Ferric sulfate
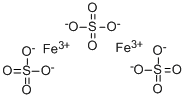
- CAS No.
- 10028-22-5
- Chemical Name:
- Ferric sulfate
- Synonyms
- Fe2(SO4)3;FERRIC SULPHATE;liusuantie;IRON (III) SULFATE;diiron tris(sulphate);Polymerized ferrous sulfate;NA-9121;FERIX-3;BETA FLOC;MONSEL SALT
- CBNumber:
- CB3496798
- Molecular Formula:
- Fe2O12S3
- Molecular Weight:
- 399.88
- MOL File:
- 10028-22-5.mol
- MSDS File:
- SDS
- Modify Date:
- 2024/3/14 15:18:26
| Melting point | 480°C |
|---|---|
| Boiling point | 101-118 °C |
| Density | 3.097 |
| vapor pressure | 0Pa at 20℃ |
| storage temp. | Keep in dark place,Inert atmosphere,Room temperature |
| solubility | slightly soluble in ethanol; insoluble in acetone |
| form | Powder |
| color | Yellow-gray |
| Water Solubility | Soluble in water. Sparingly soluble in alcohol. Almost insoluble in acetone and ethyl acetate. Insoluble in sulfuric acid and ammonia. |
| Sensitive | Hygroscopic |
| Merck | 14,4032 |
| Exposure limits |
ACGIH: TWA 1 mg/m3 NIOSH: TWA 1 mg/m3 |
| LogP | -1.031 (est) |
| CAS DataBase Reference | 10028-22-5(CAS DataBase Reference) |
| NIST Chemistry Reference | Ferric sulfate(10028-22-5) |
| EPA Substance Registry System | Ferric sulfate (10028-22-5) |
SAFETY
Risk and Safety Statements
| Symbol(GHS) |  GHS07 |
|||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Signal word | Warning | |||||||||
| Hazard statements | H315-H319-H335 | |||||||||
| Precautionary statements | P261-P280a-P304+P340-P305+P351+P338-P405-P501a | |||||||||
| Hazard Codes | Xn | |||||||||
| Risk Statements | 22-36/37 | |||||||||
| Safety Statements | 26 | |||||||||
| RIDADR | UN 9121 | |||||||||
| WGK Germany | 3 | |||||||||
| RTECS | NO8520000 | |||||||||
| TSCA | Yes | |||||||||
| HS Code | 2833298080 | |||||||||
| NFPA 704 |
|
Ferric sulfate price More Price(2)
| Manufacturer | Product number | Product description | CAS number | Packaging | Price | Updated | Buy |
|---|---|---|---|---|---|---|---|
| ALFA India | ALF-010717-14 | Iron(III) sulfate, Puratronic™, 99.998% (metals basis) | 10028-22-5 | 25g | ₹37598 | 2022-05-26 | Buy |
| ALFA India | ALF-010717-06 | Iron(III) sulfate, Puratronic™, 99.998% (metals basis) | 10028-22-5 | 5g | ₹9960 | 2022-05-26 | Buy |
Ferric sulfate Chemical Properties,Uses,Production
Description
§184.1307(a) Ferric sulfate (iron(HI)sulfate, Fe2(S04)3), a yellow substance that may be prepared by oxidizing iron(II)sulfate or by treating ferric oxide or ferric hydroxide with sulfuric acid.
Chemical Properties
Ferric Sulfate is a grayish-white powder or yellow lumpy crystals.
Physical properties
The anhydrous salt constitutes grayish-white rhombic crystals; hygroscopic; density 3.10 g/cm3; slightly soluble in cold water; decomposes in hot water. The nonahydrate is a yellow hexagonal crystalline substance; refractive index 1.54; density 2.10 g/cm3; hardness 2.5 Mohs; decomposes at 400°C; very soluble in water.
Uses
Ferric Sulfate is a nutrient and dietary supplement that is a source of iron.
Preparation
Iron(III) sulfate may be prepared by oxidation of iron(II) sulfate by hydrogen peroxide, nitric acid or any other suitable oxidizing agent. The reaction is carried out in sulfuric acid. Balanced molecular equations for the reactions with hydrogen peroxide and nitric acid are as follows:
2FeSO4 + H2SO4 + H2O2 → Fe2(SO4)3 + 2H2O
6FeSO4 + 3H2SO4 + 2HNO3 → 3Fe2(SO4)3 + 2NO + 4H2O
Even in the absence of an oxidizing agent, concentrated sulfuric acid alone can1 convert iron(II) sulfate to iron(III) sulfate: 2FeSO4 + 2H2SO4→ Fe2(SO4)3 + SO2 + 2H2O It also may be prepared by treating iron(III) oxide with sulfuric acid:
Fe2O3 + 3H2SO4 → Fe2(SO4)3 + 3H2O
.
