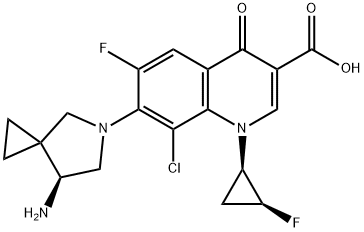
Sitafloxacin
|
- ₹0
- Product name: Sitafloxacin
- CAS: 127254-12-0
- MF: C19H18ClF2N3O3
- MW: 409.81
- EINECS:1308068-626-2
- MDL Number:MFCD00903720
- Synonyms:spifloxacin;7-[(4S)-4-Amino-6-azaspiro[2.4]heptan-6-yl]-8-chloro-6-fluoro-1-[(2S)-2-fluorocyclopropyl]-4-oxoquinoline-3-carboxylic acid;3-Quinolinecarboxylic acid, 7-[(7S)-7-aMino-5-azaspiro[2.4]hept-5-yl]-8-chloro-6-fluoro-1-[(1R,2S)-2-fluorocyclopropyl]-1,4-dihydro-4-oxo-;Sitafloxacin anhydrous;7-((S)-7-Amino-5-azaspiro[2.4]heptan-5-yl)-8-chloro-6-fluoro-1-((1R,2S)-2-fluorocyclopropyl)-4;Sitafloxacfn;Sitfloxacfn;Sitafloxacin-d4
| Manufacturer | Product number | Product description | Packaging | Price | Updated | Buy |
|---|
Properties
Boiling point :629.2±55.0 °C(Predicted)
Density :1.63±0.1 g/cm3(Predicted)
storage temp. :Store at -20°C
solubility :DMF: slightly soluble; DMSO: slightly soluble; Methanol: slightly soluble
form :A crystalline solid
pka :6.39±0.50(Predicted)
CAS DataBase Reference :127254-12-0(CAS DataBase Reference)
Density :1.63±0.1 g/cm3(Predicted)
storage temp. :Store at -20°C
solubility :DMF: slightly soluble; DMSO: slightly soluble; Methanol: slightly soluble
form :A crystalline solid
pka :6.39±0.50(Predicted)
CAS DataBase Reference :127254-12-0(CAS DataBase Reference)
Safety Information
| Symbol(GHS): |

|
|||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Signal word: | Warning | |||||||||||||||||||||||||||||||||||
| Hazard statements: |
|
|||||||||||||||||||||||||||||||||||
| Precautionary statements: |
|
Description
Sitafloxacin hydrate is the newest member (fourth generation) of the fluoroquinolone family of antibiotics that exhibits broad spectrum activity against many Gram-positive, Gramnegative, and anaerobic clinical isolates, including strains resistant to other fluoroquinolones. Since the launch of the first fluoroquinolone norfloxacin (patented in 1978), 20 other versions have made it to market with ciprofloxacin and levofloxacin experiencing the most prevalent usage. The mechanism of action involves inhibition of bacterial type II topoisomerases, both DNA gyrase and topoisomerase IV. By inhibiting these enzymes and preventing DNA supercoiling, cell division is disrupted leading to cell death. In Gram-negative bacteria, the primary target appears to be gyrase, whereas topoisomerase IV is involved in Gram-positive bacteria. Dual inhibition is attractive for widespread activity and avoidance of resistance as both encoding genes would have to acquire mutations.More related product prices
Sodium 1-heptanesulfonate Moxifloxacin Glycine Norfloxacin Lithium diisopropylamide Ciprofloxacin Ofloxacin 6-Aminocaproic acid Levofloxacin hydrochloride Rimantadine 112811-59-3 Heptane SITAFLOXACIN (1R,2S)-FLUOROCYCLOPROPYLAMINE TOSYLATE (1R,2R)-2-fluorocyclopropanecarboxylic acid Diphenyl phosphate 101513-77-3 trans-2-Fluoro-cyclopropanecarboxylic acid Ethyl 2,4,5-trifluorobenzoylacetateRelated product price
- Sodium 1-heptanesulfonate
₹306-137580 - Glycine
₹280-116823.4 - Norfloxacin
₹2140-15934.4






