Mitoxantrone hydrochloride
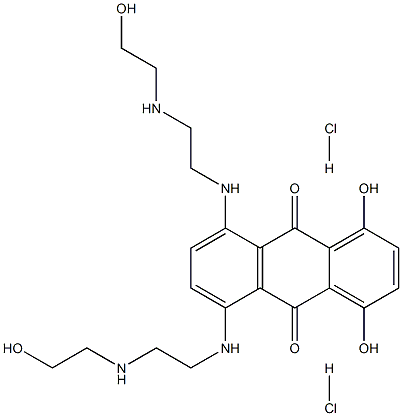
- CAS No.
- 70476-82-3
- Chemical Name:
- Mitoxantrone hydrochloride
- Synonyms
- MITOXANTRONE HCL;novantrone;DHAQ;DHAD;Mitozantrone;IMMunex;cl232315;Novantron;Novatrone;Bisantrone
- CBNumber:
- CB4246424
- Molecular Formula:
- C22H30Cl2N4O6
- Molecular Weight:
- 517.4028
- MOL File:
- 70476-82-3.mol
- MSDS File:
- SDS
- Modify Date:
- 2024/7/2 8:55:08
| Melting point | 203-205 C |
|---|---|
| storage temp. | 2-8°C |
| solubility | Sparingly soluble in water, slightly soluble in methanol, practically insoluble in acetone. |
| form | Solid |
| color | Dark Blue to Black |
| Water Solubility | Soluble to 5 mM in water and to 75 mM in DMSO |
| InChI | InChI=1S/C22H28N4O6.2ClH/c27-11-9-23-5-7-25-13-1-2-14(26-8-6-24-10-12-28)18-17(13)21(31)19-15(29)3-4-16(30)20(19)22(18)32;;/h1-4,23-30H,5-12H2;2*1H |
| InChIKey | ZAHQPTJLOCWVPG-UHFFFAOYSA-N |
| SMILES | C1(O)=C2C(C(=O)C3=C(C2=O)C(NCCNCCO)=CC=C3NCCNCCO)=C(O)C=C1.[H]Cl.[H]Cl |
| CAS DataBase Reference | 70476-82-3(CAS DataBase Reference) |
| EPA Substance Registry System | Mitoxantrone hydrochloride (70476-82-3) |
SAFETY
Risk and Safety Statements
| Symbol(GHS) |  GHS08 |
|||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Signal word | Danger | |||||||||
| Hazard statements | H340-H360 | |||||||||
| Precautionary statements | P201-P280-P308+P313 | |||||||||
| Hazard Codes | T,T+ | |||||||||
| Risk Statements | 46-61-26/27/28 | |||||||||
| Safety Statements | 53-36/37/39-45-22 | |||||||||
| RIDADR | 3249 | |||||||||
| WGK Germany | 3 | |||||||||
| RTECS | CB5748500 | |||||||||
| HazardClass | 6.1(b) | |||||||||
| PackingGroup | III | |||||||||
| HS Code | 29146100 | |||||||||
| NFPA 704 |
|
Mitoxantrone hydrochloride price More Price(4)
| Manufacturer | Product number | Product description | CAS number | Packaging | Price | Updated | Buy |
|---|---|---|---|---|---|---|---|
| Sigma-Aldrich(India) | M6545 | Mitoxantrone dihydrochloride ≥97% (HPLC) | 70476-82-3 | 10MG | ₹5910.45 | 2022-06-14 | Buy |
| Sigma-Aldrich(India) | M6545 | Mitoxantrone dihydrochloride ≥97% (HPLC) | 70476-82-3 | 50MG | ₹18012.8 | 2022-06-14 | Buy |
| TCI Chemicals (India) | M3110 | Mitoxantrone Dihydrochloride | 70476-82-3 | 25MG | ₹4400 | 2022-05-26 | Buy |
| TCI Chemicals (India) | M3110 | Mitoxantrone Dihydrochloride | 70476-82-3 | 100MG | ₹9900 | 2022-05-26 | Buy |
Mitoxantrone hydrochloride Chemical Properties,Uses,Production
Description
Mitoxantrone hydrochloride is the first of the synthetic anthracenediones related to doxorubicin to reach the marketplace. Mitoxantrone is useful in the treatment of advanced localized and metastatic mammary carcinomas. It is reported to be less cardiotoxic than doxorubicin.
Chemical Properties
Dark blue, electrostatic, hygroscopic powder.
