | |||||||||
| HS Code | 31029010 | |||||||||
| NFPA 704 |
|
Calcium cyanamide price More Price(4)
| Manufacturer | Product number | Product description | CAS number | Packaging | Price | Updated | Buy |
|---|---|---|---|---|---|---|---|
| Sigma-Aldrich | 438928 | Calcium cyanamide technical grade | 156-62-7 | 500G | ₹9774.98 | 2022-06-14 | Buy |
| Sigma-Aldrich(India) | 438928 | Calcium cyanamide technical grade | 156-62-7 | 500G | ₹9774.98 | 2022-06-14 | Buy |
| ALFA India | ALF-089462-36 | Calcium cyanamide, tech. | 156-62-7 | 500g | ₹6585 | 2022-05-26 | Buy |
| ALFA India | ALF-089462-22 | Calcium cyanamide, tech. | 156-62-7 | 100g | ₹2459 | 2022-05-26 | Buy |
Calcium cyanamide Chemical Properties,Uses,Production
Chemical Properties
Calcium cyanamide is a blackish-gray, shiny crystalline material or powder.
Physical properties
Pure product is a colorless, hexagonal crystal or white powder. Commercial grade material may be grayish-black powder or lump (the color is due to presence of calcium carbide and other impurities); density 2.29 g/cm3; melts around 1,340°C; sublimes around 1,150 to 1,200°C on rapid heating; reacts with water.
Uses
Calcium Cyanamide is used as a fertilizer, herbicide, insecticide, a steel-making additive and an ore processing material. It can also be used to make thiourea, guanidine and ferrocyanides. manufacture of calcium cyanide, melamine, dicyandiamide.
Production Methods
Calcium cyanamide was first produced commercially around 1900 as a fertilizer. The process of making calcium cyanamide involves three raw materials—coke, coal, and limestone— plus nitrogen. The limestone (calcium carbonate) is burned with coal to produce calcium oxide. The calcium oxide is then allowed to react with amorphous carbon in the furnace at 2000°C with the formation of calcium carbide (CaC2). Finely powdered calcium carbide is heated to 1000°C in an electric furnace into which pure nitrogen is passed. It is then removed and uncombined calcium carbide removed by leaching.
Definition
calcium cyanamide: A colourlesssolid, CaCN2, which sublimes at1300°C. It is prepared by heating calciumdicarbide at 800°C in a streamof nitrogen:
CaC2(s) + N2(g) → CaCN2(s) + C(s)
The reaction has been used as amethod of fixing nitrogen in countriesin which cheap electricity isavailable to make the calcium dicarbide(the cyanamide process). Calciumcyanamide can be used as afertilizer because it reacts with waterto give ammonia and calcium carbonate:
CaCN2(s) + 3H2O(l) → CaCO3(s) +2NH3(g)
It is also used in the production ofmelamine, urea, and certain cyanidesalts.
Preparation
Calcium cyanamide is prepared from calcium carbide. The carbide powder is heated at about 1,000°C in an electric furnace into which nitrogen is passed for several hours. The product is cooled to ambient temperatures and any unreacted carbide is leached out cautiously with water.
CaC2 + N2 → CaCN2 + C (ΔHƒ°= –69.0 kcal/mol at 25°C)
General Description
A colorless to gray, odorless solid. May cause illness from ingestion. May irritate the skin. If exposed to water or high temperatures, calcium cyanamide may generate toxic and flammable fumes. Used to make pesticides and in fertilizers.
Air & Water Reactions
Depending on the calcium carbide content, the cyanamide reacts with water (moisture from air or soil) to produce acetylene and hydrated calcium oxide or calcium hydroxide. Absorption of water during handling or storage of technical calcium cyanamide may cause explosion [Pieri, M. Chem. Abs. 46, 8335 1952].
Reactivity Profile
When hydrated CALCIUM CARBIDE generates salts of calcium that are basic and are generally soluble in water. The resulting solutions contain moderate concentrations of hydroxide ions and have pH's greater than 7.0. They react as bases to neutralize acids. These neutralizations generate heat, but less or far less than is generated by neutralization of the bases in reactivity group 10 (Bases) and the neutralization of amines. They usually do not react as either oxidizing agents or reducing agents but such behavior is not impossible.
