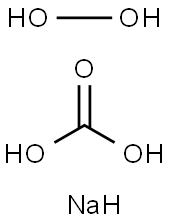
Sodium percarbonate
- CAS No.
- 15630-89-4
- Chemical Name:
- Sodium percarbonate
- Synonyms
- PCS;SODIUM CARBONATE PEROXYHYDRATE;SODIUM CARBONATE PEROXIDE;SODIUM CARBONATE HYDROGEN PEROXIDE;SODIUM PERCARBONATE 13% AVAILABLE OXYGEN;ECOX-C;oxyper;perdox;OxiClean;Oxyper S 142
- CBNumber:
- CB4853147
- Molecular Formula:
- CH5NaO5
- Molecular Weight:
- 120.04
- MOL File:
- 15630-89-4.mol
- MSDS File:
- SDS
- Modify Date:
- 2024/5/16 14:11:51
| Density | 2.09[at 20℃] |
|---|---|
| vapor pressure | < 10-3 Pa at 25°C |
| form | Granular Powder |
| color | White |
| PH | About 10.5 at 1% concentration (20°C) |
| Odor | Odorless |
| Water Solubility | Soluble in water. |
| Sensitive | Moisture Sensitive |
| Decomposition | > 131 °F (> 55 °C) |
| LogP | -0.809 (est) |
| CAS DataBase Reference | 15630-89-4(CAS DataBase Reference) |
| EPA Substance Registry System | Sodium percarbonate (15630-89-4) |
SAFETY
Risk and Safety Statements
| Symbol(GHS) |    GHS03,GHS05,GHS07 |
|||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Signal word | Danger | |||||||||
| Hazard statements | H272-H302-H318 | |||||||||
| Precautionary statements | P210-P220-P264-P280-P301+P312-P305+P351+P338 | |||||||||
| Hazard Codes | O,Xn | |||||||||
| Risk Statements | 8-22-41-36/38 | |||||||||
| Safety Statements | 17-26-39-37/39 | |||||||||
| RIDADR | UN 1479 5.1/PG 3 | |||||||||
| WGK Germany | 1 | |||||||||
| RTECS | FG0750000 | |||||||||
| F | 21 | |||||||||
| TSCA | Yes | |||||||||
| HazardClass | 5.1 | |||||||||
| PackingGroup | II | |||||||||
| HS Code | 28369990 | |||||||||
| Toxicity | mouse,LD50,oral,2200mg/kg (2200mg/kg),BEHAVIORAL: SOMNOLENCE (GENERAL DEPRESSED ACTIVITY)LUNGS, THORAX, OR RESPIRATION: DYSPNEA,Toksikologicheskii Vestnik. Vol. (3), Pg. 46, 1994. | |||||||||
| NFPA 704 |
|
Sodium percarbonate price More Price(8)
| Manufacturer | Product number | Product description | CAS number | Packaging | Price | Updated | Buy |
|---|---|---|---|---|---|---|---|
| Sigma-Aldrich(India) | 371432 | Sodium percarbonate avail. H2O2 20-30?% | 15630-89-4 | 25G | ₹1472.2 | 2022-06-14 | Buy |
| Sigma-Aldrich(India) | 371432 | Sodium percarbonate avail. H2O2 20-30?% | 15630-89-4 | 500G | ₹2554.7 | 2022-06-14 | Buy |
| Sigma-Aldrich(India) | 371432 | Sodium percarbonate avail. H2O2 20-30?% | 15630-89-4 | 2.5KG | ₹4470.73 | 2022-06-14 | Buy |
| ALFA India | ALF-A16045-36 | Sodium percarbonate, 13-14% active oxygen | 15630-89-4 | 500g | ₹2530 | 2022-05-26 | Buy |
| ALFA India | ALF-A16045-22 | Sodium percarbonate, 13-14% active oxygen | 15630-89-4 | 100g | ₹2963 | 2022-05-26 | Buy |
Sodium percarbonate Chemical Properties,Uses,Production
Chemical Properties
Sodium percarbonate is a white granular powder of sodium carbonate and hydrogen peroxide.