Definition
Ferrous sulfate,also known as ferrisulpas, green copperas, green vitriol, iron sulfate, and melanterite, is composed of blue green monoclinic crystals. It is soluble in water and is used as a mordant for dyeing wool in the textile industry. Ferrous sulfate is also used as a disinfectant and in the manufacture of ink.
General Description
A yellow crystalline solid or a grayish-white powder. The primary hazard is the threat to the environment. Immediate steps should be taken to limit its spread to the environment. Ferric sulfate is used for water purification, and as a soil conditioner.
Air & Water Reactions
Soluble in water. Hygroscopic in air. Forms acidic aqueous solutions.
Reactivity Profile
Ferric sulfate is acidic. Corrosive to copper, copper alloys, mild steel, and galvanized steel [USCG, 1999].
Health Hazard
Inhalation of dust irritates nose and throat. Ingestion causes irritation of mouth and stomach. Dust irritates eyes and can irritate skin on prolonged contact.
Agricultural Uses
Herbicide, Molluscicide, Agricultural product constituent: Ferric sulfate is used on forage alfalfa, almonds, nurseries and structural pest control. This material is also used in pigments, textile dyeing, water treatment, and metal pickling
Trade name
GREENMASTER AUTUMN®; MAXICROP MOSS KILLER®; VITAX MICRO GRAN®; VITAX TURF TONIC®
Safety Profile
A poison by intraperitoneal route. Mutation data reported. When heated to decomposition it emits toxic fumes of SOx and Fe-. See also SULFATES and other ferric salts.
Potential Exposure
This material is used in pigments, textile dyeing, water treatment; and metal pickling.
Shipping
UN3077 Environmentally hazardous substances, solid, n.o.s., Hazard class: 9; Labels: 9-Miscellaneous haz ardous material, Technical Name Required.
Purification Methods
Dissolve the sulfate in the minimum volume of dilute aqueousH2SO4 and allow it to evaporate at room temperature until yellowish-white crystals start to form. Do not concentrate by boiling off the H2O as basic salts will be formed. Various hydrates are formed; the common ones are the dodeca and nona hydrates which are violet in colour. The anhydrous salt is colourless and is very hygroscopic, but it dissolves in H2O slowly unless ferrous sulfate is added. [Gmelin’s, Iron (8th edn) pp 439-462 1932.]
Incompatibilities
Hydrolyzed slowly in aqueous solution. Incompatible with magnesium, aluminum. Corrosive to copper and its alloys, mild and galvanized steel. Light sensitive.
Waste Disposal
Treat with soda ash or dilute NaOH. Separate any precipitate and landfill. Flush solution to sewer.
Ferric sulfate Preparation Products And Raw materials
Raw materials
Preparation Products
| Supplier | Tel | Country | ProdList | Advantage | Inquiry |
|---|---|---|---|---|---|
| S.V ENTERPRISES | +919322701159 | Mumbai, India | 150 | 58 | Inquiry |
| UNILOSA INTERNATINAL PRIVATE LIMITED | +91-9999069917 +91-9999069917 | New Delhi, India | 152 | 58 | Inquiry |
| Arihant Purechem Pvt Ltd | +91-9425195253 +91-9425195253 | Madhya Pradesh, India | 18 | 58 | Inquiry |
| Nithyasri Chemicals (APURVA CHEMICALS) | +91-2512692130 +91-9323652199 | Maharashtra, India | 75 | 58 | Inquiry |
| Cosmic Chemicals | +91-8806467677 +91-9324619871 | Maharashtra, India | 22 | 58 | Inquiry |
| 99 Exports | 91--9898946190 | Gujarat, India | 20 | 58 | Inquiry |
| Arihant Purechem Pvt., Ltd.(Formerly Arihant Chemicals) | 91-7412-260380 | Madhya Pradesh, India | 3 | 58 | Inquiry |
| MaxChem | 91--9825143737 | Gujarat, India | 22 | 58 | Inquiry |
| Archit Meta Chem | 08048371779Ext 300 | Ahmedabad, India | 64 | 58 | Inquiry |
| Canton Laboratories Pvt. Ltd. | 91-265-2638084 | Gujarat, India | 60 | 58 | Inquiry |
10028-22-5(Ferric sulfate )Related Search:
1of4
chevron_right