Uses
analgesic, antipyretic
General Description
Mitoxantrone is supplied as a blue aqueous solution in 10-and 20-mg vials for IV administration in the treatment of acute lymphoid leukemia, acute myeloid leukemia, breastcancer, prostate cancer, non-Hodgkin’s lymphoma, andmultiple sclerosis. The mechanisms of resistance are thesame as those seen for the anthracyclines. The distributionhalf-life is 1.1 to 3.1 hours, and the drug has a large volumeof distribution (11 L/kg). The elimination half-life rangedfrom 23 to 215 hours, and elimination was primarily via thebile. Metabolism of the agent involves oxidation of the sidechainalcohols to give the monocarboxylic and dicarboxylicacids.Other toxicities are those seen for the anthracyclinesand include myelosuppression, nausea, vomiting, mucositis,diarrhea, and alopecia. The intense color of the parent drugand metabolites may turn the urine blue.
Biological Activity
Mitoxantrone hydrochloride (Mitoxantrone dihydrochloride) is a type II DNA topoisomerase inhibitor. Disrupts DNA synthesis and repair and induces damage by DNA cross-linking. Also inhibits PIM1 kinase (IC50 = 51 nM). Immunomodulatory, antineoplastic and chemotherapeutic agent. Also USP11 inhibitor (IC50= 3.15 μM). Induces cell death of pancreatic cancer cell lines expressing wild-type BRCA2.
Clinical Use
Mitoxantrone is used in combination with other agents during the initial treatment of acute nonlymphocytic leukemia and hormone-refractory prostate cancer. Recent studies have shown that mitoxantrone also decreases the rate of relapse and disease progression in patients with multiple sclerosis. Although too toxic for use in patients with primary progressive disease, it is available for the treatment of chronic progressive, progressive relapsing, or deteriorating relapsing-remitting multiple sclerosis.
Side effects
Common side effects of Mitoxantrone hydrochloride include: Swelling of the body; Infection, possibly in the blood, especially when white blood cell count is low; Sores in mouth and throat which may cause difficulty swallowing; Diarrhea, nausea, vomiting; Headache; Pain in belly, back, joints, or muscles; Abnormal or absence of menstrual period; Tiredness, fever; Hair loss; Hives, rash. Less common side effects include: Damage to the heart or heart failure which may cause shortness of breath, tiredness and swelling; Bruising, bleeding; Anemia which may cause tiredness, or may require transfusion; Liver damage which may cause yellowing of eyes and skin, swelling; Internal bleeding which may cause belly pain, black tarry stool, blood in vomit; Constipation, loss of appetite; Abnormal sexual function; Worry, depression; Swelling and redness at the site of the injection; Changes in weight; Swelling and redness of the whites of the eye; Increased sweating; Loss of nails, darkening of the skin and nails; Bluish/greenish discoloration of the urine, skin, eyes and saliva. A rare side effect is bone marrow cancer (leukaemia) caused by chemotherapy.
Metabolism
Mitoxantrone excretion primarily is biliary. Both the unchanged drug and inactive metabolites resulting from N-dealkylation, deamination, and oxidation of the resultant aldehyde to the carboxylic acid are observed. Both arms of the structure can be metabolized, leading to mono- or dicarboxylic acid metabolites, which are excreted as the glucuronide conjugate. The conjugated metabolites are an intense, dark blue in color and will result in blue-green urine.
Mitoxantrone hydrochloride Preparation Products And Raw materials
Raw materials
Preparation Products
| Supplier | Tel | Country | ProdList | Advantage | Inquiry |
|---|---|---|---|---|---|
| Ralington Pharma | +91-7948911722 +91-9687771722 | Gujarat, India | 1350 | 58 | Inquiry |
| Surajlok Chemicals Pvt Ltd (Neon Laboratories Ltd) | +91-2268107000 +91-9987232509 | Maharashtra, India | 30 | 58 | Inquiry |
| OCEAN TRADING CORPORATION | +91(22) 24921669 | New Delhi, India | 6211 | 58 | Inquiry |
| Manus Aktteva | +91 (79) 6512-3395 | New Delhi, India | 581 | 34 | Inquiry |
| CLEARSYNTH LABS LTD. | +91-22-45045900 | Hyderabad, India | 6351 | 58 | Inquiry |
| CHEMSWORTH | +91-261-2397244 | New Delhi, India | 6707 | 30 | Inquiry |
| A.J Chemicals | 91-9810153283 | New Delhi, India | 6124 | 58 | Inquiry |
| TCI Chemicals (India) Pvt. Ltd. | 1800 425 7889 | New Delhi, India | 6778 | 58 | Inquiry |
| Pharmaffiliates Analytics and Synthetics P. Ltd | +91-172-5066494 | Haryana, India | 6773 | 58 | Inquiry |
| SynZeal Research Pvt Ltd | +1 226-802-2078 | Gujarat, India | 6522 | 58 | Inquiry |
70476-82-3(Mitoxantrone hydrochloride)Related Search:
1of4
chevron_right