Sodium percarbonate is mainly used as a bleaching chemical in laundry detergents (tablets, compact or regular powders), laundry additives and machine dishwashing products. It is an oxidizing agent and ingredient in a number of home and laundry cleaning products, including eco-friendly bleach products such as OxiClean and Tide laundry detergent. Dissolved in water, it releases hydrogen peroxide and soda ash (sodium carbonate):
2(Na2CO31.5H202)→2Na2CO3+3H202
Uses
Sodium percarbonate is an addition salt of hydrogen peroxide and sodium carbonate that provides a solid source of hydrogen peroxide. When dissolved in water, sodium percarbonate liberates hydrogen peroxide. Sodium percarbonate is a white, granular or powdered solid oxidizer. It is used primarily as a bleaching agent in cleaning products. Other uses include algaecides, fungicides, chemical synthesis and environmental applications such as control of odor at waste treatment facilities. A small amount is used in denture cleaners and toothpaste.
Multifunctional reagent for the preparation of optically active 4-hydroxy-2-isoxazolines.
Preparation
Sodium percarbonate is produced by the reaction of sodium carbonate with hydrogen peroxide, which can be done via dry, spray and wet processes. In the dry process aqueous hydrogen peroxide solution is sprayed on solid sodium carbonate; a solid-liquid reaction yields sodium percarbonate. In the spray process sodium percarbonate is produced by a fluid bed process. Solutions of sodium carbonate and hydrogen peroxide are sprayed into a drying chamber where the water is evaporated. In the wet process sodium percarbonate is usually prepared by cristallisation possibly in combination with salting out.
Reactions
Sodium percarbonate naturally decomposes, very slowly, to form sodium carbonate and hydrogen peroxide. The hydrogen peroxide may further decompose to form water and oxygen and liberate some heat. The decomposition proceeds according to the reaction below:
2Na2CO3 • 3H2O2 → 2Na2CO3 + 3H2O + 1.5 O2 + Heat
General Description
A colorless, crystalline solid. Denser than water. May combust in contact with organic materials. Contact may irritate skin, eyes and mucous membranes. May be toxic by ingestion. Used to make other chemicals.
Air & Water Reactions
Soluble in water.
Reactivity Profile
Oxidizing agents, such as SODIUM PERCARBONATE, can react with reducing agents to generate heat and products that may be gaseous (causing pressurization of closed containers). The products may themselves be capable of further reactions (such as combustion in the air). The chemical reduction of materials in this group can be rapid or even explosive, but often requires initiation (heat, spark, catalyst, addition of a solvent). Explosive mixtures of inorganic oxidizing agents with reducing agents often persist unchanged for long periods if initiation is prevented. Such systems are typically mixtures of solids, but may involve any combination of physical states. Some inorganic oxidizing agents are salts of metals that are soluble in water; dissolution dilutes but does not nullify the oxidizing power of such materials. Organic compounds, in general, have some reducing power and can in principle react with compounds in this class. Actual reactivity varies greatly with the identity of the organic compound. Inorganic oxidizing agents can react violently with active metals, cyanides, esters, and thiocyanates.
Health Hazard
Inhalation, ingestion or contact (skin, eyes) with vapors or substance may cause severe injury, burns or death. Fire may produce irritating, corrosive and/or toxic gases. Runoff from fire control or dilution water may cause pollution.
Fire Hazard
These substances will accelerate burning when involved in a fire. Some may decompose explosively when heated or involved in a fire. May explode from heat or contamination. Some will react explosively with hydrocarbons (fuels). May ignite combustibles (wood, paper, oil, clothing, etc.). Containers may explode when heated. Runoff may create fire or explosion hazard.