Hazard
Fire risk with moisture or combined with calcium carbide. Skin, eye, and upper respiratory tract irritant. Questionable carcinogen.
Health Hazard
Inhalation or contact with vapors, substance or decomposition products may cause severe injury or death. May produce corrosive solutions on contact with water. Fire will produce irritating, corrosive and/or toxic gases. Runoff from fire control may cause pollution.
Fire Hazard
Produce flammable gases on contact with water. May ignite on contact with water or moist air. Some react vigorously or explosively on contact with water. May be ignited by heat, sparks or flames. May re-ignite after fire is extinguished. Some are transported in highly flammable liquids. Runoff may create fire or explosion hazard.
Safety Profile
Poison by ingestion, inhalation, sh contact, intravenous, and intraperitoneal routes. Moderately toxic to humans by ingestion. Questionable carcinogen with experimental tumorigenic data. Mutation data reported. The fatal dose, by ingestion, is probably around 20 to 30 g for an adult. It does not have a cyanide effect. Calcium cyanamide is not believed to have a cumulative action. Flammable. Reaction with water forms the explosive acetylene gas. When heated to decomposition it emits toxic fumes of NOx and CN-. See also CALCIUM COMPOUNDS, AMIDES, and CYANIDE
Potential Exposure
Calcium cyanamide is used in agriculture as a fertilizer, herbicide; defoliant for cotton plants; and pesticide. It is also used in the manufacture of dicyandiamide and calcium cyanide as a desulfurizer in the iron and steel industry; and in steel hardening.
Carcinogenicity
Calcium cyanamide was weakly mutagenic in Salmonella typhimurium strain TA1535 and nonmutagenic in strain TA100.
Shipping
UN1403 Calcium cyanamide with .1% calcium carbide, Hazard Class: 4.3; Labels: 4.3-Dangerous when wet material
Incompatibilities
Commercial grades of calcium cyanamide may contain calcium carbide; contact with any form of moisture solutions may cause decomposition, liberating explosive acetylene gas and ammonia. Incompatible with oxidizers (chlorates, nitrates, peroxides, permanganates, perchlorates, chlorine, bromine, fluorine, etc.); contact may cause fires or explosions. Keep away from alkaline materials, strong bases, strong acids, oxoacids, epoxides. May polymerize in water or alkaline solutions to dicyanamide. Contact with all solvents tested also causes decomposition
Calcium cyanamide Preparation Products And Raw materials
Raw materials
Preparation Products
1of4
chevron_right| Supplier | Tel | Country | ProdList | Advantage | Inquiry |
|---|---|---|---|---|---|
| TCI Chemicals (India) Pvt. Ltd. | 1800 425 7889 | New Delhi, India | 6778 | 58 | Inquiry |
| Alfa Aesar | 1 800 209 7001 | Maharashtra, India | 6913 | 58 | Inquiry |
| SynZeal Research Pvt Ltd | +1 226-802-2078 | Gujarat, India | 6522 | 58 | Inquiry |
| Triveni chemicals | 08048762458 | New Delhi, India | 6093 | 58 | Inquiry |
| Associated Agencies - Division Of Dorado Chem Private... | 08048973456 | Mumbai, India | 14 | 58 | Inquiry |
| CLEARSYNTH LABS LTD. | +91-22-45045900 | Hyderabad, India | 6351 | 58 | Inquiry |
| Hebei Mojin Biotechnology Co., Ltd | +8613288715578 | China | 12452 | 58 | Inquiry |
| Henan Tianfu Chemical Co.,Ltd. | +86-0371-55170693 +86-19937530512 | China | 21669 | 55 | Inquiry |
| SHANDONG ZHI SHANG CHEMICAL CO.LTD | +86 18953170293 | China | 2931 | 58 | Inquiry |
| Hebei Guanlang Biotechnology Co., Ltd. | +86-19930503282 | China | 8822 | 58 | Inquiry |
156-62-7(Calcium cyanamide)Related Search:
1of4
chevron_right