Safety Profile
Moderately toxic by ingestion. When heated to decomposition it emits acrid smoke and irritating vapors
Synthesis
Sodium percarbonate is prepared by the reaction of dihydrogen peroxide and sodium carbonate. The specific synthesis steps are as follows:
With florisil In water addn. of small amounts of MgSiO3 and water glass to 500kg H2O2 (dild. to 20% soln.), addn. of 1051kg Na2CO3 at 18°C, cooling to 5°C, cooling to -4°C after addn. of 500kg NaCl; centrifugation, drying, reuse of mother-liquor, finally sodium percarbonate (yield: 85.5%) is obtained.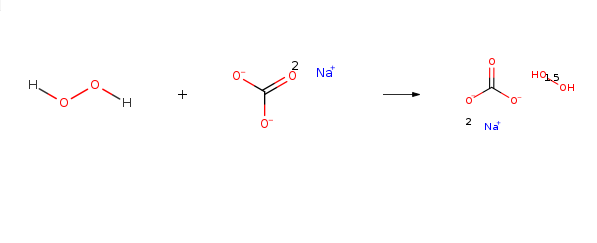
Environmental Fate
Sodium percarbonate is not persistent in the environment and readily decomposes to soda ash (sodium carbonate) and hydrogen peroxide which will subsequently decompose to water and oxygen when exposed to soils, sediments, and surface or ground waters.
Sodium percarbonate Preparation Products And Raw materials
Raw materials
1of2
chevron_rightPreparation Products
1of3
chevron_right| Supplier | Tel | Country | ProdList | Advantage | Inquiry |
|---|---|---|---|---|---|
| VITRAG CHEMICALS PVT LTD | +91-7490053194 +91-7490053194 | Gujarat, India | 47 | 58 | Inquiry |
| Belchem Industries Private Limited | +91-2224102288 +91-2224102278 | Maharashtra, India | 17 | 58 | Inquiry |
| Acuro Organics Limited | +91-8882777000 +91-8882777000 | New Delhi, India | 44 | 58 | Inquiry |
| FINAR LTD | +91 - 2717 - 616717 | New Delhi, India | 660 | 58 | Inquiry |
| Otto Chemie Pvt. Ltd. | +91 9820041841 | Mumbai, India | 5873 | 58 | Inquiry |
| Alfa Aesar | 1 800 209 7001 | Maharashtra, India | 6913 | 58 | Inquiry |
| LOBA CHEMIE PVT.LTD. | 91-22-6663 6699 | Mumbai, India | 3077 | 58 | Inquiry |
| Central Drug House(P) Ltd. | 91-11-49404040 | New Delhi, India | 6160 | 58 | Inquiry |
| Triveni chemicals | 08048762458 | New Delhi, India | 6093 | 58 | Inquiry |
| Thaker Brothers | 08048953166 | Nagpur, India | 2 | 58 | Inquiry |
| Supplier | Advantage |
|---|---|
| VITRAG CHEMICALS PVT LTD | 58 |
| Belchem Industries Private Limited | 58 |
| Acuro Organics Limited | 58 |
| FINAR LTD | 58 |
| Otto Chemie Pvt. Ltd. | 58 |
| Alfa Aesar | 58 |
| LOBA CHEMIE PVT.LTD. | 58 |
| Central Drug House(P) Ltd. | 58 |
| Triveni chemicals | 58 |
| Thaker Brothers | 58 |
Related articles
- Application of sodium peroxycarbonate
- Sodium percarbonate, referred to as SPC, commonly known as solid hydrogen peroxide, also known as sodium peroxycarbonate, soli....
- Jul 1,2021
Related Qustion
- Q:Is sodium percarbonate harmful to the human body?
- A:sodium percarbonate can be absorbed into the body through ingestion and may cause coughing, sore throat, burning sensation, an....
- Dec 27,2023
15630-89-4(Sodium percarbonate)Related Search:
1of4
chevron_right




